Ireland to India: Syringe Import Trade Route
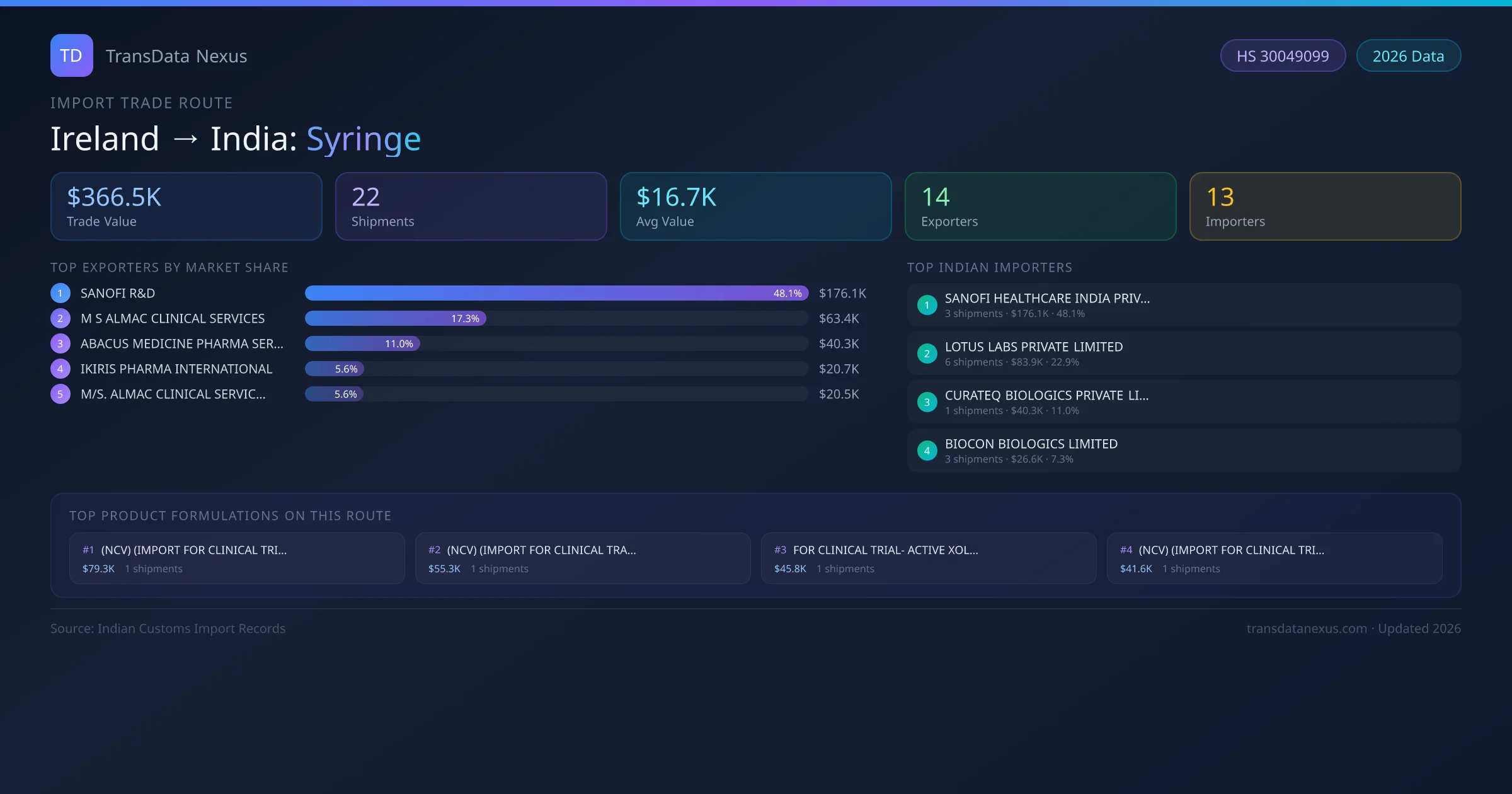
India has recorded 22 verified shipments of Syringe imported from Ireland, representing a combined trade value of $366.5K USD. This corridor is served by 14 active Ireland exporters, with an average shipment value of $16.7K USD. The leading Ireland supplier is SANOFI R&D , which accounts for 48% of total import value with 3 shipments worth $176.1K USD. On the buying side, SANOFI HEALTHCARE INDIA PRIVATE LIMITED is the largest Indian importer with $176.1K USD in purchases. The top 3 suppliers — SANOFI R&D , M S ALMAC CLINICAL SERVICES, ABACUS MEDICINE PHARMA SERVICES LTD — together control 76% of total trade value on this route. All data sourced from Indian Customs (DGFT) shipping bill of entry records. Values reported in FOB USD.
Route Intelligence Overview
The Ireland to India Syringe corridor is one of India's established pharmaceutical import routes from Ireland, with 22 shipments documented worth a combined $366.5K USD. The route is dominated by SANOFI R&D , which alone accounts for roughly 48% of all import value, reflecting the consolidated nature of Ireland's syringe export sector.
Across 14 active Ireland suppliers, the average shipment value stands at $16.7K USD — a figure that reflects both bulk commercial orders from large pharmaceutical companies and smaller specialty shipments. Sea freight dominates at 75% of all shipments, consistent with syringe's non-urgent bulk-order profile.
Shipment activity peaks during January–March, with an average transit time of 17 days port-to-port. The route has recorded an annual growth rate of 12.8%, placing it at rank #20 among India's top syringe import origins globally.
On the Indian buyer side, key Indian importers include SANOFI HEALTHCARE INDIA PRIVATE LIMITED , LOTUS LABS PRIVATE LIMITED, CURATEQ BIOLOGICS PRIVATE LIMITED and 10 others. SANOFI HEALTHCARE INDIA PRIVATE LIMITED is the single largest Indian importer with 3 shipments valued at $176.1K USD.
Route Characteristics
- Average transit17 days
- Peak seasonQ1
- Primary modeSea freight
- Top port of entryN?A
Market Position
- Global rank#20
- Annual growth+12.8%
- Demand growth+13.2%
- Regulatory ease77/100
Top 10 Syringe Exporters from Ireland to India
Showing top 10 of 14 Ireland suppliers exporting Syringe to India, ranked by total trade value (USD)
| Rank | Supplier (Ireland Exporter) | Shipments | Total Value (USD) | Market Share |
|---|---|---|---|---|
| 1 | SANOFI R&D Avg $58.7K per shipment | 3 | $176.1K | 48.1% |
| 2 | M S ALMAC CLINICAL SERVICES Avg $21.1K per shipment | 3 | $63.4K | 17.3% |
| 3 | ABACUS MEDICINE PHARMA SERVICES LTD Avg $40.3K per shipment | 1 | $40.3K | 11.0% |
| 4 | IKIRIS PHARMA INTERNATIONAL Avg $10.4K per shipment | 2 | $20.7K | 5.6% |
| 5 | M/S. ALMAC CLINICAL SERVICES., Avg $6.8K per shipment | 3 | $20.5K | 5.6% |
| 6 | ALMAC CLINICAL SERVICES Avg $10.9K per shipment | 1 | $10.9K | 3.0% |
| 7 | NEWLIFE BIOPHARMA LIMITED Avg $4.6K per shipment | 2 | $9.3K | 2.5% |
| 8 | ABACUS MEDICINE A/S Avg $6.2K per shipment | 1 | $6.2K | 1.7% |
| 9 | NEW LIFE MEDICALS LLC . Avg $5.9K per shipment | 1 | $5.9K | 1.6% |
| 10 | SRIGEN PHARMA LTD Avg $5.2K per shipment | 1 | $5.2K | 1.4% |
This table shows the top 10 of 14 Ireland companies exporting syringe to India, ranked by total trade value. The listed exporters are: SANOFI R&D , M S ALMAC CLINICAL SERVICES, ABACUS MEDICINE PHARMA SERVICES LTD , IKIRIS PHARMA INTERNATIONAL , M/S. ALMAC CLINICAL SERVICES.,, ALMAC CLINICAL SERVICES, NEWLIFE BIOPHARMA LIMITED, ABACUS MEDICINE A/S, NEW LIFE MEDICALS LLC . , SRIGEN PHARMA LTD . SANOFI R&D is the dominant supplier with 3 shipments worth $176.1K USD, giving it a 48% market share. The top 3 suppliers together account for 76% of the total trade value on this route.
Showing top 10 of 14 total Ireland exporters on the Ireland to India Syringe import route.
Top 10 Indian Syringe Importers from Ireland
Showing top 10 of 13 known Indian buyers receiving Syringe shipments from Ireland, ranked by import value
On the receiving end of this trade route, the leading Indian importers of Ireland syringe include SANOFI HEALTHCARE INDIA PRIVATE LIMITED , LOTUS LABS PRIVATE LIMITED, CURATEQ BIOLOGICS PRIVATE LIMITED , BIOCON BIOLOGICS LIMITED , PFIZER LIMITED, among 13 total buyers. The largest importer is SANOFI HEALTHCARE INDIA PRIVATE LIMITED , accounting for $176.1K USD across 3 shipments — representing 48% of all syringe imports from Ireland on this route.
| Rank | Indian Importer / Buyer | Shipments | Import Value (USD) | Market Share |
|---|---|---|---|---|
| 1 | SANOFI HEALTHCARE INDIA PRIVATE LIMITED | 3 | $176.1K | 48.1% |
| 2 | LOTUS LABS PRIVATE LIMITED | 6 | $83.9K | 22.9% |
| 3 | CURATEQ BIOLOGICS PRIVATE LIMITED | 1 | $40.3K | 11.0% |
| 4 | BIOCON BIOLOGICS LIMITED | 3 | $26.6K | 7.3% |
| 5 | PFIZER LIMITED | 1 | $10.9K | 3.0% |
| 6 | EUGIA PHARMA SPECIALITIES LIMITED | 1 | $6.2K | 1.7% |
| 7 | RELIANCE LIFE SCIENCES PRIVATE LIMITED | 1 | $6.2K | 1.7% |
| 8 | ZYDUS LIFESCIENCES LIMITED | 1 | $5.2K | 1.4% |
| 9 | LUPIN LIMITED | 1 | $3.0K | 0.8% |
| 10 | LUPIN LIMITED | 1 | $3.0K | 0.8% |
Showing top 10 of 13 Indian Syringe importers from Ireland on this route.
Top 10 Syringe Formulations Imported from Ireland
Showing top 10 of 21 product formulations shipped on the Ireland to India Syringe route, ranked by trade value
India imports a wide range of syringe formulations from Ireland, spanning tablets, capsules, suspensions, and combination drugs. The top formulation — (NCV) (IMPORT FOR CLINICAL TRIAL USE) SAR440340 (REGN3500) / ITEPEKIMAB 150MG/ML 2 ML IN PRE-FILLED SYRINGE OR PLACEBO — accounts for $79.3K USD across 1 shipments. There are 21 distinct product descriptions in the dataset, reflecting the variety of dosage forms and strengths imported.
| Rank | Product Formulation | Shipments | Trade Value (USD) | Market Share |
|---|---|---|---|---|
| 1 | (NCV) (IMPORT FOR CLINICAL TRIAL USE) SAR440340 (REGN3500) / ITEPEKIMAB 150MG/ML 2 ML IN PRE-FILLED SYRINGE OR PLACEBO | 1 | $79.3K | 21.6% |
| 2 | (NCV) (IMPORT FOR CLINICAL TRAIL USE) SAR440340 (REGN3500) / ITEPEKIMAB 300MG/ML 2ML IN PRE-FILLED SYRINGE OR PLACEBO | 1 | $55.3K | 15.1% |
| 3 | FOR CLINICAL TRIAL- ACTIVE XOLAIR OMALIZUMAB 150MG/ML PREFILLED SYRINGE FOC | 1 | $45.8K | 12.5% |
| 4 | (NCV) (IMPORT FOR CLINICAL TRIAL USE) SAR440340 (REGN3500) / ITEPEKIMAB 150MG/ML 2ML IN PRE-FILLED SYRINGE OR PLACEBO 30 | 1 | $41.6K | 11.4% |
| 5 | INVESTIGATION&TESTING PURPOSE PROLIA,60MG/ML,SOLUTION FOR INJECTION,1 PRE-FILLED SYRINGE WITH (MFG-AMGEN-IRELAND)(136NOS | 1 | $40.3K | 11.0% |
| 6 | (FOC ITEMS) SECUKINUMAB (COSENTYX) 150MG/1ML PREFILLED SYRINGES (2PFS IN 1 CARTON) | 2 | $20.7K | 5.6% |
| 7 | FOR CLINICAL TRIAL - OMALIZUMAB TEV-45779 150 MG/ML1ML FOC PRE-FILLED SYRINGE | 1 | $13.8K | 3.8% |
| 8 | FOR CLINICAL TRIAL- USACTIVE XOLAIR (OMALIZUMAB) 150MG/ML PREFILLED SYRINGE (FOC) | 1 | $13.2K | 3.6% |
| 9 | (FOC)(CLINICAL TRIAL) 1 SINGLE-DOSE PRE-FILLED SYRINGE 20-VALENT PNEUMOCOCCAL CONJUGATE (20VPNC) VACCINE (LOT NO: 22-LU- | 1 | $10.9K | 3.0% |
| 10 | FIRAZYR 30MG SOLUTION FOR INJECTION IN PRE-FILLED SYRINGE (PACK SIZE: 1)INJECTION FOR TEST AND ANALYSIS PURPOSE | 1 | $6.2K | 1.7% |
Showing top 10 of 21 Syringe formulations imported from Ireland on this route.
Shipping & Logistics Analysis
Freight mode split and port-of-origin breakdown
Freight Mode Distribution
Sea freight dominates at 75%, typical for bulk pharmaceutical shipments.
Top Indian Ports of Entry
N?A handles the highest volume with 6 shipments. Transit time averages 17 days by sea.
Market Dynamics
India's syringe imports from Ireland are driven primarily by a handful of large-scale Ireland manufacturers. SANOFI R&D with 3 shipments leads the pack, a pattern common in generic pharmaceutical corridors where manufacturing scale creates significant cost advantages. The presence of 14 active exporters signals a competitive but concentrated market — Indian buyers benefit from Ireland supplier diversity while the top tier handles the majority of volume.
The top 3 suppliers — SANOFI R&D , M S ALMAC CLINICAL SERVICES, ABACUS MEDICINE PHARMA SERVICES LTD — together account for 76% of total trade value on this route. The average shipment value of $16.7K USD reflects a mix of bulk commercial orders and smaller specialty shipments.
Beyond the primary product category, shipments on this route include closely related formulations such as (ncv) (import for clinical trail use) sar440340 (regn3500) / itepekimab 300mg/ml 2ml in pre-filled syringe or placebo and for clinical trial- active xolair omalizumab 150mg/ml prefilled syringe foc, suggesting that Indian buyers tend to consolidate orders across related product lines from the same Ireland supplier.
On the buying side, SANOFI HEALTHCARE INDIA PRIVATE LIMITED is the largest importer with 3 shipments worth $176.1K USD — representing 48% of all syringe imports from Ireland on this route. A total of 13 buyers are active on this corridor.
Route Statistics
- Trade Volume
- $366.5K
- Avg. Shipment
- $16.7K
- Suppliers
- 14
- Buyers
- 13
- Transit (Sea)
- ~17 days
- Annual Growth
- +12.8%
Unlock the Full Ireland to India Syringe Dataset
TransData Nexus provides verified shipment-level records, exporter contact details, HS code breakdowns, real-time pricing benchmarks, and regulatory compliance guides for 22 shipments on this route.
Frequently Asked Questions — Ireland to India Syringe Trade
Answers based on Indian Customs (DGFT) shipment records compiled by TransData Nexus
Q Which Ireland companies export Syringe to India?
The leading Ireland exporters of Syringe to India are SANOFI R&D , M S ALMAC CLINICAL SERVICES, ABACUS MEDICINE PHARMA SERVICES LTD . SANOFI R&D holds the largest market share at approximately 48% of total trade value on this route.
Q What is the total value of Syringe imports from Ireland to India?
India imports Syringe from Ireland worth approximately $366.5K USD across 22 recorded shipments. The average value per shipment is $16.7K USD.
Q Which Indian ports receive Syringe from Ireland?
The most active Indian port of entry is N?A with 6 shipments. Ireland exporters primarily use sea freight for this route, with 75% of shipments going by sea and 22% by air.
Q How long does shipping take from Ireland to India for Syringe?
The average transit time for Syringe shipments from Ireland to India is approximately 17 days. Sea freight typically takes longer, while air freight can reduce this significantly for urgent orders. Peak shipping activity is observed during January–March.
Q Is the Ireland to India Syringe import route growing?
Yes — this trade corridor has seen an annual growth rate of approximately 12.8% with demand growth tracking at 13.2%. The route is ranked #20 among India's top Syringe import origins globally.
Q How many Ireland suppliers are active on this Syringe import route?
There are currently 14 active Ireland suppliers exporting Syringe to India. The market is moderately concentrated with SANOFI R&D accounting for 48% of total shipment value.
Q Who are the main Indian importers of Syringe from Ireland?
The leading Indian importers of Syringe from Ireland include SANOFI HEALTHCARE INDIA PRIVATE LIMITED , LOTUS LABS PRIVATE LIMITED, CURATEQ BIOLOGICS PRIVATE LIMITED , BIOCON BIOLOGICS LIMITED , PFIZER LIMITED. SANOFI HEALTHCARE INDIA PRIVATE LIMITED is the largest buyer with 3 shipments worth $176.1K USD.
Official References & Regulatory Resources
- India Trade Statistics (DGFT)
- Invest India — Pharma Sector
- WCO HS Nomenclature
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory and trade status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Route Identification: India to Ireland export trade corridor identified from Indian Customs (DGFT) records for Syringe.
- 2.Supplier/Buyer Matching: 14 Indian exporters and 13 importers in Ireland matched using name normalization.
- 3.Value Aggregation: Total export value aggregated from 22 verified shipping bill records. Values are FOB in USD.
- 4.Market Share Analysis: Each supplier and buyer contribution calculated as a percentage of total route value. Statistical normalization applied to ensure accurate representation across varying shipment sizes.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
22 Verified Shipments
14 Indian exporters tracked
Expert-Reviewed
By pharmaceutical trade specialists