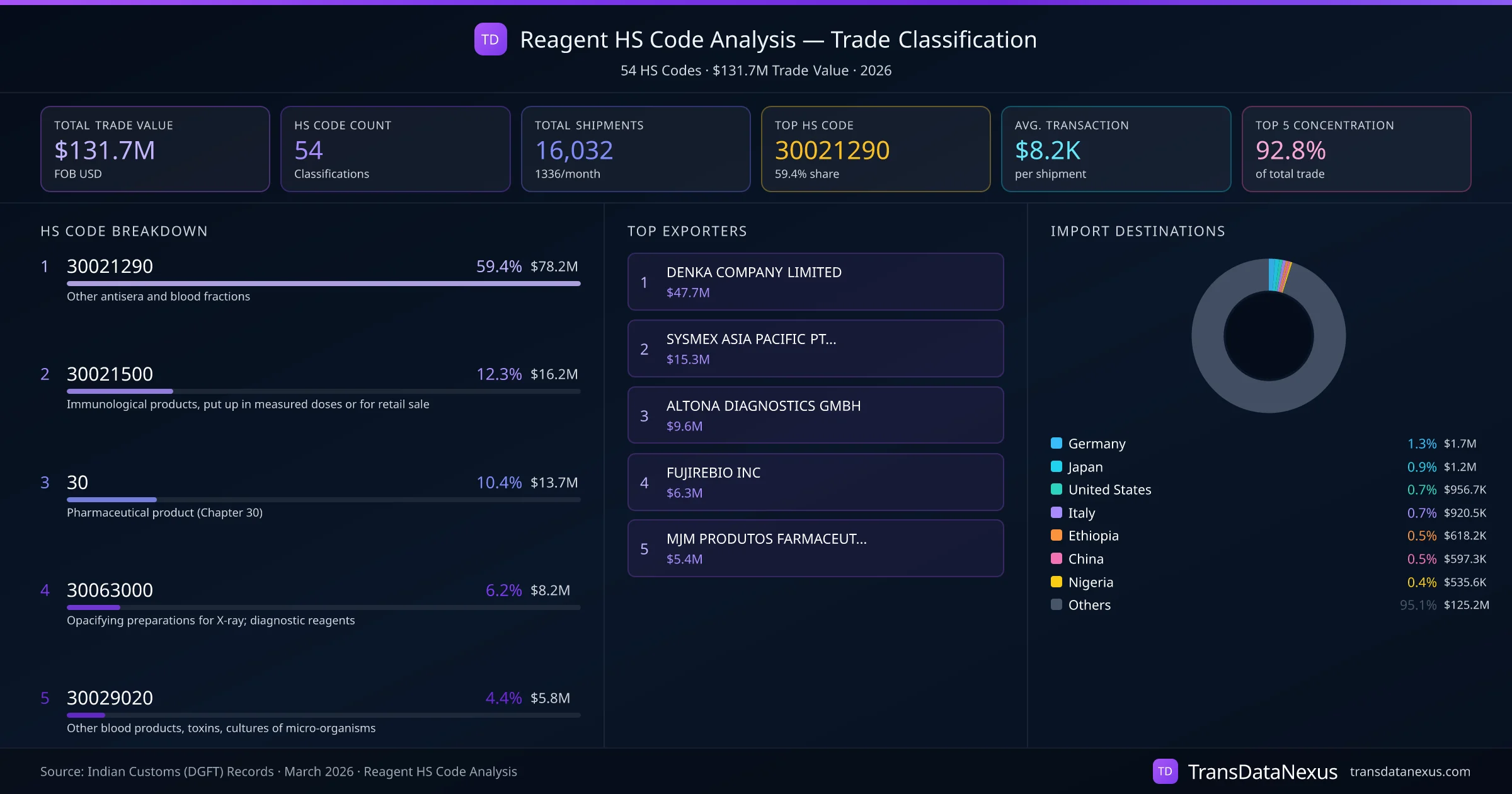
The classification of reagents under HS codes 3003 (Medicaments consisting of two or more constituents mixed together for therapeutic or prophylactic uses) versus 3004 (Medicaments consisting of mixed or unmixed products for therapeutic or prophylactic uses, put up in measured doses) can be contentious. Reagents intended for diagnostic purposes are typically classified under HS 3002, specifically 30021290 for other blood fractions and immunological products.
Common misclassification errors include:
- Misclassifying diagnostic reagents as pharmaceutical products: This can lead to incorrect tariff applications and regulatory non-compliance.
- Incorrectly assigning HS codes based on product form rather than function: For example, classifying a reagent in tablet form under HS 3004 instead of HS 3002.
Customs penalties for misclassification can be significant, including fines, delays in shipment clearance, and potential legal action. Recent rulings by the World Customs Organization (WCO) emphasize the importance of accurate classification to avoid such penalties. For instance, the WCO's amendments in HS 2028 aim to reduce litigation risks by providing clearer classifications for products like dietary supplements, which can be analogous to reagents in terms of classification challenges.
Our internal data indicates that out of 16,032 shipments of reagents, 59.4% were classified under HS 30021290, suggesting a predominant classification practice. However, the use of 54 different HS codes highlights the complexity and potential for misclassification in this category.