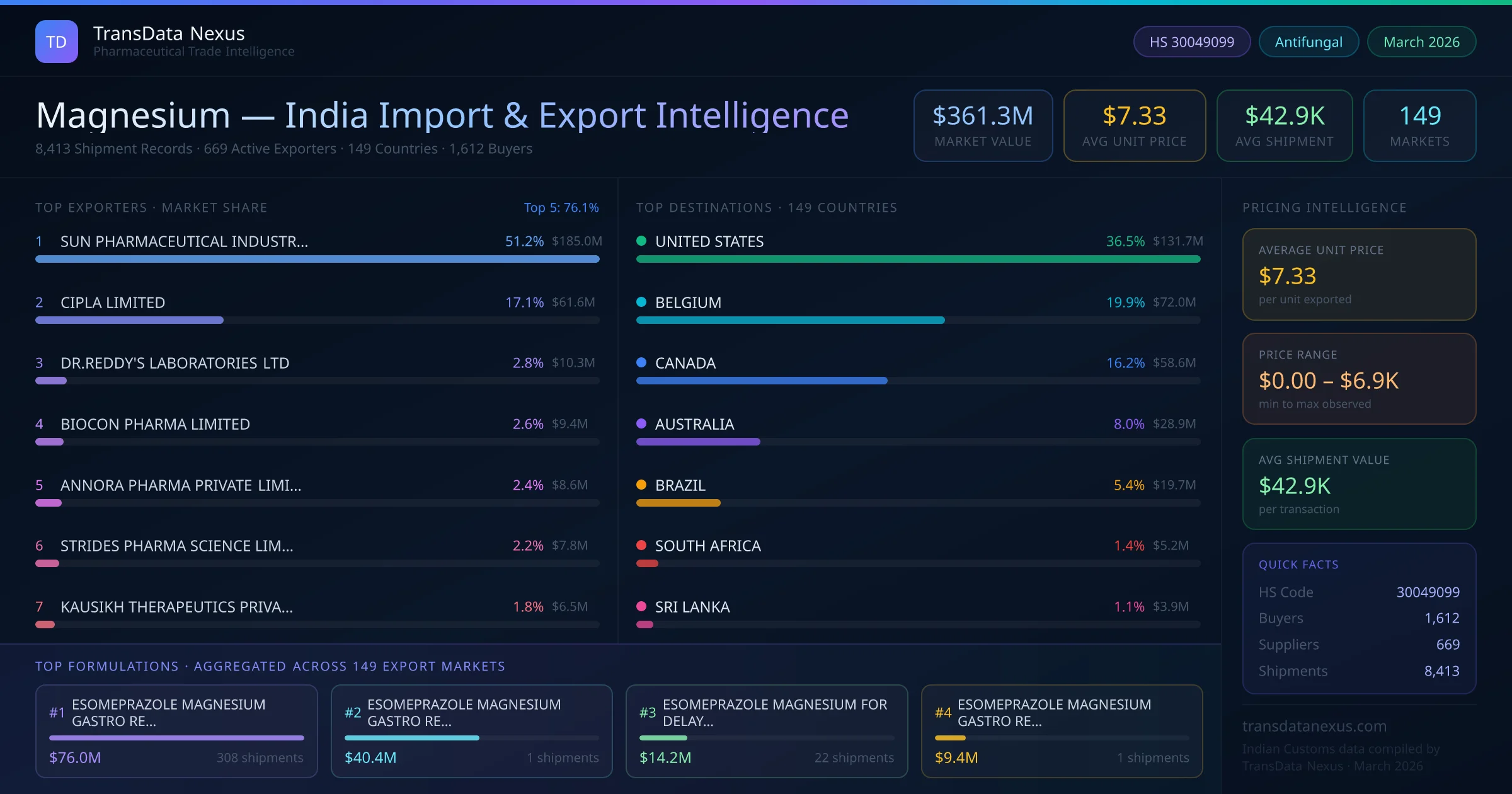
Importing finished pharmaceutical formulations containing magnesium into India requires compliance with the Drugs and Cosmetics Act, 1940, and associated rules. The Central Drugs Standard Control Organization (CDSCO) mandates that all imported drugs, including magnesium formulations, be registered and approved before distribution. The registration process involves obtaining an Import Registration Certificate and an Import License from CDSCO. The application must include detailed product information, manufacturing details, and evidence of compliance with Indian standards. The timeline for registration can vary but typically ranges from 6 to 12 months, depending on the complexity of the product and the completeness of the application. For formulations under HS Code 30049099, specific requirements include stability data demonstrating a shelf life of at least 24 months, batch manufacturing records, and a Certificate of Pharmaceutical Product (CoPP) from the manufacturer. Additionally, a No Objection Certificate (NOC) from the manufacturer is required to confirm that the product is authorized for export to India.