Italy to India Pharmaceutical Import
Bilateral Trade Intelligence · $471.9M Total Trade · 133 Foreign Suppliers · 88 Indian Buyers · DGFT Verified · Updated March 2026
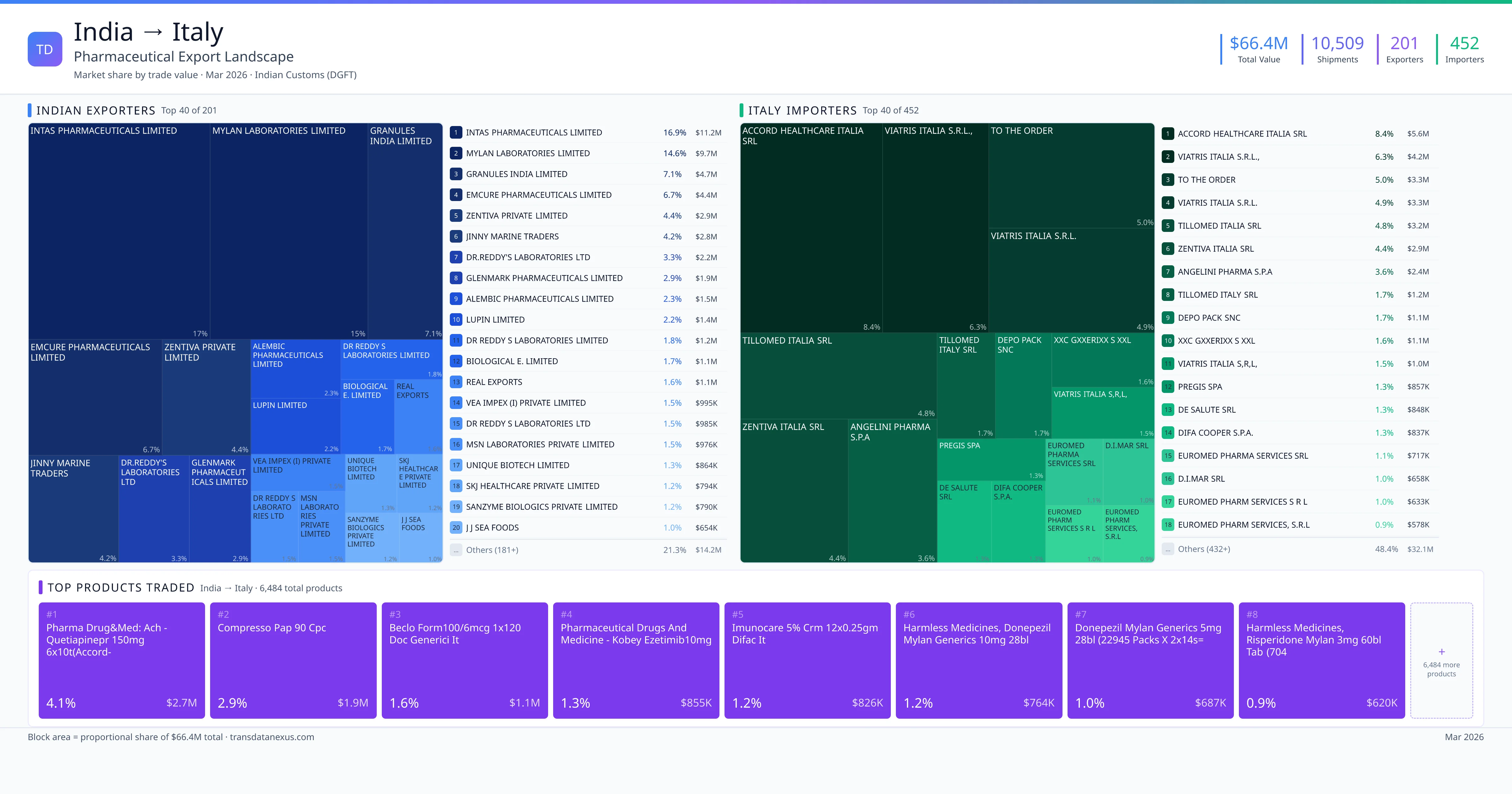
India imported $471.9M worth of pharmaceutical formulations from Italy across 1,710 verified shipments, from 133 foreign suppliers to 88 Indian buyers. The top suppliers are PFIZER SERVICE COMPANY BV ($79.5M) and Takeda Pharmaceuticals International AG ($71.0M). The leading products are Valsartan ($103.0M) and Serum ($84.5M). Average shipment value: $275.9K.
Top Pharmaceutical Formulations — Italy to India
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Valsartan | $103.0M | 21.8% |
| 2 | Serum | $84.5M | 17.9% |
| 3 | Insulin | $78.0M | 16.5% |
| 4 | Prednisolone | $57.5M | 12.2% |
| 5 | Methylprednisolone | $49.0M | 10.4% |
| 6 | Vaccine | $20.8M | 4.4% |
| 7 | Teicoplanin | $20.0M | 4.2% |
| 8 | Canagliflozin | $18.0M | 3.8% |
| 9 | Sodium | $10.1M | 2.1% |
| 10 | Cefuroxime | $9.6M | 2.0% |
| 11 | Sunitinib | $5.0M | 1.1% |
| 12 | Darunavir | $3.1M | 0.7% |
| 13 | Mycophenolate | $2.7M | 0.6% |
| 14 | Budesonide | $2.5M | 0.5% |
| 15 | Protein | $1.8M | 0.4% |
India imports 20+ pharmaceutical formulations from Italy with a combined trade value of $471.9M. Key products include Valsartan ($103.0M), Serum ($84.5M), Insulin ($78.0M), Prednisolone ($57.5M), Methylprednisolone ($49.0M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 1,710 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — Italy to India Trade Routes
These are the top pharmaceutical products exported from India to Italy, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Italy buyers, regulatory requirements, and logistics for that specific product corridor. Products include Valsartan ($103.0M), Serum ($84.5M), Insulin ($78.0M), Prednisolone ($57.5M), Methylprednisolone ($49.0M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Italy Suppliers to India
133 Indian pharmaceutical companies export finished formulations to Italy. Leading exporters include Pfizer Service Company Bv, Takeda Pharmaceuticals International Ag, Novartis Pharma Ag, Eli Lilly Export S.a.. The top exporter accounts for 16.8% of total India–Italy pharma exports. Source: Indian Customs (DGFT).
Top Indian Buyers from Italy
88 companies in Italy import pharmaceutical formulations from India. Top buyers include Pfizer Products India Private Limited, Sandoz Private Limited, Takeda Biopharmaceuticals India Private Limited, Eli Lilly And Company (india ) Private Limited. The largest buyer accounts for 20.6% of India–Italy pharma imports. Source: Indian Customs (DGFT).
Trade Statistics
Other Import Origins
Product Routes
India–Italy Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade corridor between Italy and India has experienced significant growth over the past decade. In 2022, the total import value stood at $471.9 million USD, with 1,710 shipments from 133 Italian suppliers to 88 Indian buyers. This reflects a robust and expanding trade relationship, underscoring Italy's increasing role in supplying pharmaceutical products to the Indian market.
Key milestones in this evolution include the establishment of streamlined regulatory processes and enhanced logistical frameworks. The introduction of the CDSCO's SUGAM portal in 2016 facilitated more efficient registration and approval procedures for foreign pharmaceutical companies, including those from Italy. This digitalization has significantly reduced processing times and improved transparency in the import process.
2Italy's Role in India's Pharma Imports
Italy has become a significant contributor to India's pharmaceutical imports, with a total import value of $471.9 million USD over the past five years. The top 10 imported products from Italy include valsartan ($103.0 million, 21.8%), serum ($84.5 million, 17.9%), insulin ($78.0 million, 16.5%), and prednisolone ($57.5 million, 12.2%). These products highlight Italy's strength in supplying essential medications, particularly in the cardiovascular and endocrine therapeutic areas.
Italian pharmaceutical companies such as Pfizer Service Company BV, Takeda Pharmaceuticals International AG, and Novartis Pharma AG have been instrumental in this trade, supplying a diverse range of products to meet India's growing healthcare needs. Their consistent presence underscores the trust and demand for Italian pharmaceutical products in the Indian market.
3Recent Developments
Between 2024 and 2026, several developments have influenced the Italy-India pharmaceutical trade corridor. The CDSCO has implemented policy changes to streamline the registration process for foreign pharmaceutical companies, including those from Italy. The SUGAM portal has been enhanced to support electronic submissions, reducing processing times and improving transparency.
Additionally, trade agreements between India and the European Union have been under negotiation, aiming to reduce tariffs and non-tariff barriers. These agreements are expected to further facilitate the import of pharmaceutical products from Italy to India, promoting a more integrated and efficient trade relationship.
Italy Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1CDSCO Registration Process
Italian pharmaceutical companies seeking to export products to India must navigate the CDSCO's registration process. The first step involves registering on the SUGAM portal, where applicants must provide details such as the Corporate Identification Number (CIN), Customs House Agent (CHA) information, and authorized personnel details. Required documents include an undertaking form, valid address proof of the firm (e.g., Certificate of Incorporation, GST Certificate), and a valid ID proof of the authorized person.
Once registered, companies can submit applications for import licenses using Form 8, accompanied by an undertaking in Form 9. The CDSCO reviews these applications to ensure compliance with Indian regulations. For products requiring clinical trials, additional documentation such as clinical trial data and ethical committee approvals are necessary.
2GMP & Manufacturing Standards
India requires that foreign manufacturing sites adhere to Good Manufacturing Practices (GMP) standards as outlined by the World Health Organization (WHO). Italian manufacturers must provide a GMP certificate issued by the National Regulatory Authority of their country, ensuring that their facilities meet international quality standards. The CDSCO may conduct inspections to verify compliance with these standards.
3Import Documentation
To import pharmaceutical products into India, Italian companies must obtain an import license (Form 10) from the CDSCO. This requires submitting a registration certificate, a No Objection Certificate (NOC) from the CDSCO, a test license, and adhering to customs procedures. The SUGAM portal facilitates the submission of these documents, streamlining the approval process.
Product Categories & Therapeutic Trends — India to Italy
Dominant categories, emerging opportunities, and demand drivers
1Dominant Import Categories
India's pharmaceutical imports from Italy are predominantly in the form of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. The top 10 imported products reflect a strong demand for cardiovascular drugs like valsartan, endocrine therapies such as insulin and prednisolone, and biologics like serum. This trend indicates Italy's specialization in supplying high-quality, essential medications that complement India's domestic production capabilities.
2Innovation & Specialty Imports
Italian pharmaceutical companies are known for their innovative approaches, offering novel formulations and advanced therapies not yet manufactured domestically in India. Products like teicoplanin, a glycopeptide antibiotic, and canagliflozin, a sodium-glucose co-transporter 2 (SGLT2) inhibitor for diabetes, exemplify Italy's contribution to India's healthcare sector by providing specialized treatments that address specific medical needs.
3Import Demand Drivers
India's import demand from Italy is driven by several factors, including the need for patented drugs not available locally, technological advancements in pharmaceutical manufacturing, and stringent quality requirements. The high disease burden in India, particularly in areas like cardiovascular diseases and diabetes, further propels the demand for effective treatments, many of which are supplied by Italian pharmaceutical companies.
Trade Policy & Tariff Intelligence — India and Italy
Tariff structure, trade agreements, IP and patent landscape
1India's Import Tariff Structure
India's import tariff structure for pharmaceutical formulations includes the Most Favored Nation (MFN) tariff rates, basic customs duty, Integrated Goods and Services Tax (IGST), and health cess. These duties are subject to change based on government policies and trade agreements. Pharmaceutical imports from Italy are typically classified under specific HS codes, which determine the applicable duty rates. Importers should consult the latest customs notifications and the Directorate General of Foreign Trade (DGFT) guidelines for accurate and up-to-date information.
2Trade Agreements & Preferences
India and Italy, as part of the European Union, are engaged in trade agreements aimed at reducing tariffs and non-tariff barriers. These agreements are expected to facilitate the import of pharmaceutical products from Italy to India by providing preferential duty rates and streamlining customs procedures. The Regional Comprehensive Economic Partnership (RCEP) and other bilateral negotiations also play a role in shaping the trade dynamics between the two countries.
3IP, Patents & Price Control
India's patent regime, particularly Section 3(d) of the Indian Patents Act, allows for the granting of compulsory licenses under certain conditions, such as public health emergencies. The National Pharmaceutical Pricing Authority (NPPA) regulates the prices of essential drugs, including imported formulations, to ensure affordability. Imported drugs are subject to price controls, which can impact the pricing strategies of Italian pharmaceutical companies exporting to India.
Supply Chain & Logistics — India to Italy Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical products from Italy to India are primarily transported via sea and air routes. Major Italian export ports include Genoa and Naples, while Indian import ports handling pharmaceutical shipments include Mumbai, Chennai, and Kolkata. Sea freight typically takes 20-30 days, depending on the shipping line and route, while air freight is faster, usually taking 5-7 days. The reliability of these routes is generally high, with established shipping lines and air cargo services ensuring timely deliveries.
2Port Infrastructure
Italian export ports like Genoa and Naples are well-equipped to handle pharmaceutical exports, offering facilities that comply with international shipping standards. In India, ports such as Mumbai, Chennai, and Kolkata have dedicated terminals for pharmaceutical imports, equipped with cold storage facilities to maintain the integrity of temperature-sensitive products. These ports adhere to the guidelines set by the Directorate General of Shipping and the Directorate General of Foreign Trade (DGFT) to ensure efficient handling and clearance of pharmaceutical shipments.
3Indian Customs & Cold Chain
Upon arrival at Indian ports, pharmaceutical imports undergo customs clearance procedures, which include documentation verification, inspection, and testing by the CDSCO to ensure compliance with Indian regulations. Cold chain management is critical for temperature-sensitive products; therefore, importers must ensure that shipments are accompanied by a valid Certificate of Analysis and comply with the Good Distribution Practice (GDP) guidelines. The CDSCO may conduct inspections to verify compliance with these standards, and any discrepancies can lead to delays or rejections of shipments.
Market Opportunity Assessment — Italy for Indian Pharma
Market size, healthcare system, growth outlook
1India's Import Dependency
Despite being a major manufacturer of generic pharmaceuticals, India imports a significant portion of its pharmaceutical requirements from Italy. This dependency is largely due to the need for patented drugs, advanced biologics, and specialized formulations that are not yet produced domestically. Italy's strong pharmaceutical industry, characterized by innovation and high-quality standards, positions it as a key supplier to meet these specific needs in the Indian market.
2Italy Companies in India
Major Italian pharmaceutical companies, such as Pfizer Service Company BV, Takeda Pharmaceuticals International AG, and Novartis Pharma AG, have established a strong presence in India. These companies employ strategies like direct exports, partnerships with local
FAQ — India to Italy Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Italy?
India exported pharmaceuticals worth $471.9M to Italy across 1,710 verified shipments.
Who are the top Indian pharmaceutical exporters to Italy?
1. PFIZER SERVICE COMPANY BV — $79.5M. 2. Takeda Pharmaceuticals International AG — $71.0M. 3. NOVARTIS PHARMA AG — $67.5M. Total: 133 suppliers.
Which companies in Italy import pharmaceuticals from India?
1. PFIZER PRODUCTS INDIA PRIVATE LIMITED — $97.0M. 2. SANDOZ PRIVATE LIMITED — $72.0M. 3. TAKEDA BIOPHARMACEUTICALS INDIA PRIVATE LIMITED — $62.5M. 88 buyers total.
What pharmaceutical products does India export most to Italy?
1. Valsartan ($103.0M, 21.8%); 2. Serum ($84.5M, 17.9%); 3. Insulin ($78.0M, 16.5%); 4. Prednisolone ($57.5M, 12.2%); 5. Methylprednisolone ($49.0M, 10.4%)
Which ports handle pharmaceutical shipments from India to Italy?
Export: . Import: .
Why does Italy import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $471.9M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Italy?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Italy pharmaceutical trade?
$275.9K per consignment across 1,710 shipments.
How many Indian pharmaceutical companies export to Italy?
133 Indian companies. Largest: PFIZER SERVICE COMPANY BV with $79.5M.
How can I find verified Indian pharmaceutical suppliers for Italy?
TransData Nexus covers 133 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Italy Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 1,710 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Italy identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 1,710 verified shipments from 133 Indian exporters to 88 Italy buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
133 Exporters
88 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists