Sanofi India Limited — Ramipril Exporter Profile
Indian Pharmaceutical Exporter · #4 for Ramipril · $4.5M export value · DGFT Verified
Sanofi India Limited is the #4 Indian exporter of Ramipril with $4.5M in export value and 90 verified shipments. Sanofi India Limited holds a 5.0% market share in Ramipril exports across 12 countries. The company exports 4 pharmaceutical products worth $9.0M across 4 therapeutic categories.
Sanofi India Limited — Ramipril Export Profile: Buyers & Destinations
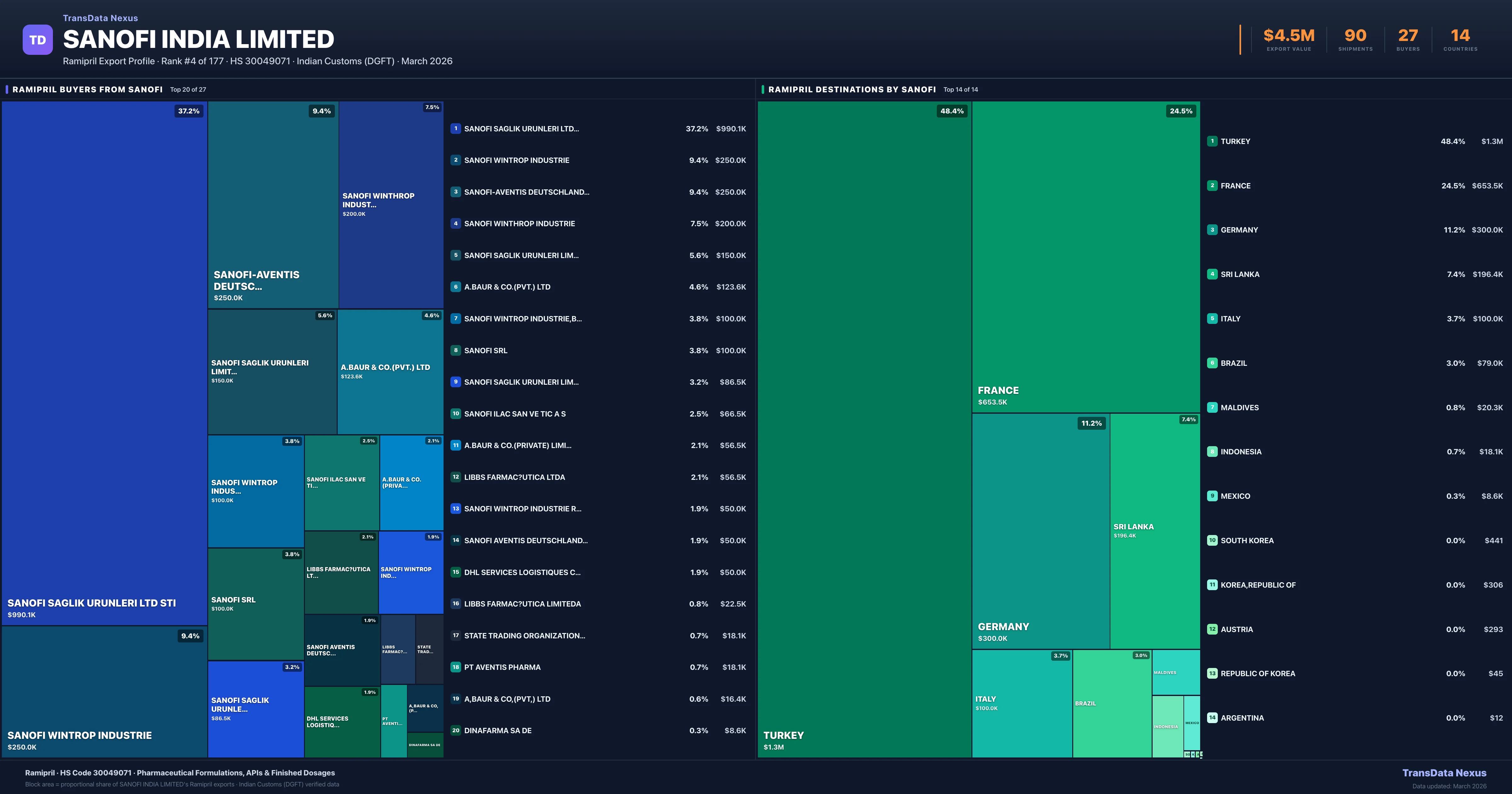
Where Does Sanofi India Limited Export Ramipril?
| Country | Value | Shipments | Share |
|---|---|---|---|
| TURKEY | $1.3M | 29 | 48.4% |
| FRANCE | $653.5K | 16 | 24.5% |
| GERMANY | $300.0K | 6 | 11.2% |
| SRI LANKA | $196.4K | 13 | 7.4% |
| ITALY | $100.0K | 2 | 3.7% |
| BRAZIL | $79.0K | 6 | 3.0% |
| MALDIVES | $20.3K | 10 | 0.8% |
| INDONESIA | $18.1K | 2 | 0.7% |
| MEXICO | $8.6K | 1 | 0.3% |
| SOUTH KOREA | $441 | 1 | 0.0% |
Sanofi India Limited exports Ramipril to 14 countries. The largest destination is TURKEY accounting for 48.4% of Sanofi India Limited's Ramipril shipments, followed by FRANCE (24.5%) and GERMANY (11.2%). These destinations reflect Sanofi India Limited's established distribution network and regulatory approvals in key markets.
Who Are the Top Buyers of Ramipril from Sanofi India Limited?
| Buyer | Country | Value | Orders |
|---|---|---|---|
| SANOFI SAGLIK URUNLERI LTD STI | TURKEY | $990.1K | 22 |
| SANOFI WINTROP INDUSTRIE | FRANCE | $250.0K | 5 |
| SANOFI-AVENTIS DEUTSCHLAND GMBH | GERMANY | $250.0K | 5 |
| SANOFI WINTHROP INDUSTRIE | FRANCE | $200.0K | 4 |
| SANOFI SAGLIK URUNLERI LIMITED STI | TURKEY | $150.0K | 3 |
| A.BAUR & CO.(PVT.) LTD | SRI LANKA | $123.6K | 8 |
| SANOFI WINTROP INDUSTRIE,BOLLORE LO | FRANCE | $100.0K | 2 |
| SANOFI SRL | ITALY | $100.0K | 2 |
| SANOFI SAGLIK URUNLERI LIMITED | TURKEY | $86.5K | 2 |
| SANOFI ILAC SAN VE TIC A S | TURKEY | $66.5K | 2 |
Sanofi India Limited supplies Ramipril to 27 buyers globally. The largest buyer is SANOFI SAGLIK URUNLERI LTD STI (TURKEY), followed by SANOFI WINTROP INDUSTRIE (FRANCE) and SANOFI-AVENTIS DEUTSCHLAND GMBH (GERMANY). These buyers represent pharmaceutical distributors, hospital groups, and government procurement agencies in their respective markets.
What Is India's Total Ramipril Export Value and How Much Does Sanofi India Limited Contribute?
India exported $82.5M worth of Ramipril through 4,977 shipments from 177 suppliers to 93 countries, serving 389 buyers globally. Sanofi India Limited contributes $4.5M to this total, accounting for 5.0% of India's Ramipril exports. Sanofi India Limited ships Ramipril to 14 countries through 27 buyers.
What Is the Average Shipment Value for Sanofi India Limited's Ramipril Exports?
Sanofi India Limited's average Ramipril shipment value is $50.0K per consignment, based on 90 shipments totaling $4.5M. The largest destination is TURKEY (48.4% of Sanofi India Limited's Ramipril exports).
How Does Sanofi India Limited Compare to Other Indian Ramipril Exporters?
Sanofi India Limited ranks #4 among 177 Indian Ramipril exporters with a 5.0% market share. The top 3 exporters are MYLAN LABORATORIES LIMITED ($16.0M), GENO PHARMACEUTICALS PRIVATE LIMITED ($14.0M), AUROBINDO PHARMA LTD ($13.3M). Sanofi India Limited processed 90 shipments to 12 destination countries.
What Ramipril Formulations Does Sanofi India Limited Export?
| Product Description / Formulation | Value | Shipments |
|---|---|---|
| PHARMACEUTICAL PRODUCT, RAMIPRIL PC GRAN | $100.8K | 8 |
| RAMIPRIL PC GRANULES 850G/1KG AG GMID: 8 | $100.0K | 2 |
| RAMIPRIL PC GRANULES 850G/1KG (273.60 KG | $100.0K | 2 |
| PHARMACEUTICAL PRODUCTS RAMIPRIL PC Gra | $100.0K | 2 |
| PHARMACEUTICAL PRODUCTS: RAMIPRIL PC GRANULES 850G/1KG (173.35 KG) Exempted Material (W/O Wastage) RAMIPRIL 147.348 KG | $100.0K | 2 |
| PHARMACEUTICAL PRODUCTS: RAMIPRIL PC GRANULES 850G/1KG (30.000 KG) Exempted Material (W/O Wastage) RAMIPRIL 25.500 KG | $56.5K | 2 |
| RAMIPRIL 850G/1KG GRANULES (43.170 KG) E | $50.0K | 1 |
| RAMIPRIL 850G/1KG GRANULES (130.160 KG) | $50.0K | 1 |
| RAMIPRIL PC 850G/1KG GRANULES (124.910 K | $50.0K | 1 |
| RAMIPRIL PC Granules 850G/1KG (129.960 K | $50.0K | 1 |
Sanofi India Limited exports 74 distinct Ramipril formulations including tablets, capsules, syrups, and combination drugs. The top formulation is PHARMACEUTICAL PRODUCT, RAMIPRIL PC GRAN with 8 shipments worth $100.8K.
Regulatory Requirements: Exporting Ramipril to Key Markets
What Sanofi India Limited must comply with to export Ramipril to its top destination countries
Germany — BfArM/EMA
Approval Process
Marketing Authorisation via Decentralized or Mutual Recognition Procedure under EU Directive 2001/83/EC. CTD Module 1–5 dossier required.
Timeline: 12–18 months via Decentralized Procedure
GMP & Export Requirements
EU GMP (EudraLex Vol. 4); GMP certificate from EU competent authority; WHO Prequalification accepted as supporting evidence
Certificate of Pharmaceutical Product (CPP) from CDSCO; Written Confirmation for API export per EU requirements; EU FMD serialization mandatory
Note: Indian manufacturers must hold EU GMP certificate. EU Falsified Medicines Directive (FMD) serialization and tamper-evident packaging mandatory.
Regulatory information is provided for general reference based on publicly available guidelines. Requirements vary by formulation, dosage form, and therapeutic classification. Exporters should consult qualified regulatory affairs professionals for case-specific guidance.
How Does Sanofi India Limited Compare to Nearest Ramipril Exporters?
Exporters ranked immediately above and below #4 by export value
| # | Supplier | Export Value | Shipments | Countries | Avg/Shipment |
|---|---|---|---|---|---|
| 6 | ZENTIVA PRIVATE LIMITED | $4.9M | 233 | 5 | $21.0K |
| 7 | SUN PHARMACEUTICAL INDUSTRIES LIMITED | $4.8M | 361 | 5 | $13.2K |
| 4 | SANOFI INDIA LIMITED ★ | $4.5M | 90 | 12 | $50.0K |
| 8 | MICRO LABS LIMITED | $4.2M | 141 | 2 | $30.1K |
| 9 | AUROBINDO PHARMA LIMITED | $4.0M | 276 | 8 | $14.6K |
Sanofi India Limited ranks #4 among 177 Indian Ramipril exporters. Average shipment value of $50.0K compared to the market average of $466.2K. The closest competitors by value are ZENTIVA PRIVATE LIMITED and SUN PHARMACEUTICAL INDUSTRIES LIMITED.
Which Indian Ports Ship Ramipril Exports?
| Port | Shipments | % Share |
|---|---|---|
| NHAVA SHEVA SEA (INNSA1) | 658 | 13.2% |
| JNPT/ NHAVA SHEVA SEA | 553 | 11.1% |
| SAHAR AIR CARGO ACC (INBOM4) | 305 | 6.1% |
| SAHAR AIR | 291 | 5.8% |
| JNPT | 271 | 5.4% |
| DELHI AIR CARGO ACC (INDEL4) | 214 | 4.3% |
| DELHI AIR | 201 | 4.0% |
| VIZAG SEA | 194 | 3.9% |
Geopolitical & Trade Policy Impact on Sanofi India Limited's Ramipril Exports
Recent geopolitical developments have introduced significant challenges for Indian pharmaceutical exporters like Sanofi India. The escalation of conflicts in the Middle East, particularly around the Red Sea and Suez Canal, has disrupted key shipping routes. Major carriers have suspended or restricted transits, leading to extended transit times by 10–20 days and increased freight rates by 40–50% on India–Europe routes. (livemint.com) These disruptions pose risks to the timely delivery of temperature-sensitive pharmaceuticals, potentially affecting product efficacy and market competitiveness.
In the United States, the implementation of tariffs on pharmaceutical imports, including a 25% reciprocal tariff and an additional 25% penalty tied to India's energy policies, has created an uncertain trade environment. Although pharmaceuticals have been exempt from some of these tariffs, the potential for future escalations remains a concern. (en.wikipedia.org) Additionally, the European Union's stringent regulatory frameworks, such as the Falsified Medicines Directive, necessitate rigorous compliance measures, adding complexity to market entry and sustainability for exporters like Sanofi India.
Sanofi India Limited — Regulatory Compliance & Quality Standards
Compliance with international regulatory standards is paramount for Indian pharmaceutical exporters. Sanofi India has consistently adhered to guidelines set by the U.S. Food and Drug Administration (FDA), the World Health Organization (WHO), and the European Union's Good Manufacturing Practices (EU GMP). This commitment ensures product quality and facilitates access to global markets. However, evolving regulatory requirements, such as the EU's Falsified Medicines Directive, demand continuous adaptation and investment in compliance infrastructure. For instance, the directive's serialization and traceability mandates require significant technological upgrades, impacting operational costs and necessitating strategic planning.
About Sanofi India Limited
Sanofi India Limited exports 4 products worth $9.0M. Beyond Ramipril, top products include Codeine, Insulin, Teicoplanin. View the complete Sanofi India Limited profile for full portfolio analysis.
Market Intelligence
Latest developments and market context
Detailed market intelligence for Ramipril — including regulatory updates, trade policy changes, and competitive landscape analysis — will be available in an upcoming update. This section will feature AI-researched insights from verified industry sources, government notifications, and market reports.
Official References & Regulatory Resources
- CDSCO India
- Pharmexcil
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- India Trade Statistics (DGFT)
Verify manufacturer licensing and export certifications with the official agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Product Matching: Ramipril shipments identified using fuzzy matching on DGFT product description fields, validated against Indian Pharmacopoeia nomenclature.
- 2.Supplier Identification: Sanofi India Limited matched across shipments using name normalization, accounting for company name formatting variations.
- 3.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation. This removes the impact of unusually large one-off transactions that could distort company rankings.
- 4.Verified Records: This page is backed by 90 individual customs records matching Sanofi India Limited exporting Ramipril, covering 74 formulations to 14 countries via 27 buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
Global Coverage
Exports to 93+ countries, 389+ buyers
Expert-Reviewed
Analyzed by pharmaceutical trade specialists
Get Ramipril Export Data from Sanofi India Limited
Access shipment-level details, pricing data, buyer connections, and competitive analysis for Sanofi India Limited's Ramipril exports. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Sanofi India Limited
Full Company Profile →
4 products · $9.0M total trade · 4 categories
Ramipril Stats
Company Overview
Top Products by Sanofi India Limited
Related Analysis
Need Detailed Export Data?
Access shipment-level records, pricing trends, and buyer contacts for Sanofi India Limited.
Request DemoView Pricing →Data Source
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Market share calculated relative to total Indian pharmaceutical exports for Ramipril. For current shipment-level data, contact TransData Nexus.