Who Buys Teicoplanin from India — 108 Importers Behind a $6.3M Market
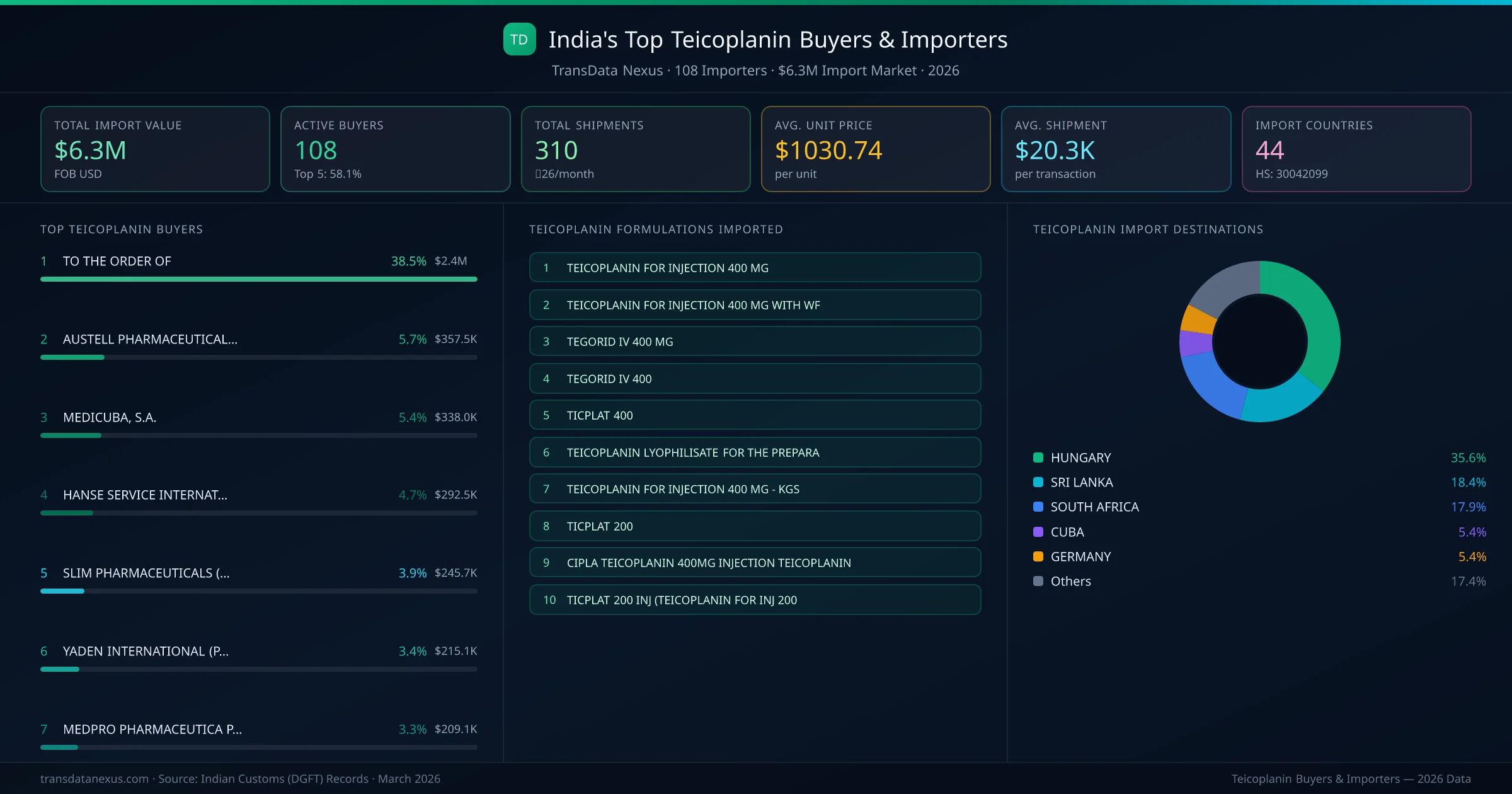
India's teicoplanin import market is served by 108 active buyers who collectively imported $6.3M across 310 shipments. AUSTELL PHARMACEUTICALS ( PTY) LTD (SOUTH AFRICA) leads with a 5.7% market share, followed by MEDICUBA, S.A. and HANSE SERVICE INTERNATIONALE. The top 5 buyers together control 23.0% of total import value, reflecting a moderately competitive buyer landscape.
Top Teicoplanin Importers — Ranked by Import Value
AUSTELL PHARMACEUTICALS ( PTY) LTD (SOUTH AFRICA) is the leading teicoplanin importer from India, holding a 5.7% share of the $6.3M market across 310 shipments from 108 buyers. The top 5 buyers — AUSTELL PHARMACEUTICALS ( PTY) LTD (SOUTH AFRICA), MEDICUBA, S.A. (CUBA), HANSE SERVICE INTERNATIONALE (GERMANY), SLIM PHARMACEUTICALS (PVT) LTD, (SRI LANKA), YADEN INTERNATIONAL (PVT) LTD (SRI LANKA) — collectively control 23.0% of total import value.
Top Teicoplanin Buyers & Importers
Ranked by import value · 108 active buyers · Indian Customs (DGFT) data
| # | Buyer & Formulations | Value | Suppliers | Share |
|---|---|---|---|---|
| 1 | AUSTELL PHARMACEUTICALS ( PTY) LTDSOUTH AFRICA TEICOPLANIN FOR INJECTION 400 MG , 64420 VIALS, PACK SIZE-TEGORID IV 400 MGTEGORID IV 400 | $357.5K | 5 | 5.7% |
| 2 | MEDICUBA, S.A.CUBA TEICOPLANIN FOR INJECTION 400 MG WITH WF | $338.0K | 5 | 5.4% |
| 3 | HANSE SERVICE INTERNATIONALEGERMANY TEICOPLANIN EBERTH 400 MG PACK SIZE 1X1 S VLS 24000 VLSTEICOPLANIN FOR INJECTION 400 MGTEICOPLANIN EBERTH 200 MG PACK SIZE 1X1 S VLS 9000 VLS | $292.5K | 5 | 4.7% |
| 4 | SLIM PHARMACEUTICALS (PVT) LTD,SRI LANKA TEICOPLANIN FOR INJECTION 400 MGTICPLAT 200 INJ (TEICOPLANIN FOR INJ 200TICPLAT 400 INJ (TEICOPLANIN FOR INJ 400 | $245.7K | 5 | 3.9% |
| 5 | YADEN INTERNATIONAL (PVT) LTDSRI LANKA TEICOPLANIN FOR INJECTION 400 MGTICPLAT 200 INJ (TEICOPLANIN FOR INJ 200TICPLAT 400 INJ (TEICOPLANIN FOR INJ 400 | $215.1K | 5 | 3.4% |
| 6 | MEDPRO PHARMACEUTICA PTY LTDSOUTH AFRICA TEICOPLANIN FOR INJECTION 400 MG , 64420 VIALS, PACK SIZE-TEGORID IV 400 MGTEGORID IV 400 | $209.1K | 5 | 3.3% |
| 7 | PEOPLE'S BANK,SRI LANKA TEICOPLANIN FOR INJECTION 400 MGTICPLAT 200 INJ (TEICOPLANIN FOR INJ 200TICPLAT 400 INJ (TEICOPLANIN FOR INJ 400 | $197.5K | 5 | 3.1% |
| 8 | AUSTELL PHARMACEUTICALS (PTY) LTD,SOUTH AFRICA TEICOPLANIN FOR INJECTION 400 MG , 64420 VIALS, PACK SIZE-TEGORID IV 400 MGTEGORID IV 400 | $195.1K | 5 | 3.1% |
| 9 | AUSTELL PHARMACEUTICALS ( PTY) LIMITEDSOUTH AFRICA TEICOPLANIN FOR INJECTION 400 MG , 64420 VIALS, PACK SIZE-TEGORID IV 400 MGTEGORID IV 400 | $168.6K | 5 | 2.7% |
| 10 | M/S. OOO JODAS EXPOIM..RUSSIA TEICOPLANIN LYOPHILISATE FOR THE PREPARATARGOCID INJ 400 MG B NO:2TARGOCID INJ 200 MG B NO:2 | $160.3K | 5 | 2.5% |
| 11 | BANK OF CEYLONSRI LANKA TEICOPLANIN FOR INJECTION 400 MGTICPLAT 200 INJ (TEICOPLANIN FOR INJ 200TICPLAT 400 INJ (TEICOPLANIN FOR INJ 400 | $113.2K | 5 | 1.8% |
| 12 | HATTON NATIONAL BANK PLC,SRI LANKA TEICOPLANIN FOR INJECTION 400 MGTICPLAT 200 INJ (TEICOPLANIN FOR INJ 200TICPLAT 400 INJ (TEICOPLANIN FOR INJ 400 | $110.5K | 5 | 1.8% |
| 13 | YETI PHARMACHEM DISTRIBUTORS PVT LTNEPAL TARGOCID INJ 400 MG B NO:2TARGOCID INJ 200 MG B NO:2TARGOCID INJ 400 MG TEICOPLANIN B NO 2J3261 | $101.2K | 5 | 1.6% |
| 14 | OOOJODAS EXPOIMRUSSIA TEICOPLANIN LYOPHILISATE FOR THE PREPARATEICOPLANIN LYOPHILISATE FOR PREPARATION | $96.7K | 5 | 1.5% |
| 15 | LEADERSHIP SARLLEBANON CELPLANIN 200 MG TEICOPLANIN INJECTION 200MGCELPLANIN 400 MG TEICOPLANIN INJECTION 400MGTEICOPLANIN FOR INJECTION TECOCIDE 400MG | $74.6K | 5 | 1.2% |
Related Analysis
Market Analysis: Top Importing Countries & Importers
Comprehensive geographic market intelligence dashboard
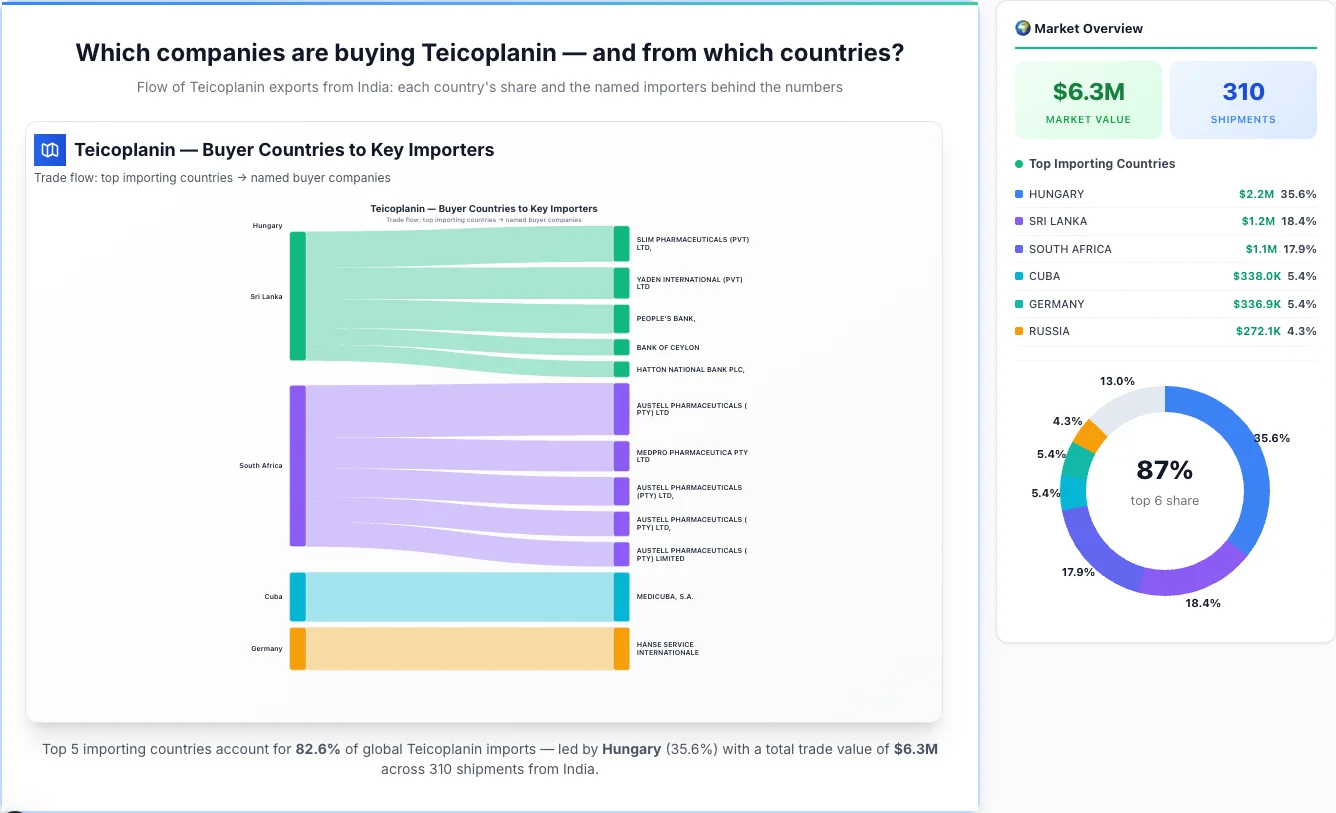
Which companies are buying Teicoplanin — and from which countries?
Flow of Teicoplanin exports from India: each country's share and the named importers behind the numbers
Teicoplanin — Buyer Countries to Key Importers
Trade flow: top importing countries → named buyer companies
Market Analysis: Top Importing Countries & Their Importers
1Hungary
Hungary emerges as the first largest importing country, achieving a total trade value of $2.2M through 0 shipments. This represents a market share of 35.6% of the total market activity, demonstrating their significant presence in the industry.
Key Insight: Hungary demonstrates strong market positioning with an average shipment value of $2.2M, indicating premium pricing strategy compared to the market average of $20.3K.
Hungary is a key market for Teicoplanin imports, representing 35.6% of total trade value.
2Sri Lanka
Sri Lanka emerges as the second largest importing country, achieving a total trade value of $1.2M through 28 shipments. This represents a market share of 18.4% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Sri Lanka has a diverse importer base, with their top importers including:
• SLIM PHARMACEUTICALS (PVT) LTD, - $245.7K (21.2% of country's total business)
• YADEN INTERNATIONAL (PVT) LTD - $215.1K (18.6% of country's total business)
• PEOPLE'S BANK, - $197.5K (17.0% of country's total business)
• BANK OF CEYLON - $113.2K (9.8% of country's total business)
• HATTON NATIONAL BANK PLC, - $110.5K (9.5% of country's total business)
• Rest of Sri Lanka's importers - $276.4K (23.9% of country's total business)
Key Insight: Sri Lanka demonstrates strong market positioning with an average shipment value of $41.4K, indicating premium pricing strategy compared to the market average of $20.3K.
Sri Lanka is a key market for Teicoplanin imports, representing 18.4% of total trade value.
3South Africa
South Africa emerges as the third largest importing country, achieving a total trade value of $1.1M through 15 shipments. This represents a market share of 17.9% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: South Africa has a diverse importer base, with their top importers including:
• AUSTELL PHARMACEUTICALS ( PTY) LTD - $357.5K (31.7% of country's total business)
• MEDPRO PHARMACEUTICA PTY LTD - $209.1K (18.6% of country's total business)
• AUSTELL PHARMACEUTICALS (PTY) LTD, - $195.1K (17.3% of country's total business)
• AUSTELL PHARMACEUTICALS ( PTY) LIMITED - $168.6K (15.0% of country's total business)
Key Insight: South Africa demonstrates strong market positioning with an average shipment value of $75.1K, indicating premium pricing strategy compared to the market average of $20.3K.
South Africa is a key market for Teicoplanin imports, representing 17.9% of total trade value.
4Cuba
Cuba emerges as the fourth largest importing country, achieving a total trade value of $338.0K through 2 shipments. This represents a market share of 5.4% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Cuba has a diverse importer base, with their top importers including:
• MEDICUBA, S.A. - $338.0K (100.0% of country's total business)
Key Insight: Cuba demonstrates strong market positioning with an average shipment value of $169.0K, indicating premium pricing strategy compared to the market average of $20.3K.
Cuba is a key market for Teicoplanin imports, representing 5.4% of total trade value.
5Germany
Germany emerges as the fifth largest importing country, achieving a total trade value of $336.9K through 3 shipments. This represents a market share of 5.4% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Germany has a diverse importer base, with their top importers including:
• HANSE SERVICE INTERNATIONALE - $292.5K (86.8% of country's total business)
Key Insight: Germany demonstrates strong market positioning with an average shipment value of $112.3K, indicating premium pricing strategy compared to the market average of $20.3K.
Germany is a key market for Teicoplanin imports, representing 5.4% of total trade value.
Buyer Segment Analysis
TransData Nexus Teicoplanin buyer market intelligence
1Buyer Segment Analysis
The importation of Teicoplanin from India involves various buyer segments, each with distinct purchasing behaviors and order patterns.
Distributors: Entities such as Austell Pharmaceuticals (Pty) Ltd in South Africa and Hanse Service Internationale in Germany function as distributors. They procure Teicoplanin in bulk to supply hospitals, clinics, and pharmacies within their respective regions. Their purchasing behavior is characterized by large-volume orders, often through tender processes, to ensure a steady supply chain and meet the demands of multiple healthcare providers.
Government Procurement Agencies: Organizations like Medicuba S.A. in Cuba and People's Bank in Sri Lanka represent government procurement agencies. They acquire Teicoplanin to distribute within public healthcare systems, often through national tenders or agreements. Their order patterns are typically large and periodic, aligning with government budget cycles and healthcare program needs.
Hospital Groups: Slim Pharmaceuticals (Pvt) Ltd and Yaden International (Pvt) Ltd in Sri Lanka, as well as Medpro Pharmaceutica Pty Ltd in South Africa, are likely associated with hospital groups. They purchase Teicoplanin to meet the specific needs of their affiliated hospitals, focusing on ensuring the availability of essential antibiotics for patient care. Their purchasing behavior includes regular orders based on hospital demand and treatment protocols.
Country-Specific Import Regulations
TransData Nexus Teicoplanin buyer market intelligence
1Country-Specific Import Regulations
Hungary:
1. Drug Regulatory Authority: National Institute of Pharmacy and Nutrition (OGYÉI).
2. Import Registration Process for Teicoplanin: Importers must submit a marketing authorization application to OGYÉI, including comprehensive data on the drug's quality, safety, and efficacy. The process involves dossier evaluation, potential inspections, and compliance with EU regulations.
3. Estimated Timeline for Registration: Approximately 210 days, subject to the completeness of the application and any additional information requests.
4. Import Tariff/Duty for HS 30042099: As an EU member, Hungary applies the EU's Common External Tariff, which may allow for duty-free importation of pharmaceutical products under HS code 30042099.
Sri Lanka:
1. Drug Regulatory Authority: National Medicines Regulatory Authority (NMRA).
2. Import Registration Process for Teicoplanin: Importers are required to obtain registration from NMRA by submitting an application with detailed information on the product, including manufacturing details, quality control measures, and clinical data.
3. Estimated Timeline for Registration: Approximately 6 to 12 months, depending on the review process and any additional information required.
4. Import Tariff/Duty for HS 30042099: Pharmaceutical products under HS code 30042099 are typically subject to a duty of 0% to 15%, depending on the specific product and applicable trade agreements.
South Africa:
1. Drug Regulatory Authority: South African Health Products Regulatory Authority (SAHPRA).
2. Import Registration Process for Teicoplanin: Importers must apply for registration with SAHPRA, providing comprehensive data on the product's quality, safety, and efficacy. The process includes dossier evaluation and potential facility inspections.
3. Estimated Timeline for Registration: Approximately 12 to 24 months, subject to the complexity of the application and any additional information requests.
4. Import Tariff/Duty for HS 30042099: Pharmaceutical products under HS code 30042099 are generally subject to a duty of 0%, aligning with South Africa's commitment to facilitating access to essential medicines.
Cuba:
1. Drug Regulatory Authority: Centro para el Control Estatal de Medicamentos, Equipos y Dispositivos Médicos (CECMED).
2. Import Registration Process for Teicoplanin: Importers must submit a registration application to CECMED, including detailed information on the product's quality, safety, and efficacy. The process involves dossier evaluation and compliance with national health regulations.
3. Estimated Timeline for Registration: Approximately 12 months, depending on the completeness of the application and any additional information required.
4. Import Tariff/Duty for HS 30042099: Cuba applies import duties on pharmaceutical products, which can vary; specific rates for HS code 30042099 should be confirmed with Cuban customs authorities.
Germany:
1. Drug Regulatory Authority: Federal Institute for Drugs and Medical Devices (BfArM).
2. Import Registration Process for Teicoplanin: Importers must obtain a marketing authorization from BfArM, submitting a comprehensive dossier that includes data on the drug's quality, safety, and efficacy. The process aligns with EU regulations and may involve mutual recognition procedures.
3. Estimated Timeline for Registration: Approximately 210 days, subject to the completeness of the application and any additional information requests.
4. Import Tariff/Duty for HS 30042099: As an EU member, Germany applies the EU's Common External Tariff, which may allow for duty-free importation of pharmaceutical products under HS code 30042099.
Demand Drivers & Market Opportunity
TransData Nexus Teicoplanin buyer market intelligence
1Demand Drivers & Market Opportunity
The demand for Teicoplanin in importing countries is driven by several factors:
1. Disease Prevalence: Teicoplanin is an antibiotic used to treat serious infections caused by Gram-positive bacteria, including methicillin-resistant *Staphylococcus aureus* (MRSA). The prevalence of such infections necessitates the availability of effective antibiotics like Teicoplanin.
2. Government Healthcare Programs: Many countries have implemented programs to combat antimicrobial resistance and ensure the availability of essential antibiotics. For instance, the WHO's AWaRe classification system, updated in 2019, categorizes antibiotics to guide their appropriate use and procurement. Teicoplanin falls into the 'Watch' category, indicating its importance in treating specific infections and the need for careful monitoring of its use. (who.int)
3. Universal Health Coverage Expansion: Efforts to expand universal health coverage have led to increased procurement of essential medicines, including antibiotics. Countries are investing in their healthcare systems to provide comprehensive care, which includes access to critical medications like Teicoplanin.
4. WHO Essential Medicines List Procurement: The WHO Model List of Essential Medicines serves as a guide for countries to develop their national lists. The 2019 update included revisions to the AWaRe classification, emphasizing the need for access to a range of antibiotics, including those in the 'Watch' category like Teicoplanin. (who.int)
5. Tender-Based Purchasing: Many countries utilize tendering processes to procure medicines for their public healthcare systems. This approach ensures competitive pricing and consistent supply of essential medicines. For example, South Africa employs a centralized tender system for pharmaceutical procurement, which has been effective in standardizing processes and achieving economies of scale.
With 108 buyers across 44 countries importing Teicoplanin from India, there is a significant market opportunity. The repeat buyer rate of 62.0% indicates sustained demand and satisfaction with the product, suggesting potential for continued growth in these markets.
Common Questions — Teicoplanin Buyers & Importers
Answers based on Indian Customs (DGFT) shipment records compiled by TransData Nexus
QWho is the largest teicoplanin buyer importing from India?
Based on import volume and value, AUSTELL PHARMACEUTICALS ( PTY) LTD (SOUTH AFRICA) leads with $357.5K in imports and a 5.7% market share — the highest of any single teicoplanin importer. MEDICUBA, S.A. (CUBA) and HANSE SERVICE INTERNATIONALE (GERMANY) are the next largest buyers.
QHow many companies buy teicoplanin from India?
There are 108 active teicoplanin buyers importing from India, with a combined market of $6.3M across 310 shipments to 44 countries. The top 5 buyers hold 23.0% of total import value, while the remaining 103 buyers handle the other 77.0%.
QWhich countries import the most teicoplanin from India?
The top importing countries for teicoplanin from India are Hungary (35.6%), Sri Lanka (18.4%), South Africa (17.9%), Cuba (5.4%), Germany (5.4%). These markets represent the largest demand centres for Indian pharmaceutical exports of teicoplanin, with buyers ranging from government procurement agencies to private pharmaceutical distributors.
QWhat is the average import order value for teicoplanin from India?
The average import transaction value for teicoplanin from India is $20.3K, with an average unit price of $1030.74 per unit. Order sizes vary significantly based on buyer type — government tenders tend to be larger while private distributors place more frequent smaller orders.
Buyer Segment Analysis
TransData Nexus Teicoplanin buyer market intelligence
1Buyer Segment Analysis
The importation of Teicoplanin from India involves various buyer segments, each with distinct purchasing behaviors and order patterns.
Distributors: Entities such as Austell Pharmaceuticals (Pty) Ltd in South Africa and Hanse Service Internationale in Germany function as distributors. They procure Teicoplanin in bulk to supply hospitals, clinics, and pharmacies within their respective regions. Their purchasing behavior is characterized by large-volume orders, often through tender processes, to ensure a steady supply chain and meet the demands of multiple healthcare providers.
Government Procurement Agencies: Organizations like Medicuba S.A. in Cuba and People's Bank in Sri Lanka represent government procurement agencies. They acquire Teicoplanin to distribute within public healthcare systems, often through national tenders or agreements. Their order patterns are typically large and periodic, aligning with government budget cycles and healthcare program needs.
Hospital Groups: Slim Pharmaceuticals (Pvt) Ltd and Yaden International (Pvt) Ltd in Sri Lanka, as well as Medpro Pharmaceutica Pty Ltd in South Africa, are likely associated with hospital groups. They purchase Teicoplanin to meet the specific needs of their affiliated hospitals, focusing on ensuring the availability of essential antibiotics for patient care. Their purchasing behavior includes regular orders based on hospital demand and treatment protocols.
Country-Specific Import Regulations
TransData Nexus Teicoplanin buyer market intelligence
1Country-Specific Import Regulations
Hungary:
1. Drug Regulatory Authority: National Institute of Pharmacy and Nutrition (OGYÉI).
2. Import Registration Process for Teicoplanin: Importers must submit a marketing authorization application to OGYÉI, including comprehensive data on the drug's quality, safety, and efficacy. The process involves dossier evaluation, potential inspections, and compliance with EU regulations.
3. Estimated Timeline for Registration: Approximately 210 days, subject to the completeness of the application and any additional information requests.
4. Import Tariff/Duty for HS 30042099: As an EU member, Hungary applies the EU's Common External Tariff, which may allow for duty-free importation of pharmaceutical products under HS code 30042099.
Sri Lanka:
1. Drug Regulatory Authority: National Medicines Regulatory Authority (NMRA).
2. Import Registration Process for Teicoplanin: Importers are required to obtain registration from NMRA by submitting an application with detailed information on the product, including manufacturing details, quality control measures, and clinical data.
3. Estimated Timeline for Registration: Approximately 6 to 12 months, depending on the review process and any additional information required.
4. Import Tariff/Duty for HS 30042099: Pharmaceutical products under HS code 30042099 are typically subject to a duty of 0% to 15%, depending on the specific product and applicable trade agreements.
South Africa:
1. Drug Regulatory Authority: South African Health Products Regulatory Authority (SAHPRA).
2. Import Registration Process for Teicoplanin: Importers must apply for registration with SAHPRA, providing comprehensive data on the product's quality, safety, and efficacy. The process includes dossier evaluation and potential facility inspections.
3. Estimated Timeline for Registration: Approximately 12 to 24 months, subject to the complexity of the application and any additional information requests.
4. Import Tariff/Duty for HS 30042099: Pharmaceutical products under HS code 30042099 are generally subject to a duty of 0%, aligning with South Africa's commitment to facilitating access to essential medicines.
Cuba:
1. Drug Regulatory Authority: Centro para el Control Estatal de Medicamentos, Equipos y Dispositivos Médicos (CECMED).
2. Import Registration Process for Teicoplanin: Importers must submit a registration application to CECMED, including detailed information on the product's quality, safety, and efficacy. The process involves dossier evaluation and compliance with national health regulations.
3. Estimated Timeline for Registration: Approximately 12 months, depending on the completeness of the application and any additional information required.
4. Import Tariff/Duty for HS 30042099: Cuba applies import duties on pharmaceutical products, which can vary; specific rates for HS code 30042099 should be confirmed with Cuban customs authorities.
Germany:
1. Drug Regulatory Authority: Federal Institute for Drugs and Medical Devices (BfArM).
2. Import Registration Process for Teicoplanin: Importers must obtain a marketing authorization from BfArM, submitting a comprehensive dossier that includes data on the drug's quality, safety, and efficacy. The process aligns with EU regulations and may involve mutual recognition procedures.
3. Estimated Timeline for Registration: Approximately 210 days, subject to the completeness of the application and any additional information requests.
4. Import Tariff/Duty for HS 30042099: As an EU member, Germany applies the EU's Common External Tariff, which may allow for duty-free importation of pharmaceutical products under HS code 30042099.
Demand Drivers & Market Opportunity
TransData Nexus Teicoplanin buyer market intelligence
1Demand Drivers & Market Opportunity
The demand for Teicoplanin in importing countries is driven by several factors:
1. Disease Prevalence: Teicoplanin is an antibiotic used to treat serious infections caused by Gram-positive bacteria, including methicillin-resistant *Staphylococcus aureus* (MRSA). The prevalence of such infections necessitates the availability of effective antibiotics like Teicoplanin.
2. Government Healthcare Programs: Many countries have implemented programs to combat antimicrobial resistance and ensure the availability of essential antibiotics. For instance, the WHO's AWaRe classification system, updated in 2019, categorizes antibiotics to guide their appropriate use and procurement. Teicoplanin falls into the 'Watch' category, indicating its importance in treating specific infections and the need for careful monitoring of its use. (who.int)
3. Universal Health Coverage Expansion: Efforts to expand universal health coverage have led to increased procurement of essential medicines, including antibiotics. Countries are investing in their healthcare systems to provide comprehensive care, which includes access to critical medications like Teicoplanin.
4. WHO Essential Medicines List Procurement: The WHO Model List of Essential Medicines serves as a guide for countries to develop their national lists. The 2019 update included revisions to the AWaRe classification, emphasizing the need for access to a range of antibiotics, including those in the 'Watch' category like Teicoplanin. (who.int)
5. Tender-Based Purchasing: Many countries utilize tendering processes to procure medicines for their public healthcare systems. This approach ensures competitive pricing and consistent supply of essential medicines. For example, South Africa employs a centralized tender system for pharmaceutical procurement, which has been effective in standardizing processes and achieving economies of scale.
With 108 buyers across 44 countries importing Teicoplanin from India, there is a significant market opportunity. The repeat buyer rate of 62.0% indicates sustained demand and satisfaction with the product, suggesting potential for continued growth in these markets.
Official References & Regulatory Resources
- European Medicines Agency
- WHO Essential Medicines
- Invest India — Pharma Sector
- India Trade Statistics (DGFT)
Verify import regulations and drug registration requirements with the official agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Identification: 108 global importers of Teicoplanin identified from consignee fields in DGFT shipping bill records.
- 2.Import Value Analysis: Total import value aggregated from 310 individual shipping bill records. Values are FOB in USD.
- 3.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 4.Geographic Distribution: Buyer imports distributed across 44 destination countries with market share per buyer calculated from capped values.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
310 Verified Shipments
108 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists