STADAPHARM GMBH — Leflunomide Importer Profile
Pharmaceutical Importer · #8 buyer for Leflunomide · $478.9K import value · 31 shipments · GERMANY · DGFT Verified
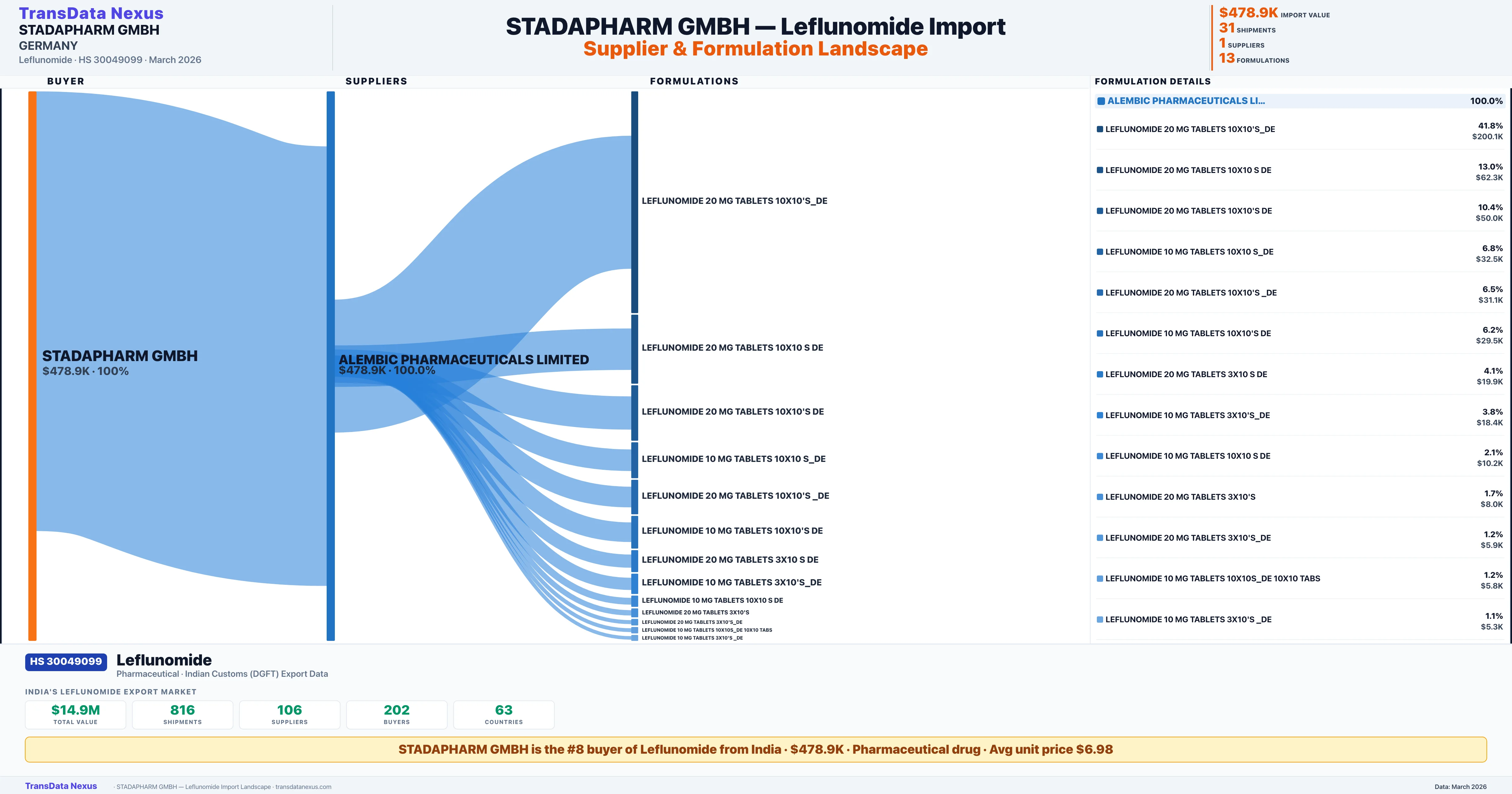
STADAPHARM GMBH is the #8 importer of Leflunomide from India with $478.9K in import value and 31 verified shipments. STADAPHARM GMBH holds a 2.2% market share in Leflunomide imports based in GERMANY. The company sources from 1 verified supplier across 13 formulations shipped to 1 country.
STADAPHARM GMBH — Leflunomide Import Profile: Suppliers, Formulations & Sources
Who Actually Supplies Leflunomide to STADAPHARM GMBH?
Verified from 31 customs shipment records
| Supplier | Value | Shipments | % Share |
|---|---|---|---|
| ALEMBIC PHARMACEUTICALS LIMITED | $478.9K | 31 | 100.0% |
STADAPHARM GMBH sources Leflunomide from 1 verified Indian supplier based on 31 customs records. The primary supplier is ALEMBIC PHARMACEUTICALS LIMITED accounting for 100.0% of imports.
How Does STADAPHARM GMBH Compare to Nearest Leflunomide Importers?
Buyers ranked immediately above and below #8 by import value
| # | Buyer | Country | Import Value | Shipments | Avg/Shipment |
|---|---|---|---|---|---|
| 4 | NATIONS TRUST BANK PLC. | SRI LANKA | $457.9K | 10 | $45.8K |
| 5 | EMCURE PHARMA CHILE S.P.A. | CHILE | $373.7K | 21 | $17.8K |
| 8 | STADAPHARM GMBH ★ | GERMANY | $320.8K | 17 | $18.9K |
| 10 | LLP "DARI-PHARM (KAZAKHSTAN) | KAZAKHSTAN | $269.0K | 10 | $26.9K |
| 11 | ALLOGA (NEDERLAND) B.V | NETHERLANDS | $260.0K | 18 | $14.4K |
STADAPHARM GMBH ranks #8 among 202 Leflunomide importers from India. Average shipment value of $18.9K compared to the market average of $41.3K. The closest competitors by value are NATIONS TRUST BANK PLC. and EMCURE PHARMA CHILE S.P.A..
Where Does STADAPHARM GMBH Import Leflunomide To?
Destination countries from 31 verified shipments
| Country | Value | Shipments | Share |
|---|---|---|---|
| GERMANY | $478.9K | 31 | 100.0% |
STADAPHARM GMBH imports Leflunomide to 1 country. The primary destination is GERMANY receiving 100.0% of shipments worth $478.9K.
What Leflunomide Formulations Does STADAPHARM GMBH Import?
| Formulation | Value | Shipments |
|---|---|---|
| LEFLUNOMIDE 20 MG TABLETS 10X10'S_DE | $200.1K | 9 |
| LEFLUNOMIDE 20 MG TABLETS 10X10 S DE | $62.3K | 3 |
| LEFLUNOMIDE 20 MG TABLETS 10X10'S DE | $50.0K | 1 |
| LEFLUNOMIDE 10 MG TABLETS 10X10 S_DE | $32.5K | 2 |
| LEFLUNOMIDE 20 MG TABLETS 10X10'S _DE | $31.1K | 2 |
| LEFLUNOMIDE 10 MG TABLETS 10X10'S DE | $29.5K | 1 |
| LEFLUNOMIDE 20 MG TABLETS 3X10 S DE | $19.9K | 4 |
| LEFLUNOMIDE 10 MG TABLETS 3X10'S_DE | $18.4K | 3 |
| LEFLUNOMIDE 10 MG TABLETS 10X10 S DE | $10.2K | 1 |
| LEFLUNOMIDE 20 MG TABLETS 3X10'S | $8.0K | 1 |
STADAPHARM GMBH imports 13 distinct Leflunomide formulations from India. The top formulation is LEFLUNOMIDE 20 MG TABLETS 10X10'S_DE with 9 shipments worth $200.1K.
Regulatory Requirements: Importing Leflunomide into Germany
What STADAPHARM GMBH and other importers must comply with to import Leflunomide from India
Regulatory Authority
Federal Institute for Drugs and Medical Devices (BfArM) / European Medicines Agency (EMA) (BfArM/EMA)
Approval Process for Generic Leflunomide
Marketing Authorisation Application (MAA) under EU Directive 2001/83/EC. Decentralized or Mutual Recognition Procedure for generics. Requires CTD Module 1–5 dossier.
Typical timeline: 12–18 months via Decentralized Procedure
GMP Requirements
EU GMP (EudraLex Vol. 4) compliance; GMP certificate issued by competent authority; WHO Prequalification accepted as supporting evidence
Import Licence & Documentation
Wholesale Distribution Authorisation (WDA) and Manufacturing/Import Authorisation (MIA) required for importer. Qualified Person (QP) must certify each batch.
Labeling & Packaging
EU labeling per Directive 2001/83/EC; German language patient leaflet (PIL) mandatory; serialization under EU Falsified Medicines Directive (FMD)
Note: EU FMD serialization and tamper-evident packaging mandatory since February 2019. Indian API/FDF manufacturers must hold EU GMP certificate.
Regulatory information is provided for general reference based on publicly available BfArM/EMA guidelines for pharmaceutical imports. Requirements may vary by specific drug formulation, dosage form, and therapeutic classification. Importers should consult directly with BfArM/EMA or qualified regulatory affairs professionals for case-specific guidance.
Who Are the Top Buyers of Leflunomide from India and How Much Does STADAPHARM GMBH Import?
India exported $8.3M worth of Leflunomide through 816 shipments from 106 suppliers to 63 countries, serving 202 buyers globally. STADAPHARM GMBH imports $478.9K of this total, accounting for 2.2% of India's Leflunomide exports with 31 shipments from GERMANY. This is verified from 31 customs shipment records covering 13 distinct formulations.
What Is the Average Order Value for STADAPHARM GMBH's Leflunomide Imports?
STADAPHARM GMBH's average Leflunomide order value is $15.4K per consignment, based on 31 shipments totaling $478.9K. STADAPHARM GMBH is based in GERMANY and ranks #8 among all Leflunomide importers from India by value. The company sources from 1 verified supplier in India.
Who Supplies Leflunomide to STADAPHARM GMBH?
STADAPHARM GMBH sources Leflunomide from 1 verified Indian supplier: ALEMBIC PHARMACEUTICALS LIMITED ($478.9K, 100.0%). This is based on 31 customs records.
How Does STADAPHARM GMBH Compare to Other Leflunomide Importers?
STADAPHARM GMBH ranks #8 among 202 Leflunomide importers from India with a 2.2% market share. The top 3 importers are EVERSANA LIFE SCIENCE SERVICES ($4.3M), ROPACK ($511.9K), NATIONS TRUST BANK PLC. ($457.9K). STADAPHARM GMBH processed 31 shipments from GERMANY.
Market Intelligence
Latest developments and market context
Detailed market intelligence for Leflunomide — including regulatory updates, trade policy changes, and competitive landscape analysis — will be available in an upcoming update. This section will feature AI-researched insights from verified industry sources, government notifications, and market reports.
Official References & Regulatory Resources
- European Medicines Agency
- WHO Essential Medicines
- Invest India — Pharma Sector
- India Trade Statistics (DGFT)
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Product Matching: Leflunomide shipments identified using fuzzy matching on DGFT product description fields, validated against Indian Pharmacopoeia nomenclature and HS code classification.
- 2.Buyer Identification: STADAPHARM GMBH matched across shipments using first-3-word normalization of the consignee field, accounting for company name formatting variations.
- 3.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation. This removes the impact of unusually large one-off transactions that could distort buyer rankings.
- 4.Market Share: Calculated as STADAPHARM GMBH's capped import value divided by total capped export value for Leflunomide across all Indian exporters.
- 5.Verified Records: This page is backed by 31 individual customs records matching STADAPHARM GMBH importing Leflunomide, covering 13 formulations from 1 verified supplier.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
Global Coverage
Exports to 63+ countries, 202+ buyers
Expert-Reviewed
Analyzed by pharmaceutical trade specialists
Get Leflunomide Import Data for STADAPHARM GMBH
Access order-level details, pricing data, supplier connections, and competitive analysis for STADAPHARM GMBH's Leflunomide imports. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
STADAPHARM GMBH
Company Profile
Multiple products · $0 total trade · Multiple categories
STADAPHARM GMBH × Leflunomide
Verified from customs records
Related Analysis
Need Detailed Import Data?
Access shipment-level records, supplier contacts, and formulation details for STADAPHARM GMBH.
Request DemoView Pricing →Data Source
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Market share calculated relative to total Indian pharmaceutical exports for Leflunomide. For current order-level data, contact TransData Nexus.