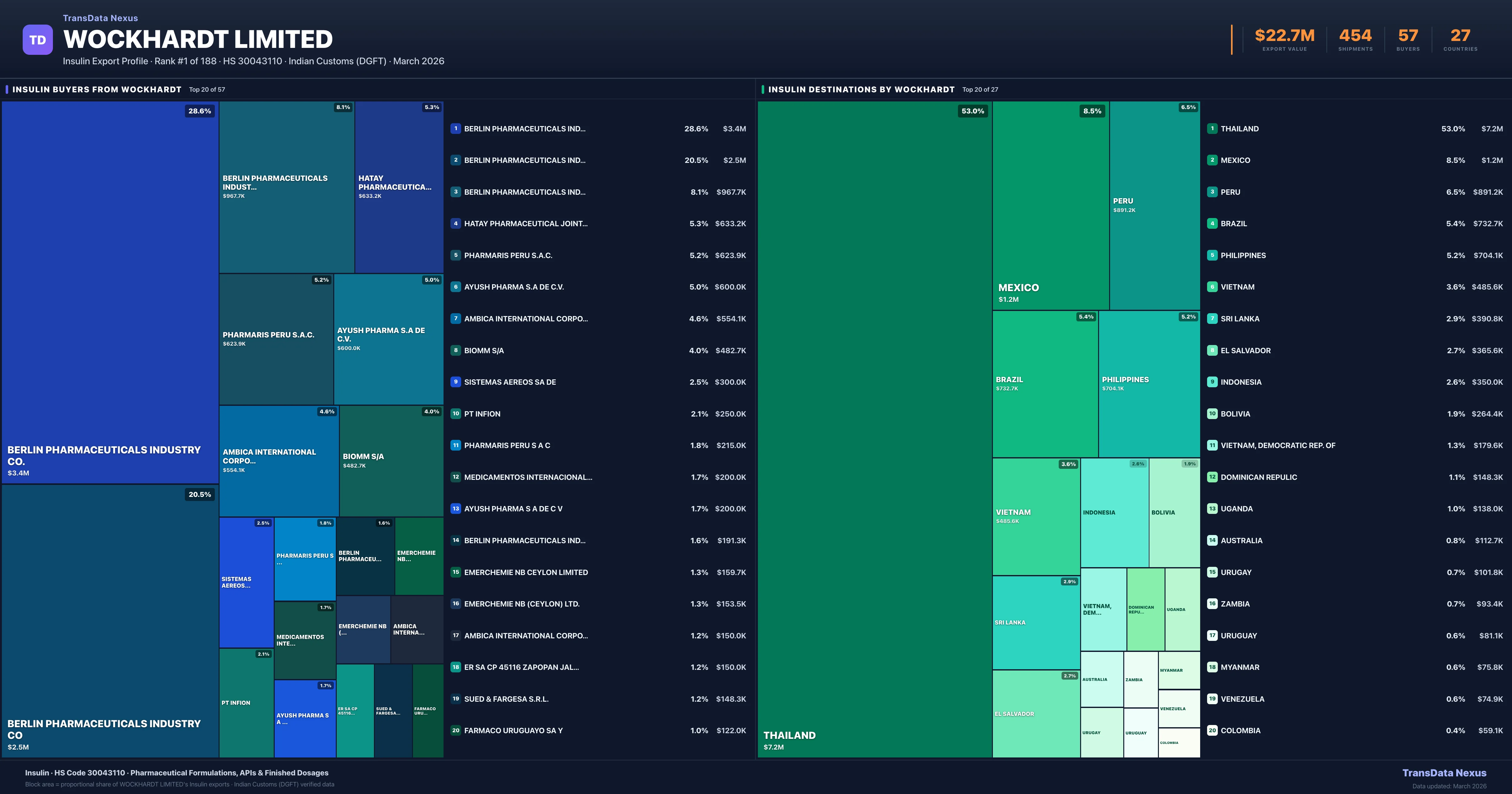
Geopolitical & Trade Policy Impact on Wockhardt Limited's Insulin Exports
Wockhardt's export operations are influenced by various geopolitical factors. The Israel-Iran conflict has disrupted Red Sea shipping routes, leading to increased shipping costs and extended transit times for Indian pharmaceutical exporters. These disruptions pose challenges for time-sensitive products, particularly those requiring stringent cold-chain logistics. (pharmaceuticalcommerce.com)
In the U.S., recent tariff policies have introduced additional complexities. In August 2025, the U.S. imposed broad tariffs on imports from over 60 countries, including India, with rates reaching up to 39%. These tariffs have impacted nearly 55% of India's exports to the U.S., placing Indian pharmaceutical companies at a competitive disadvantage. (apnews.com)
Conversely, the U.S.-China trade tensions have created opportunities for Indian pharmaceutical companies to fill supply gaps in the American market. However, the overall impact of U.S. tariff policies remains a significant concern for exporters like Wockhardt.
In the European Union, compliance with the Falsified Medicines Directive (FMD) has become increasingly stringent. Indian exporters must invest in advanced serialization and traceability systems to meet these requirements, ensuring the authenticity and safety of pharmaceutical products in the EU market.
Wockhardt Limited — Regulatory Compliance & Quality Standards
The global pharmaceutical industry is witnessing heightened regulatory scrutiny, with agencies like the U.S. FDA and the European Medicines Agency (EMA) enforcing stringent quality standards. India, housing over 600 U.S. FDA-approved manufacturing sites, faces challenges in maintaining compliance. In 2023, the U.S. FDA conducted more than 200 inspections in India, with plans to increase this number in 2024. (fact.net.in)
For Wockhardt, adherence to Good Manufacturing Practices (GMP) and other regulatory standards is crucial to sustain its export operations. The company's strategic focus on high-impact areas necessitates continuous investment in quality control systems and infrastructure to meet evolving global standards.