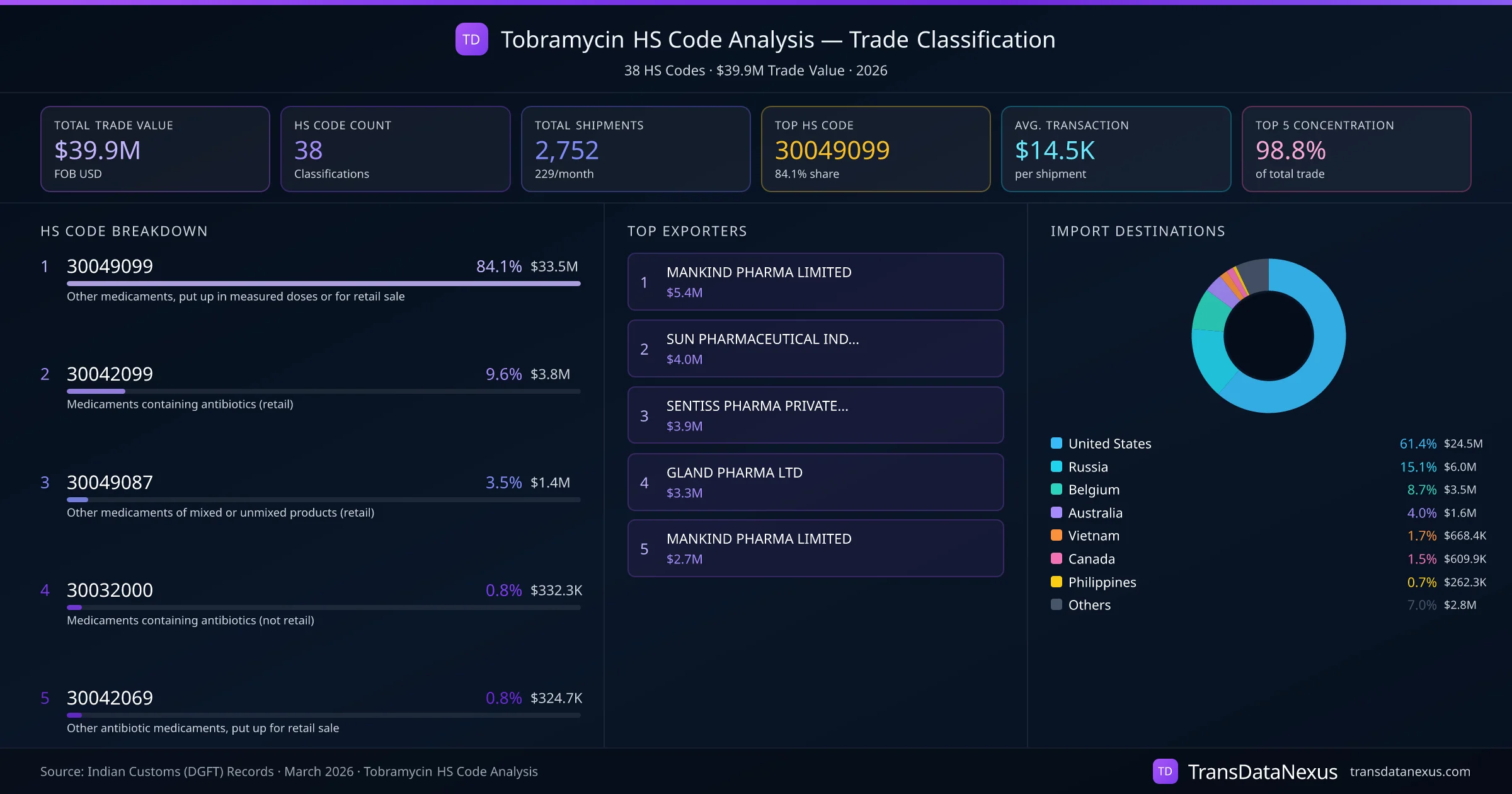
Tobramycin can be classified under HS codes 3003 or 3004, depending on its form and intended use:
- HS Code 3003: This code covers "Medicaments (excluding goods of heading 30.02, 30.05, or 30.06) consisting of two or more constituents which have been mixed together for therapeutic or prophylactic uses, not put up in measured doses or in forms or packings for retail sale."
- HS Code 3004: This code includes "Medicaments (excluding goods of heading 30.02, 30.05, or 30.06) consisting of mixed or unmixed products for therapeutic or prophylactic uses, put up in measured doses (including those in the form of transdermal administration systems) or in forms or packings for retail sale."
Common misclassification errors occur when the packaging and dosing form are not accurately considered. For instance, bulk Tobramycin intended for further processing should be classified under HS code 3003, while finished products ready for retail sale fall under HS code 3004. Misclassification can lead to customs penalties, including fines and delays in shipment clearance. Our data indicates that 84.1% of Tobramycin shipments are classified under HS code 30049099, suggesting a predominant classification as finished products for retail sale.
Recent rulings by the World Customs Organization (WCO) and national customs authorities emphasize the importance of accurate classification based on product form and packaging. Importers and exporters should ensure compliance by thoroughly reviewing product specifications and consulting with customs experts when necessary.