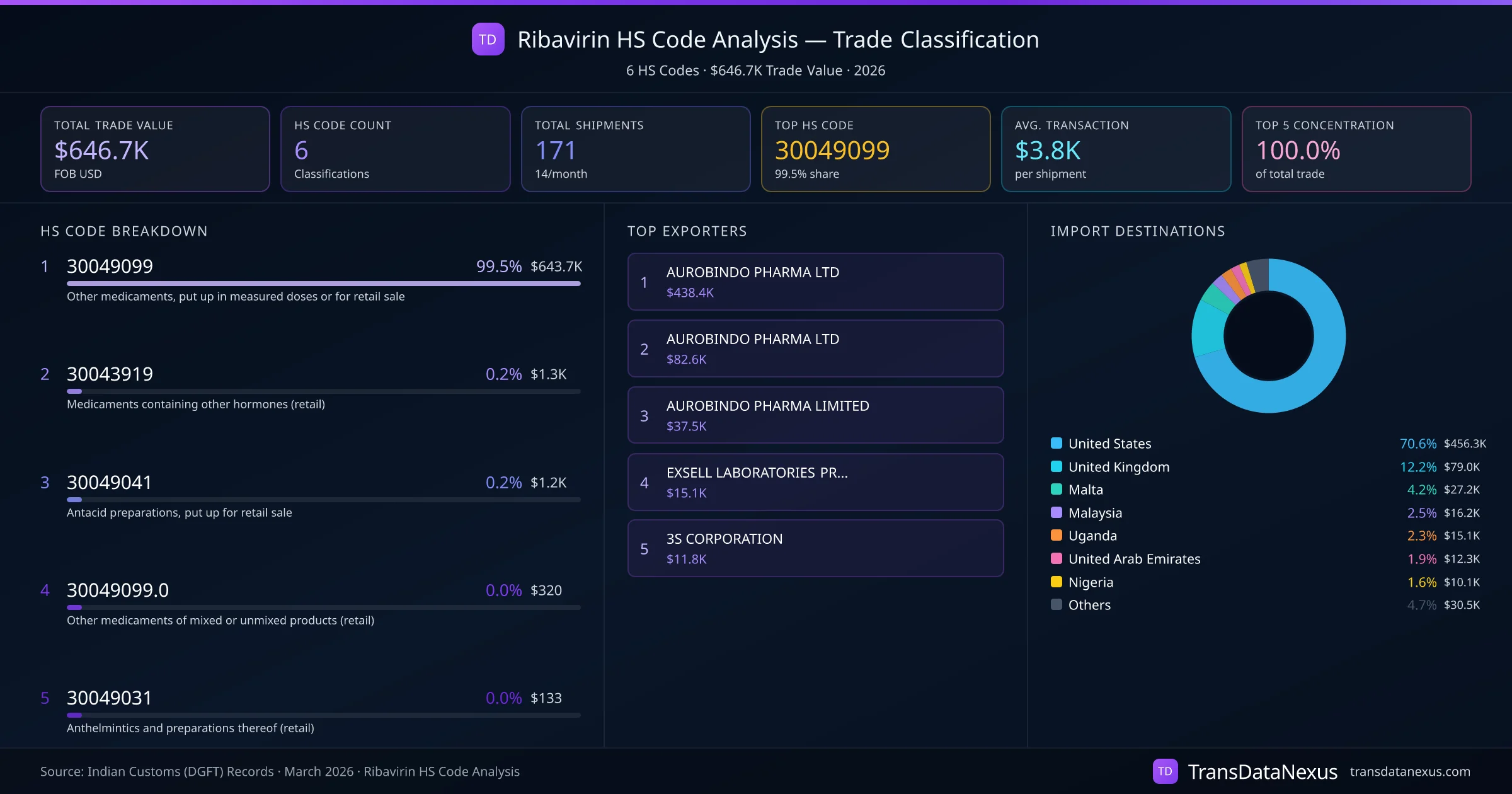
Ribavirin is predominantly classified under HS code 30049099, as evidenced by TransData Nexus data indicating that 99.5% of trade transactions for Ribavirin utilize this code. However, there are instances where other HS codes, such as 30043919 and 30049041, have been applied.
The classification of pharmaceutical products like Ribavirin hinges on their form and intended use:
- HS Code 3003: This code pertains to medicaments consisting of two or more constituents mixed together for therapeutic or prophylactic uses, not put up in measured doses or forms for retail sale.
- HS Code 3004: This code covers medicaments consisting of mixed or unmixed products for therapeutic or prophylactic uses, put up in measured doses or forms for retail sale.
Ribavirin, when formulated and packaged for retail sale in specific dosages, should be classified under HS code 3004. Misclassification can occur if the product's form or packaging is not accurately represented, leading to incorrect tariff applications.
Common errors include misclassifying retail-packaged Ribavirin under HS code 3003, which is intended for bulk medicaments not in measured doses. Such misclassification can result in customs penalties, including fines and delays in clearance. To mitigate these risks, it is essential to ensure accurate product descriptions and classifications in all shipping and customs documentation.
While specific recent rulings from the World Customs Organization (WCO) or national customs authorities regarding Ribavirin classification are not available in the provided sources, it is advisable for importers and exporters to stay informed about any updates or changes in classification guidelines to ensure compliance.