Who Buys Torsemide from India — 232 Importers Behind a $18.2M Market
India's torsemide import market is served by 232 active buyers who collectively imported $18.2M across 1,997 shipments. MICRO NOVA PHARMACEUTICALS IND (NIGERIA) leads with a 6.1% market share, followed by AUROLOGISTICS LLC, (AUROBINDO PHARM and AUROLOGISTICS LLC. The top 5 buyers together control 20.9% of total import value, reflecting a moderately competitive buyer landscape.
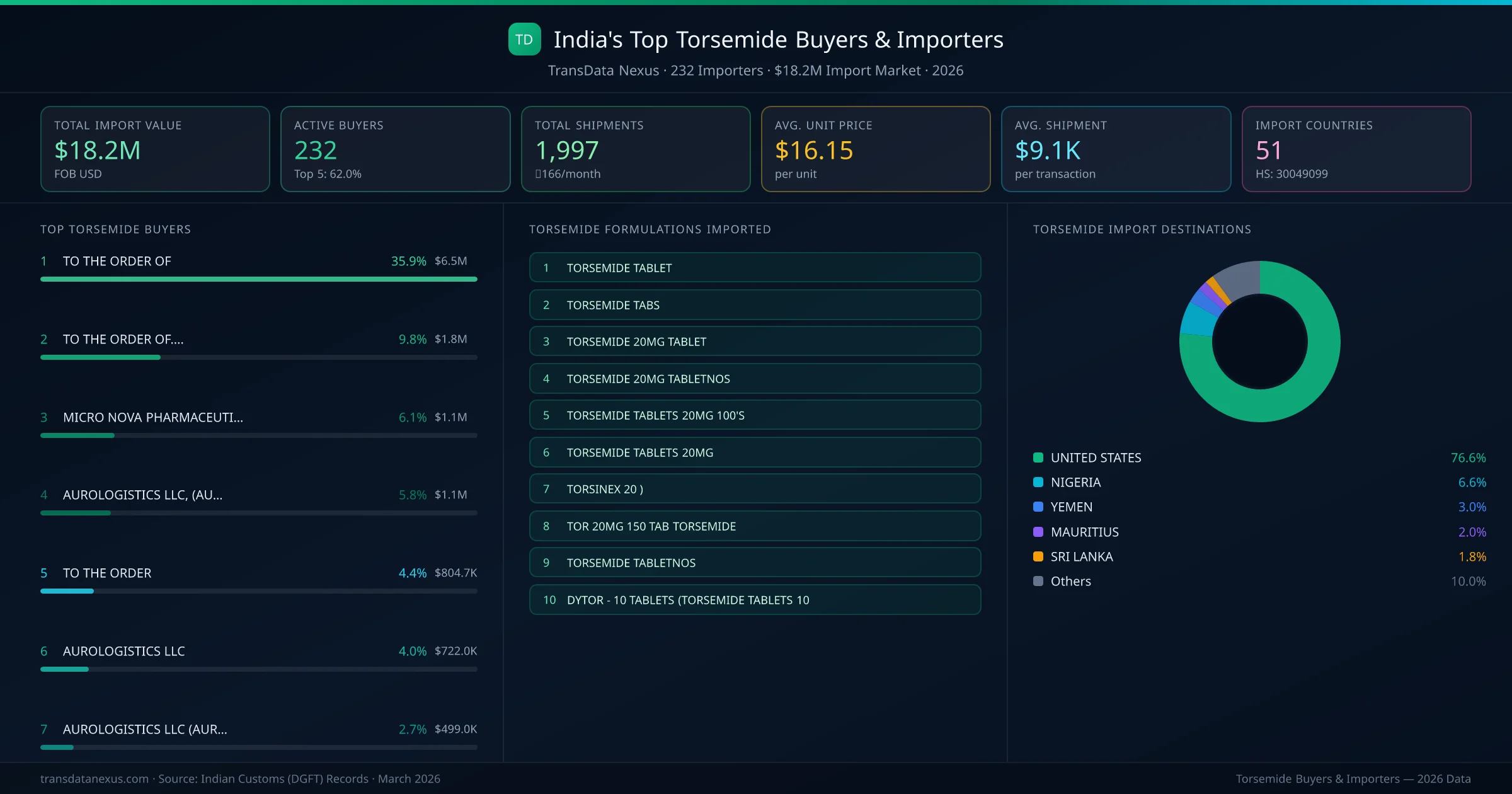
Top Torsemide Importers — Ranked by Import Value
MICRO NOVA PHARMACEUTICALS IND (NIGERIA) is the leading torsemide importer from India, holding a 6.1% share of the $18.2M market across 1,997 shipments from 232 buyers. The top 5 buyers — MICRO NOVA PHARMACEUTICALS IND (NIGERIA), AUROLOGISTICS LLC, (AUROBINDO PHARM (UNITED STATES), AUROLOGISTICS LLC (UNITED STATES), AUROLOGISTICS LLC (AUROBINDO PHARMA (UNITED STATES), CAMBER PHARMACEUTICALS INC (UNITED STATES) — collectively control 20.9% of total import value.
Top Torsemide Buyers & Importers
Ranked by import value · 232 active buyers · Indian Customs (DGFT) data
| # | Buyer & Formulations | Value | Suppliers | Share |
|---|---|---|---|---|
| 1 | MICRO NOVA PHARMACEUTICALS INDNIGERIA TORSINEX 20 TORSEMIDE 20MG TABLETS 3 XTORSINEX 20 - 3TORSINEX 20 TORSEMIDE 20MG TABLETS 3 X10'S | $1.1M | 5 | 6.1% |
| 2 | AUROLOGISTICS LLC, (AUROBINDO PHARMUNITED STATES TORSEMIDE TABLETTORSEMIDE TABSTORSEMIDE 20MG TABLET | $1.1M | 5 | 5.8% |
| 3 | AUROLOGISTICS LLCUNITED STATES TORSEMIDE TABLETTORSEMIDE TABSTORSEMIDE 20MG TABLET | $722.0K | 5 | 4.0% |
| 4 | AUROLOGISTICS LLC (AUROBINDO PHARMAUNITED STATES TORSEMIDE TABLETTORSEMIDE TABSTORSEMIDE 20MG TABLET | $499.0K | 5 | 2.7% |
| 5 | CAMBER PHARMACEUTICALS INCUNITED STATES TORSEMIDE TABLETTORSEMIDE TABSTORSEMIDE 20MG TABLET | $423.3K | 5 | 2.3% |
| 6 | CIPLAMEDICA PHARMACEUTICAL INDUSTRIYEMEN DYTOR 10 MG TABLETS | $383.3K | 5 | 2.1% |
| 7 | REGENCY PHARMA LIMITEDMAURITIUS TOR 20MG 150 TAB TORSEMIDEDYTOR 10MG 1500 TAB TORSEMIDETOR 10MG 150 TAB TORSEMIDE USP | $308.1K | 5 | 1.7% |
| 8 | AUROLOGISTICS LLC AUROBINDO PHARMUNITED STATES TORSEMIDE TABLETTORSEMIDE TABSTORSEMIDE 20MG TABLET | $285.8K | 5 | 1.6% |
| 9 | AUROLOGISTICS LLC(AUROBINDO PHARMAUNITED STATES TORSEMIDE TABLETTORSEMIDE TABSTORSEMIDE 20MG TABLET | $279.0K | 5 | 1.5% |
| 10 | BREATHEFREE LANKA PVT LTDSRI LANKA DYTOR - 10 TABLETS (TORSEMIDE TABLETS 10DYTOR 10 MG TABLETSDYTOR-10 TABLETS TORSEMIDE TABLETS 10 M | $250.8K | 5 | 1.4% |
| 11 | AUROBINDO PHARMA USA.INC.,.UNITED STATES TORSEMIDE TABLETTORSEMIDE TABSTORSEMIDE 20MG TABLET | $187.4K | 5 | 1.0% |
| 12 | AUROBINDO PHARMA (PTY) LTD,SOUTH AFRICA TORSEMIDE TABLETTORSEMIDE TABSTORSEMIDE 20MG TABLET | $172.8K | 5 | 0.9% |
| 13 | E.P.DISFRANCE TORSINEX 20 )TORSINEX 10 TORSEMIDE TABLETS 10MG 3X10STORSINEX 10 ) | $140.6K | 5 | 0.8% |
| 14 | MIRTRFZUNITED ARAB EMIRATES | $111.2K | 5 | 0.6% |
| 15 | E.P.DIS.,.,FRANCE TORSINEX 20 )TORSINEX 10 TORSEMIDE TABLETS 10MG 3X10STORSINEX 10 ) | $106.8K | 5 | 0.6% |
Related Analysis
Market Analysis: Top Importing Countries & Importers
Comprehensive geographic market intelligence dashboard
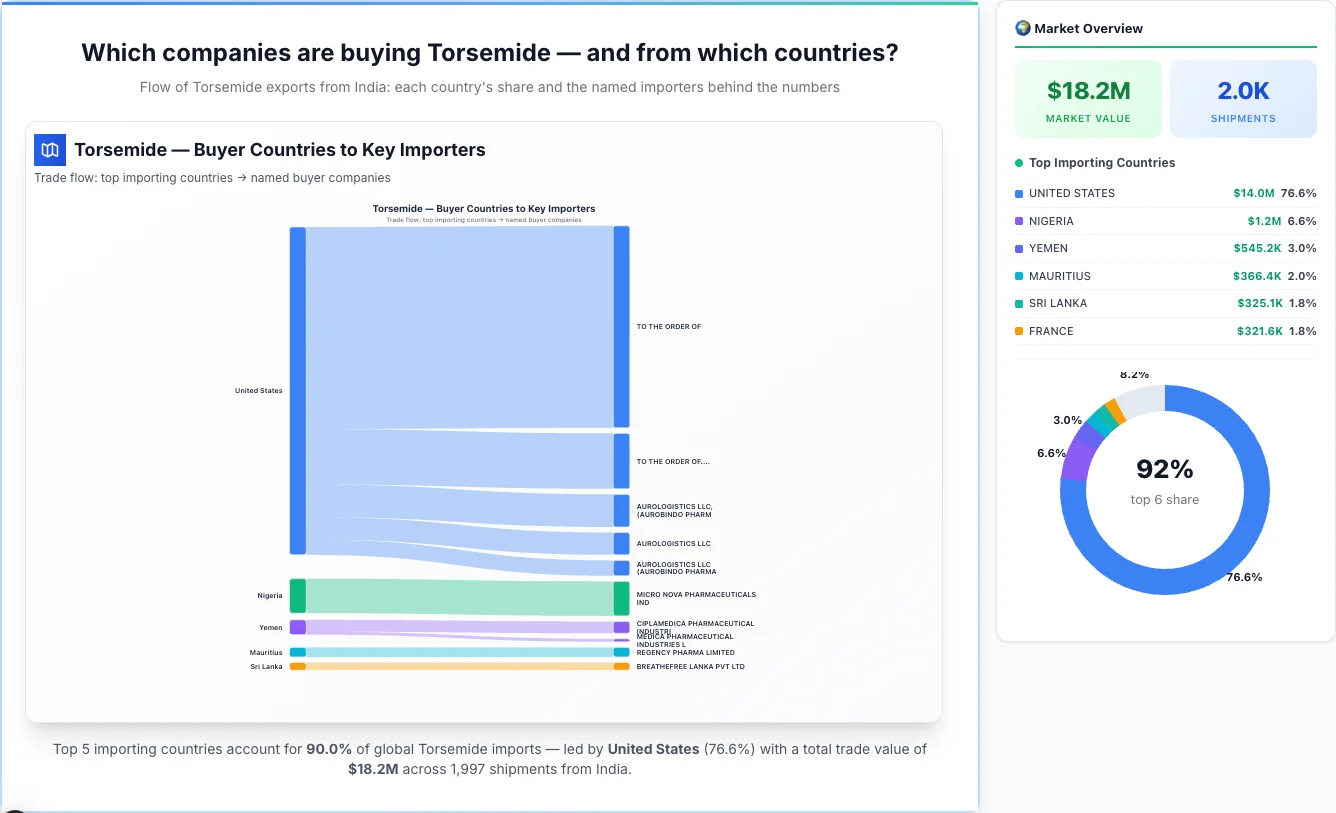
Which companies are buying Torsemide — and from which countries?
Flow of Torsemide exports from India: each country's share and the named importers behind the numbers
Torsemide — Buyer Countries to Key Importers
Trade flow: top importing countries → named buyer companies
Market Analysis: Top Importing Countries & Their Importers
1United States
United States emerges as the first largest importing country, achieving a total trade value of $14.0M through 138 shipments. This represents a market share of 76.6% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: United States has a diverse importer base, with their top importers including:
• AUROLOGISTICS LLC, (AUROBINDO PHARM - $1.1M (7.6% of country's total business)
• AUROLOGISTICS LLC - $722.0K (5.2% of country's total business)
• AUROLOGISTICS LLC (AUROBINDO PHARMA - $499.0K (3.6% of country's total business)
• CAMBER PHARMACEUTICALS INC - $423.3K (3.0% of country's total business)
• AUROLOGISTICS LLC AUROBINDO PHARM - $285.8K (2.0% of country's total business)
• Rest of United States's importers - $11.0M (78.6% of country's total business)
Key Insight: United States demonstrates strong market positioning with an average shipment value of $101.3K, indicating premium pricing strategy compared to the market average of $9.1K.
United States is a key market for Torsemide imports, representing 76.6% of total trade value.
2Nigeria
Nigeria emerges as the second largest importing country, achieving a total trade value of $1.2M through 34 shipments. This represents a market share of 6.6% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Nigeria has a diverse importer base, with their top importers including:
• MICRO NOVA PHARMACEUTICALS IND - $1.1M (92.6% of country's total business)
Key Insight: Nigeria demonstrates strong market positioning with an average shipment value of $35.5K, indicating premium pricing strategy compared to the market average of $9.1K.
Nigeria is a key market for Torsemide imports, representing 6.6% of total trade value.
3Yemen
Yemen emerges as the third largest importing country, achieving a total trade value of $545.2K through 15 shipments. This represents a market share of 3.0% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Yemen has a diverse importer base, with their top importers including:
• CIPLAMEDICA PHARMACEUTICAL INDUSTRI - $383.3K (70.3% of country's total business)
• MEDICA PHARMACEUTICAL INDUSTRIES L - $97.5K (17.9% of country's total business)
Key Insight: Yemen demonstrates strong market positioning with an average shipment value of $36.3K, indicating premium pricing strategy compared to the market average of $9.1K.
Yemen is a key market for Torsemide imports, representing 3.0% of total trade value.
4Mauritius
Mauritius emerges as the fourth largest importing country, achieving a total trade value of $366.4K through 417 shipments. This represents a market share of 2.0% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Mauritius has a diverse importer base, with their top importers including:
• REGENCY PHARMA LIMITED - $308.1K (84.1% of country's total business)
Key Insight: Mauritius demonstrates strong market positioning with an average shipment value of $879, indicating competitive pricing strategy compared to the market average of $9.1K.
Mauritius is a key market for Torsemide imports, representing 2.0% of total trade value.
5Sri Lanka
Sri Lanka emerges as the fifth largest importing country, achieving a total trade value of $325.1K through 27 shipments. This represents a market share of 1.8% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Sri Lanka has a diverse importer base, with their top importers including:
• BREATHEFREE LANKA PVT LTD - $250.8K (77.1% of country's total business)
Key Insight: Sri Lanka demonstrates strong market positioning with an average shipment value of $12.0K, indicating premium pricing strategy compared to the market average of $9.1K.
Sri Lanka is a key market for Torsemide imports, representing 1.8% of total trade value.
Buyer Segment Analysis
TransData Nexus Torsemide buyer market intelligence
1Buyer Segment Analysis
The importers of Torsemide from India can be categorized into several key segments:
1. Distributors: These entities purchase large quantities of Torsemide to supply to various healthcare providers, pharmacies, and retailers within their respective countries. For instance, MICRO NOVA PHARMACEUTICALS IND in Nigeria, which imported Torsemide worth $1.12 million, likely operates as a distributor, ensuring widespread availability of the medication across the Nigerian market.
2. Contract Manufacturers: Companies that import Torsemide for the purpose of further manufacturing or repackaging before distribution. AUROLOGISTICS LLC, associated with AUROBINDO PHARMA in the United States, appears multiple times among the top buyers, with combined imports totaling approximately $2.57 million. This suggests a role in contract manufacturing or distribution within the U.S. pharmaceutical supply chain.
3. Retail Chains and Hospital Groups: While specific data on retail chains and hospital groups is not detailed in the provided information, it's common for such entities to import pharmaceuticals directly to meet the needs of their patients and customers.
4. Government Procurement Agencies: These agencies import medications to supply public healthcare facilities. The data does not specify any government procurement agencies among the top buyers of Torsemide from India.
5. Re-exporters: Companies that import pharmaceuticals with the intention of exporting them to other markets. The data does not provide specific information on re-exporters in the context of Torsemide imports.
Each segment exhibits distinct purchasing behaviors. Distributors and contract manufacturers often place large, recurring orders to maintain a steady supply chain, as evidenced by the 64.2% repeat buyer rate among the 232 buyers. Retail chains and hospital groups may order based on patient demand and inventory levels, leading to variable order sizes and frequencies. Government agencies typically procure through tenders, resulting in bulk purchases aligned with budget cycles and public health initiatives. Re-exporters' purchasing patterns depend on the demand in their target export markets.
Country-Specific Import Regulations
TransData Nexus Torsemide buyer market intelligence
1Country-Specific Import Regulations
United States
1. Drug Regulatory Authority: U.S. Food and Drug Administration (FDA).
2. Import Registration Process for Torsemide: Importers must ensure that Torsemide is FDA-approved. The process involves submitting an Abbreviated New Drug Application (ANDA) for generic drugs, demonstrating bioequivalence to the brand-name counterpart.
3. Estimated Timeline for Registration: The FDA review process for an ANDA typically takes 10 months, though it can vary based on the completeness of the application and FDA workload.
4. Import Tariff/Duty for HS 30049099: Pharmaceutical products under HS Code 30049099 are generally duty-free in the United States.
Nigeria
1. Drug Regulatory Authority: National Agency for Food and Drug Administration and Control (NAFDAC).
2. Import Registration Process for Torsemide: Importers must register the product with NAFDAC, providing documentation such as a Certificate of Pharmaceutical Product (CPP), Good Manufacturing Practice (GMP) certificate, and samples for laboratory analysis.
3. Estimated Timeline for Registration: The registration process can take approximately 6 to 12 months, depending on the completeness of the submission and NAFDAC's review process.
4. Import Tariff/Duty for HS 30049099: Pharmaceutical products under this HS code are subject to a 5% import duty in Nigeria.
Yemen
1. Drug Regulatory Authority: Supreme Board of Drugs and Medical Appliances (SBDMA).
2. Import Registration Process for Torsemide: Importers are required to submit a registration dossier to the SBDMA, including product samples, manufacturing licenses, and quality certificates.
3. Estimated Timeline for Registration: The registration process typically takes 6 to 9 months, contingent upon the thoroughness of the application and the SBDMA's evaluation procedures.
4. Import Tariff/Duty for HS 30049099: Yemen imposes a 5% import duty on pharmaceutical products classified under this HS code.
Mauritius
1. Drug Regulatory Authority: Pharmacy Board of Mauritius.
2. Import Registration Process for Torsemide: Importers must obtain approval from the Pharmacy Board by submitting a detailed application, including product specifications, manufacturing details, and evidence of registration in the country of origin.
3. Estimated Timeline for Registration: The approval process usually takes between 3 to 6 months, depending on the completeness of the application and the Board's review schedule.
4. Import Tariff/Duty for HS 30049099: Pharmaceuticals under this HS code are generally exempt from import duties in Mauritius.
Sri Lanka
1. Drug Regulatory Authority: National Medicines Regulatory Authority (NMRA).
2. Import Registration Process for Torsemide: Importers are required to register the product with the NMRA, providing comprehensive documentation, including a Certificate of Pharmaceutical Product (CPP), GMP certificate, and product samples.
3. Estimated Timeline for Registration: The registration process can take approximately 6 to 12 months, depending on the completeness of the submission and the NMRA's review process.
4. Import Tariff/Duty for HS 30049099: Sri Lanka imposes a 0% import duty on pharmaceutical products under this HS code.
Demand Drivers & Market Opportunity
TransData Nexus Torsemide buyer market intelligence
1Demand Drivers & Market Opportunity
The demand for Torsemide in importing countries is primarily driven by the prevalence of cardiovascular diseases, particularly heart failure and hypertension, conditions for which Torsemide is commonly prescribed. According to the World Health Organization (WHO), cardiovascular diseases remain the leading cause of death globally, accounting for approximately 17.9 million deaths annually as of 2024. This high disease burden necessitates the availability of effective diuretics like Torsemide.
Government healthcare programs and the expansion of universal health coverage also significantly influence the demand for essential medicines. Many countries have included Torsemide in their national Essential Medicines Lists, aligning with the WHO's Model List of Essential Medicines, which emphasizes the importance of such medications in addressing public health needs. Additionally, procurement through international organizations and tender-based purchasing mechanisms ensures a steady demand. For example, the WHO's Prequalification Programme facilitates the procurement of quality-assured medicines by various countries, thereby supporting consistent demand for drugs like Torsemide. (iris.who.int)
The presence of 232 buyers across 51 countries, with a repeat buyer rate of 64.2%, underscores the sustained global demand for Torsemide. This consistent purchasing pattern reflects the medication's critical role in managing cardiovascular conditions worldwide.
Common Questions — Torsemide Buyers & Importers
Answers based on Indian Customs (DGFT) shipment records compiled by TransData Nexus
QWho is the largest torsemide buyer importing from India?
Based on import volume and value, MICRO NOVA PHARMACEUTICALS IND (NIGERIA) leads with $1.1M in imports and a 6.1% market share — the highest of any single torsemide importer. AUROLOGISTICS LLC, (AUROBINDO PHARM (UNITED STATES) and AUROLOGISTICS LLC (UNITED STATES) are the next largest buyers.
QHow many companies buy torsemide from India?
There are 232 active torsemide buyers importing from India, with a combined market of $18.2M across 1,997 shipments to 51 countries. The top 5 buyers hold 20.9% of total import value, while the remaining 227 buyers handle the other 79.1%.
QWhich countries import the most torsemide from India?
The top importing countries for torsemide from India are United States (76.6%), Nigeria (6.6%), Yemen (3.0%), Mauritius (2.0%), Sri Lanka (1.8%). These markets represent the largest demand centres for Indian pharmaceutical exports of torsemide, with buyers ranging from government procurement agencies to private pharmaceutical distributors.
QWhat is the average import order value for torsemide from India?
The average import transaction value for torsemide from India is $9.1K, with an average unit price of $16.15 per unit. Order sizes vary significantly based on buyer type — government tenders tend to be larger while private distributors place more frequent smaller orders.
Buyer Segment Analysis
TransData Nexus Torsemide buyer market intelligence
1Buyer Segment Analysis
The importers of Torsemide from India can be categorized into several key segments:
1. Distributors: These entities purchase large quantities of Torsemide to supply to various healthcare providers, pharmacies, and retailers within their respective countries. For instance, MICRO NOVA PHARMACEUTICALS IND in Nigeria, which imported Torsemide worth $1.12 million, likely operates as a distributor, ensuring widespread availability of the medication across the Nigerian market.
2. Contract Manufacturers: Companies that import Torsemide for the purpose of further manufacturing or repackaging before distribution. AUROLOGISTICS LLC, associated with AUROBINDO PHARMA in the United States, appears multiple times among the top buyers, with combined imports totaling approximately $2.57 million. This suggests a role in contract manufacturing or distribution within the U.S. pharmaceutical supply chain.
3. Retail Chains and Hospital Groups: While specific data on retail chains and hospital groups is not detailed in the provided information, it's common for such entities to import pharmaceuticals directly to meet the needs of their patients and customers.
4. Government Procurement Agencies: These agencies import medications to supply public healthcare facilities. The data does not specify any government procurement agencies among the top buyers of Torsemide from India.
5. Re-exporters: Companies that import pharmaceuticals with the intention of exporting them to other markets. The data does not provide specific information on re-exporters in the context of Torsemide imports.
Each segment exhibits distinct purchasing behaviors. Distributors and contract manufacturers often place large, recurring orders to maintain a steady supply chain, as evidenced by the 64.2% repeat buyer rate among the 232 buyers. Retail chains and hospital groups may order based on patient demand and inventory levels, leading to variable order sizes and frequencies. Government agencies typically procure through tenders, resulting in bulk purchases aligned with budget cycles and public health initiatives. Re-exporters' purchasing patterns depend on the demand in their target export markets.
Country-Specific Import Regulations
TransData Nexus Torsemide buyer market intelligence
1Country-Specific Import Regulations
United States
1. Drug Regulatory Authority: U.S. Food and Drug Administration (FDA).
2. Import Registration Process for Torsemide: Importers must ensure that Torsemide is FDA-approved. The process involves submitting an Abbreviated New Drug Application (ANDA) for generic drugs, demonstrating bioequivalence to the brand-name counterpart.
3. Estimated Timeline for Registration: The FDA review process for an ANDA typically takes 10 months, though it can vary based on the completeness of the application and FDA workload.
4. Import Tariff/Duty for HS 30049099: Pharmaceutical products under HS Code 30049099 are generally duty-free in the United States.
Nigeria
1. Drug Regulatory Authority: National Agency for Food and Drug Administration and Control (NAFDAC).
2. Import Registration Process for Torsemide: Importers must register the product with NAFDAC, providing documentation such as a Certificate of Pharmaceutical Product (CPP), Good Manufacturing Practice (GMP) certificate, and samples for laboratory analysis.
3. Estimated Timeline for Registration: The registration process can take approximately 6 to 12 months, depending on the completeness of the submission and NAFDAC's review process.
4. Import Tariff/Duty for HS 30049099: Pharmaceutical products under this HS code are subject to a 5% import duty in Nigeria.
Yemen
1. Drug Regulatory Authority: Supreme Board of Drugs and Medical Appliances (SBDMA).
2. Import Registration Process for Torsemide: Importers are required to submit a registration dossier to the SBDMA, including product samples, manufacturing licenses, and quality certificates.
3. Estimated Timeline for Registration: The registration process typically takes 6 to 9 months, contingent upon the thoroughness of the application and the SBDMA's evaluation procedures.
4. Import Tariff/Duty for HS 30049099: Yemen imposes a 5% import duty on pharmaceutical products classified under this HS code.
Mauritius
1. Drug Regulatory Authority: Pharmacy Board of Mauritius.
2. Import Registration Process for Torsemide: Importers must obtain approval from the Pharmacy Board by submitting a detailed application, including product specifications, manufacturing details, and evidence of registration in the country of origin.
3. Estimated Timeline for Registration: The approval process usually takes between 3 to 6 months, depending on the completeness of the application and the Board's review schedule.
4. Import Tariff/Duty for HS 30049099: Pharmaceuticals under this HS code are generally exempt from import duties in Mauritius.
Sri Lanka
1. Drug Regulatory Authority: National Medicines Regulatory Authority (NMRA).
2. Import Registration Process for Torsemide: Importers are required to register the product with the NMRA, providing comprehensive documentation, including a Certificate of Pharmaceutical Product (CPP), GMP certificate, and product samples.
3. Estimated Timeline for Registration: The registration process can take approximately 6 to 12 months, depending on the completeness of the submission and the NMRA's review process.
4. Import Tariff/Duty for HS 30049099: Sri Lanka imposes a 0% import duty on pharmaceutical products under this HS code.
Demand Drivers & Market Opportunity
TransData Nexus Torsemide buyer market intelligence
1Demand Drivers & Market Opportunity
The demand for Torsemide in importing countries is primarily driven by the prevalence of cardiovascular diseases, particularly heart failure and hypertension, conditions for which Torsemide is commonly prescribed. According to the World Health Organization (WHO), cardiovascular diseases remain the leading cause of death globally, accounting for approximately 17.9 million deaths annually as of 2024. This high disease burden necessitates the availability of effective diuretics like Torsemide.
Government healthcare programs and the expansion of universal health coverage also significantly influence the demand for essential medicines. Many countries have included Torsemide in their national Essential Medicines Lists, aligning with the WHO's Model List of Essential Medicines, which emphasizes the importance of such medications in addressing public health needs. Additionally, procurement through international organizations and tender-based purchasing mechanisms ensures a steady demand. For example, the WHO's Prequalification Programme facilitates the procurement of quality-assured medicines by various countries, thereby supporting consistent demand for drugs like Torsemide. (iris.who.int)
The presence of 232 buyers across 51 countries, with a repeat buyer rate of 64.2%, underscores the sustained global demand for Torsemide. This consistent purchasing pattern reflects the medication's critical role in managing cardiovascular conditions worldwide.
Official References & Regulatory Resources
- European Medicines Agency
- WHO Essential Medicines
- Invest India — Pharma Sector
- India Trade Statistics (DGFT)
Verify import regulations and drug registration requirements with the official agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Identification: 232 global importers of Torsemide identified from consignee fields in DGFT shipping bill records.
- 2.Import Value Analysis: Total import value aggregated from 1,997 individual shipping bill records. Values are FOB in USD.
- 3.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 4.Geographic Distribution: Buyer imports distributed across 51 destination countries with market share per buyer calculated from capped values.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
1,997 Verified Shipments
232 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists