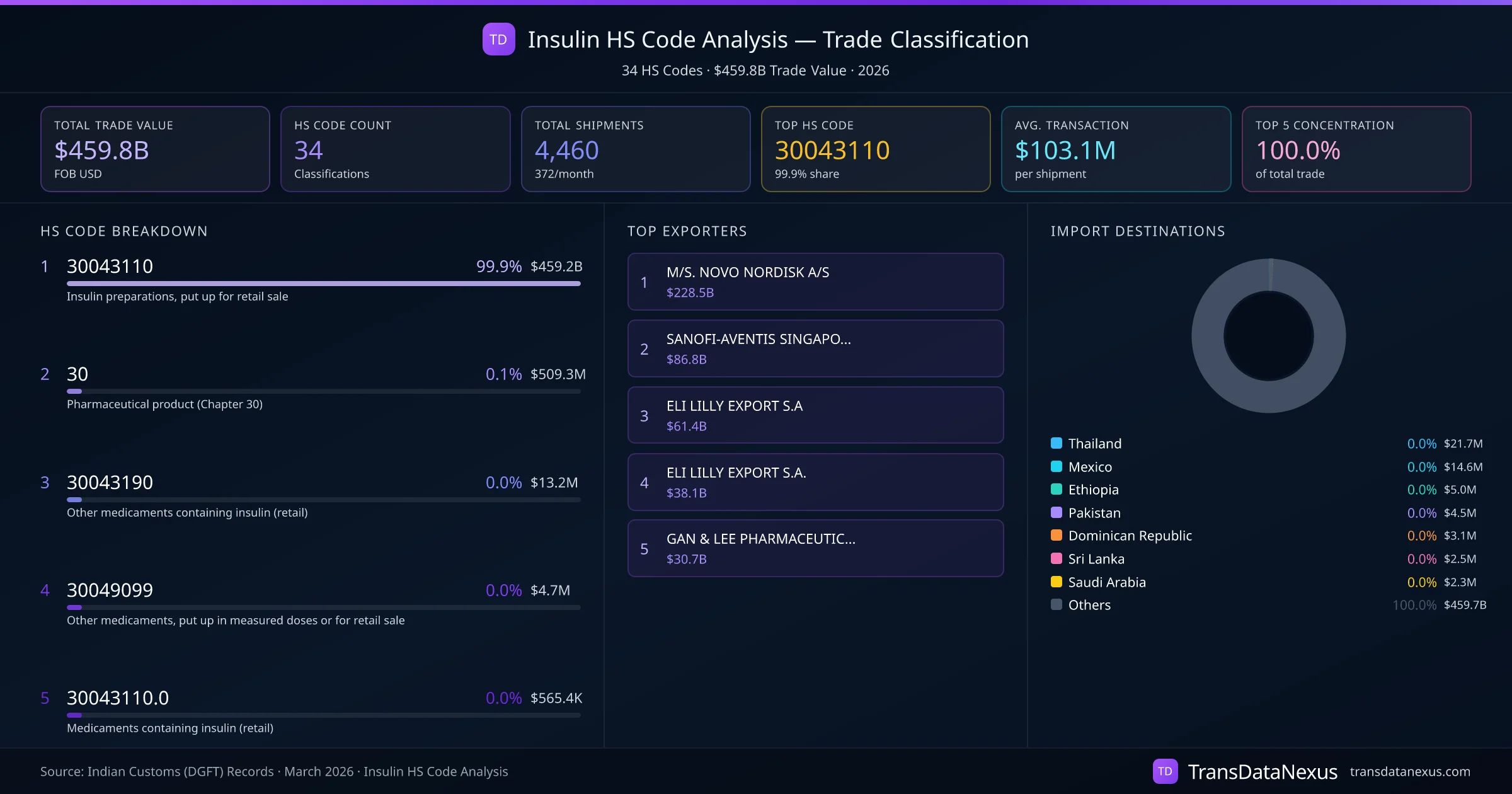
Insulin products are primarily classified under HS code 30043110, which covers medicaments containing insulin, packaged for retail sale. However, classification disputes can arise when determining whether insulin should be classified under HS code 3003 or 3004.
- HS Code 3003: This code pertains to medicaments consisting of two or more constituents mixed together for therapeutic or prophylactic uses, not put up in measured doses or in forms or packings for retail sale. Insulin preparations intended for further processing or bulk supply, not in final retail packaging, fall under this category.
- HS Code 3004: This code includes medicaments consisting of mixed or unmixed products for therapeutic or prophylactic uses, put up in measured doses or in forms or packings for retail sale. Finished insulin products ready for consumer use are classified here.
Common misclassification errors occur when bulk insulin intended for further processing is incorrectly classified under HS 3004 instead of HS 3003. Such misclassifications can lead to customs penalties, including fines and delays in shipment clearance. For instance, a U.S. Customs ruling (NY L80595) dated November 10, 2004, classified five PHASULIN® human insulin preparations in glass vials for injection under subheading 3004.31.0000, emphasizing the importance of correct classification based on packaging and intended use.
Given that 99.9% of insulin trade utilizes HS code 30043110, it is crucial for importers and exporters to ensure accurate classification to avoid regulatory issues.