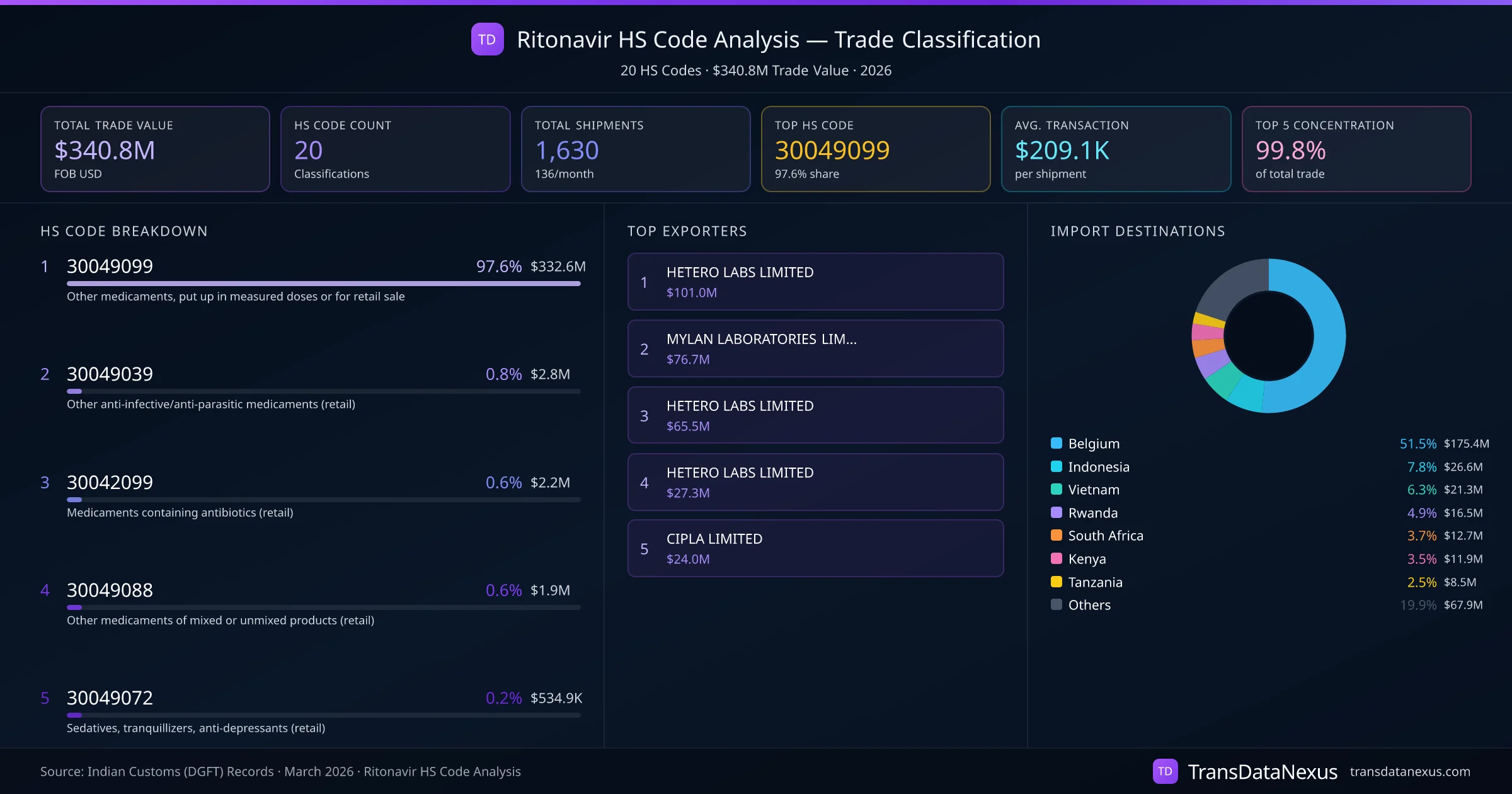
Ritonavir's classification under the Harmonized System can lead to disputes, particularly when determining whether it falls under HS code 3003 or 3004. The distinction hinges on the product's form and intended use:
- HS Code 3003: This code covers "Medicaments (excluding goods of heading 30.02, 30.05, or 30.06) consisting of two or more constituents which have been mixed together for therapeutic or prophylactic uses, not put up in measured doses or in forms or packings for retail sale."
- HS Code 3004: This code pertains to "Medicaments (excluding goods of heading 30.02, 30.05, or 30.06) consisting of mixed or unmixed products for therapeutic or prophylactic uses, put up in measured doses (including those in the form of transdermal administration systems) or in forms or packings for retail sale."
Given that Ritonavir is predominantly traded in finished dosage forms intended for retail sale, it is appropriately classified under HS code 3004. Misclassification can occur if the product is incorrectly declared under HS code 3003, which is intended for bulk forms not packaged for retail. Such errors can lead to customs penalties, delays in clearance, and potential legal disputes.
Our data indicates that Ritonavir is primarily classified under HS code 30049099, accounting for 97.6% of trade, with 1,424 transactions. This consistency suggests a general consensus on its appropriate classification. However, the use of 20 different HS codes across 1,630 shipments highlights the potential for misclassification. Importers and exporters should ensure accurate classification to avoid compliance issues.