Who Buys Insulin from India — 384 Importers Behind a $76.0M Market
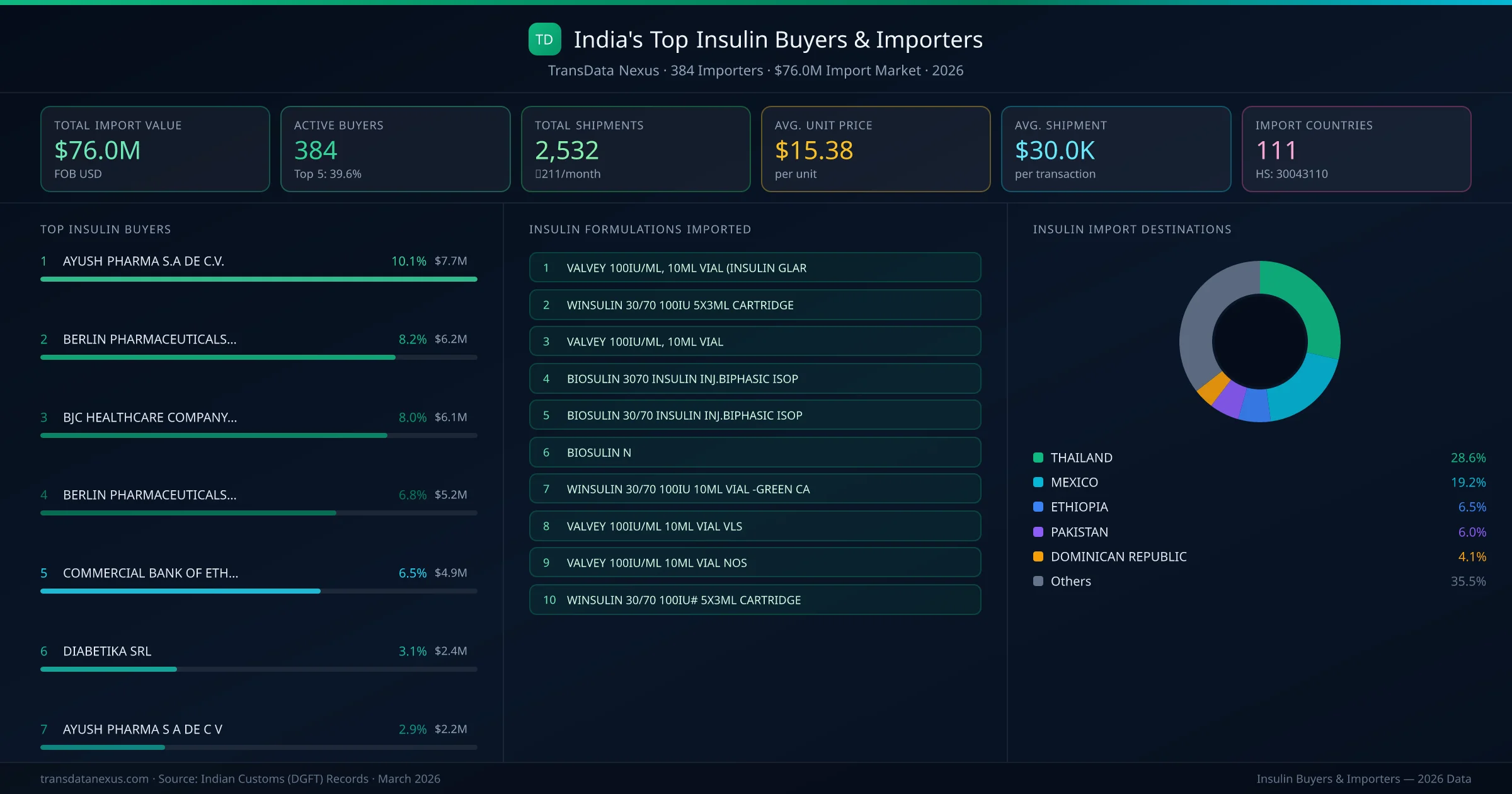
India's insulin import market is served by 384 active buyers who collectively imported $76.0M across 2,532 shipments. AYUSH PHARMA S.A DE C.V. (MEXICO) leads with a 10.1% market share, followed by BERLIN PHARMACEUTICALS INDUSTRY CO. and BJC HEALTHCARE COMPANY LIMITED. The top 5 buyers together control 39.6% of total import value, reflecting a moderately competitive buyer landscape.
Top Insulin Importers — Ranked by Import Value
AYUSH PHARMA S.A DE C.V. (MEXICO) is the leading insulin importer from India, holding a 10.1% share of the $76.0M market across 2,532 shipments from 384 buyers. The top 5 buyers — AYUSH PHARMA S.A DE C.V. (MEXICO), BERLIN PHARMACEUTICALS INDUSTRY CO. (THAILAND), BJC HEALTHCARE COMPANY LIMITED (THAILAND), BERLIN PHARMACEUTICALS INDUSTRY CO (THAILAND), COMMERCIAL BANK OF ETHIOPIA (ETHIOPIA) — collectively control 39.6% of total import value.
Top Insulin Buyers & Importers
Ranked by import value · 384 active buyers · Indian Customs (DGFT) data
| # | Buyer & Formulations | Value | Suppliers | Share |
|---|---|---|---|---|
| 1 | AYUSH PHARMA S.A DE C.V.MEXICO VALVEY 100IU/ML, 10ML VIAL (INSULIN GLARVALVEY 100IU/ML, 10ML VIALVALVEY 100IU/ML 10ML VIAL VLS | $7.7M | 5 | 10.1% |
| 2 | BERLIN PHARMACEUTICALS INDUSTRY CO.THAILAND WINSULIN 30/70 100IU 5X3ML CARTRIDGEWINSULIN 30/70 100IU 10ML VIAL -GREEN CAWINSULIN 30/70 100IU# 5X3ML CARTRIDGE | $6.2M | 5 | 8.2% |
| 3 | BJC HEALTHCARE COMPANY LIMITEDTHAILAND WINSULIN 30/70 100IU 5X3ML CARTRIDGEWINSULIN 30/70 100IU 10ML VIAL -GREEN CAWINSULIN 30/70 100IU# 5X3ML CARTRIDGE | $6.1M | 5 | 8.0% |
| 4 | BERLIN PHARMACEUTICALS INDUSTRY COTHAILAND WINSULIN 30/70 100IU 5X3ML CARTRIDGEWINSULIN 30/70 100IU 10ML VIAL -GREEN CAWINSULIN 30/70 100IU# 5X3ML CARTRIDGE | $5.2M | 5 | 6.8% |
| 5 | COMMERCIAL BANK OF ETHIOPIAETHIOPIA BIOSULIN 30/70 INSULIN INJ.BIPHASIC ISOPBIOSULIN NBIOSULIN 3070 INSULIN INJ.BIPHASIC ISOP | $4.9M | 5 | 6.5% |
| 6 | DIABETIKA SRLDOMINICAN REPUBLIC BIOSULIN 30/70 INSULIN INJECTION BIPHASIBIOSULIN N INSULIN INJECTION ISOPHANE PH | $2.4M | 5 | 3.1% |
| 7 | AYUSH PHARMA S A DE C VMEXICO VALVEY 100IU/ML, 10ML VIAL (INSULIN GLARVALVEY 100IU/ML, 10ML VIALVALVEY 100IU/ML 10ML VIAL VLS | $2.2M | 5 | 2.9% |
| 8 | BERLIN PHARMACEUTICALS INDUSTRY CO,THAILAND WINSULIN 30/70 100IU 5X3ML CARTRIDGEWINSULIN 30/70 100IU 10ML VIAL -GREEN CAWINSULIN 30/70 100IU# 5X3ML CARTRIDGE | $2.0M | 5 | 2.7% |
| 9 | INNODRIVE GROUP S.A DE CVMEXICO VALVEY 100IU/ML, 10ML VIAL (INSULIN GLARVALVEY 100IU/ML, 10ML VIALVALVEY 100IU/ML 10ML VIAL VLS | $2.0M | 5 | 2.7% |
| 10 | PHARMARIS PERU S.A.C.PERU GLARITUS 100IU/ML, 10ML VIALGLARITUS 10ML VIALWOSULIN N 100IU/ML 10ML VIAL | $1.7M | 5 | 2.3% |
| 11 | ELI LILLY PAKISTAN (PVT ) LTDPAKISTAN | $1.7M | 5 | 2.2% |
| 12 | ELI LILLY PAKISTAN (PRIVATE) LTDPAKISTAN | $1.6M | 5 | 2.0% |
| 13 | NEW GPC INCGUYANA | $1.2M | 5 | 1.6% |
| 14 | BANK OF CEYLONSRI LANKA BIOSULIN 30/70 INSULIN INJ.BIPHASIC ISOPBIOSULIN NBIOSULIN 3070 INSULIN INJ.BIPHASIC ISOP | $1.2M | 5 | 1.6% |
| 15 | SISTEMAS AEREOS SA DEEL SALVADOR | $1.2M | 5 | 1.6% |
Related Analysis
Market Analysis: Top Importing Countries & Importers
Comprehensive geographic market intelligence dashboard
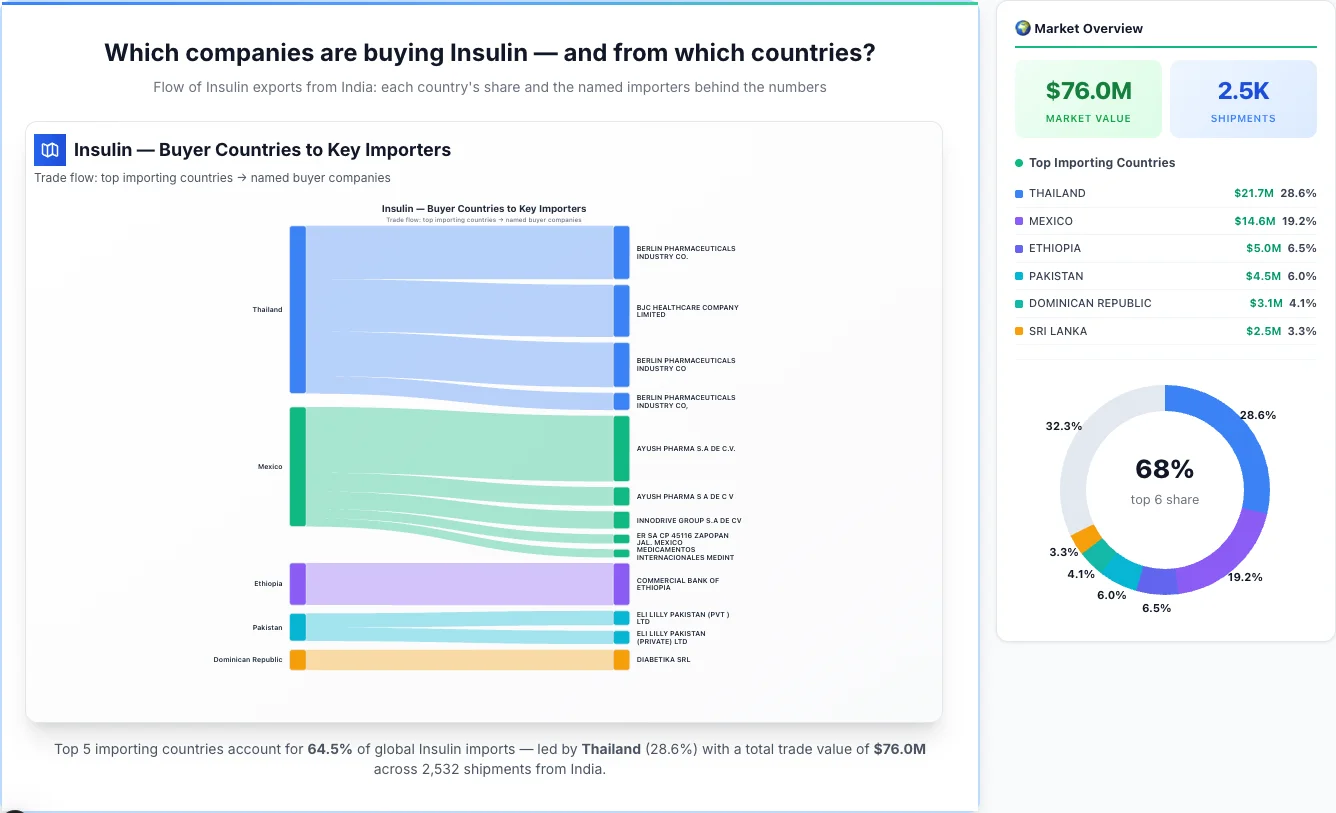
Which companies are buying Insulin — and from which countries?
Flow of Insulin exports from India: each country's share and the named importers behind the numbers
Insulin — Buyer Countries to Key Importers
Trade flow: top importing countries → named buyer companies
Market Analysis: Top Importing Countries & Their Importers
1Thailand
Thailand emerges as the first largest importing country, achieving a total trade value of $21.7M through 339 shipments. This represents a market share of 28.6% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Thailand has a diverse importer base, with their top importers including:
• BERLIN PHARMACEUTICALS INDUSTRY CO. - $6.2M (28.6% of country's total business)
• BJC HEALTHCARE COMPANY LIMITED - $6.1M (28.0% of country's total business)
• BERLIN PHARMACEUTICALS INDUSTRY CO - $5.2M (23.9% of country's total business)
• BERLIN PHARMACEUTICALS INDUSTRY CO, - $2.0M (9.3% of country's total business)
Key Insight: Thailand demonstrates strong market positioning with an average shipment value of $64.1K, indicating premium pricing strategy compared to the market average of $30.0K.
Thailand is a key market for Insulin imports, representing 28.6% of total trade value.
2Mexico
Mexico emerges as the second largest importing country, achieving a total trade value of $14.6M through 31 shipments. This represents a market share of 19.2% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Mexico has a diverse importer base, with their top importers including:
• AYUSH PHARMA S.A DE C.V. - $7.7M (52.4% of country's total business)
• AYUSH PHARMA S A DE C V - $2.2M (14.9% of country's total business)
• INNODRIVE GROUP S.A DE CV - $2.0M (13.9% of country's total business)
• ER SA CP 45116 ZAPOPAN JAL. MEXICO - $1.1M (7.4% of country's total business)
• MEDICAMENTOS INTERNACIONALES MEDINT - $976.1K (6.7% of country's total business)
• Rest of Mexico's importers - $700.2K (4.8% of country's total business)
Key Insight: Mexico demonstrates strong market positioning with an average shipment value of $472.0K, indicating premium pricing strategy compared to the market average of $30.0K.
Mexico is a key market for Insulin imports, representing 19.2% of total trade value.
3Ethiopia
Ethiopia emerges as the third largest importing country, achieving a total trade value of $5.0M through 13 shipments. This represents a market share of 6.5% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Ethiopia has a diverse importer base, with their top importers including:
• COMMERCIAL BANK OF ETHIOPIA - $4.9M (98.8% of country's total business)
Key Insight: Ethiopia demonstrates strong market positioning with an average shipment value of $382.2K, indicating premium pricing strategy compared to the market average of $30.0K.
Ethiopia is a key market for Insulin imports, representing 6.5% of total trade value.
4Pakistan
Pakistan emerges as the fourth largest importing country, achieving a total trade value of $4.5M through 59 shipments. This represents a market share of 6.0% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Pakistan has a diverse importer base, with their top importers including:
• ELI LILLY PAKISTAN (PVT ) LTD - $1.7M (36.8% of country's total business)
• ELI LILLY PAKISTAN (PRIVATE) LTD - $1.6M (34.3% of country's total business)
Key Insight: Pakistan demonstrates strong market positioning with an average shipment value of $77.1K, indicating premium pricing strategy compared to the market average of $30.0K.
Pakistan is a key market for Insulin imports, representing 6.0% of total trade value.
5Dominican Republic
Dominican Republic emerges as the fifth largest importing country, achieving a total trade value of $3.1M through 36 shipments. This represents a market share of 4.1% of the total market activity, demonstrating their significant presence in the industry.
Top Importers: Dominican Republic has a diverse importer base, with their top importers including:
• DIABETIKA SRL - $2.4M (76.7% of country's total business)
Key Insight: Dominican Republic demonstrates strong market positioning with an average shipment value of $86.7K, indicating premium pricing strategy compared to the market average of $30.0K.
Dominican Republic is a key market for Insulin imports, representing 4.1% of total trade value.
Buyer Segment Analysis
TransData Nexus Insulin buyer market intelligence
1Buyer Segment Analysis
The analysis of insulin imports from India reveals a diverse buyer landscape, encompassing several key segments:
- Distributors: Entities such as AYUSH PHARMA S.A DE C.V. (Mexico) and DIABETIKA SRL (Dominican Republic) function as intermediaries, procuring insulin in bulk to supply to various healthcare providers and pharmacies within their respective regions. Their purchasing patterns typically involve large, periodic orders to maintain a steady supply chain.
- Hospital Groups: Organizations like BJC HEALTHCARE COMPANY LIMITED (Thailand) represent hospital networks that import insulin directly to meet the needs of their inpatient and outpatient services. Their procurement is characterized by consistent, scheduled orders aligned with patient demand and treatment protocols.
- Government Procurement: The COMMERCIAL BANK OF ETHIOPIA (Ethiopia) likely acts on behalf of government health initiatives, facilitating large-scale purchases of insulin for national distribution through public healthcare facilities. This segment's buying behavior is often driven by public health policies and tender-based purchasing systems.
- Retail Chains: Entities such as INNODRIVE GROUP S.A DE CV (Mexico) may represent pharmacy chains that import insulin to stock their retail outlets, ensuring availability for consumers. Their order patterns are influenced by consumer demand and inventory turnover rates.
- Re-exporters: Some buyers might engage in re-exporting activities, purchasing insulin from India to supply neighboring countries or regions lacking direct access. This segment's purchasing behavior is opportunistic, capitalizing on market demands and regulatory environments.
- Contract Manufacturers: While not explicitly identified among the top buyers, contract manufacturers may import insulin for formulation, packaging, or further processing before distribution. Their procurement is typically project-based, aligned with contractual obligations and production schedules.
Understanding these segments is crucial for tailoring market strategies, forecasting demand, and ensuring compliance with diverse regulatory requirements across importing countries.
Country-Specific Import Regulations
TransData Nexus Insulin buyer market intelligence
1Country-Specific Import Regulations
Navigating the import regulations for insulin in the top importing countries is essential for market entry and compliance. Below is an overview for each:
Thailand
1. Drug Regulatory Authority: Food and Drug Administration (FDA) Thailand.
2. Import Registration Process: Importers must obtain an import permit from the FDA Thailand. This involves submitting detailed product information, including composition, manufacturing process, and quality control measures.
3. Estimated Timeline for Registration: The registration process can take several months, depending on the completeness of the application and the FDA's review workload.
4. Import Tariff/Duty for HS 30043110: Specific tariff rates should be confirmed with the Thai Customs Department, as they are subject to change and may vary based on trade agreements.
Mexico
1. Drug Regulatory Authority: Comisión Federal para la Protección contra Riesgos Sanitarios (COFEPRIS).
2. Import Registration Process: Importers are required to register the product with COFEPRIS, providing comprehensive documentation on the product's safety, efficacy, and quality.
3. Estimated Timeline for Registration: The registration process typically ranges from 6 to 12 months, contingent upon the complexity of the product and the regulatory body's assessment.
4. Import Tariff/Duty for HS 30043110: Tariff rates can vary; it is advisable to consult the Mexican Customs Authority for the most current information.
Ethiopia
1. Drug Regulatory Authority: Ethiopian Food and Drug Authority (EFDA).
2. Import Registration Process: Importers must register pharmaceutical products with the EFDA, submitting dossiers that include clinical data, manufacturing details, and labeling information.
3. Estimated Timeline for Registration: The process may take approximately 6 to 9 months, depending on the EFDA's review procedures and any additional information requests.
4. Import Tariff/Duty for HS 30043110: Import duties are subject to periodic revisions; consulting the Ethiopian Revenue and Customs Authority is recommended for accurate rates.
Pakistan
1. Drug Regulatory Authority: Drug Regulatory Authority of Pakistan (DRAP).
2. Import Registration Process: Registration with DRAP requires submission of a comprehensive dossier, including product specifications, clinical trial data, and manufacturing practices.
3. Estimated Timeline for Registration: The registration timeline can extend from 12 to 18 months, influenced by the product's complexity and DRAP's evaluation process.
4. Import Tariff/Duty for HS 30043110: Tariff rates are subject to change; it is advisable to verify current rates with the Pakistan Customs Department.
Dominican Republic
1. Drug Regulatory Authority: Dirección General de Medicamentos, Alimentos y Productos Sanitarios (DIGEMAPS).
2. Import Registration Process: Importers must obtain sanitary registration from DIGEMAPS, providing detailed information on the product's composition, manufacturing, and quality control.
3. Estimated Timeline for Registration: The process generally takes between 6 to 12 months, depending on the thoroughness of the application and the regulatory body's review process.
4. Import Tariff/Duty for HS 30043110: Import duties can vary; consulting the Dominican Republic Customs Authority is recommended for the latest rates.
Staying informed about these regulations and maintaining compliance is vital for successful market entry and sustained operations in these countries.
Demand Drivers & Market Opportunity
TransData Nexus Insulin buyer market intelligence
1Demand Drivers & Market Opportunity
The demand for insulin in importing countries is driven by several key factors:
- Disease Prevalence: The global rise in diabetes cases, particularly in low- and middle-income countries (LMICs), significantly increases the need for insulin. (who.int)
- Government Healthcare Programs: Many governments are implementing initiatives to improve diabetes care, including subsidizing insulin costs and enhancing healthcare infrastructure.
- Universal Health Coverage Expansion: Efforts to achieve universal health coverage are leading to increased access to essential medicines, including insulin, thereby boosting demand.
- WHO Essential Medicines List Procurement: The inclusion of insulin in the WHO Essential Medicines List encourages countries to prioritize its procurement, ensuring availability in public health systems.
- Tender-Based Purchasing: Many countries utilize tender systems for bulk purchasing of insulin, aiming to secure lower prices and ensure a consistent supply for their populations.
With 384 buyers across 111 countries, the market for insulin imported from India is extensive. This widespread demand underscores the importance of understanding and addressing the diverse needs and regulatory environments of each importing country to effectively capture market opportunities.
Common Questions — Insulin Buyers & Importers
Answers based on Indian Customs (DGFT) shipment records compiled by TransData Nexus
QWho is the largest insulin buyer importing from India?
Based on import volume and value, AYUSH PHARMA S.A DE C.V. (MEXICO) leads with $7.7M in imports and a 10.1% market share — the highest of any single insulin importer. BERLIN PHARMACEUTICALS INDUSTRY CO. (THAILAND) and BJC HEALTHCARE COMPANY LIMITED (THAILAND) are the next largest buyers.
QHow many companies buy insulin from India?
There are 384 active insulin buyers importing from India, with a combined market of $76.0M across 2,532 shipments to 111 countries. The top 5 buyers hold 39.6% of total import value, while the remaining 379 buyers handle the other 60.4%.
QWhich countries import the most insulin from India?
The top importing countries for insulin from India are Thailand (28.6%), Mexico (19.2%), Ethiopia (6.5%), Pakistan (6.0%), Dominican Republic (4.1%). These markets represent the largest demand centres for Indian pharmaceutical exports of insulin, with buyers ranging from government procurement agencies to private pharmaceutical distributors.
QWhat is the average import order value for insulin from India?
The average import transaction value for insulin from India is $30.0K, with an average unit price of $15.38 per unit. Order sizes vary significantly based on buyer type — government tenders tend to be larger while private distributors place more frequent smaller orders.
Buyer Segment Analysis
TransData Nexus Insulin buyer market intelligence
1Buyer Segment Analysis
The analysis of insulin imports from India reveals a diverse buyer landscape, encompassing several key segments:
- Distributors: Entities such as AYUSH PHARMA S.A DE C.V. (Mexico) and DIABETIKA SRL (Dominican Republic) function as intermediaries, procuring insulin in bulk to supply to various healthcare providers and pharmacies within their respective regions. Their purchasing patterns typically involve large, periodic orders to maintain a steady supply chain.
- Hospital Groups: Organizations like BJC HEALTHCARE COMPANY LIMITED (Thailand) represent hospital networks that import insulin directly to meet the needs of their inpatient and outpatient services. Their procurement is characterized by consistent, scheduled orders aligned with patient demand and treatment protocols.
- Government Procurement: The COMMERCIAL BANK OF ETHIOPIA (Ethiopia) likely acts on behalf of government health initiatives, facilitating large-scale purchases of insulin for national distribution through public healthcare facilities. This segment's buying behavior is often driven by public health policies and tender-based purchasing systems.
- Retail Chains: Entities such as INNODRIVE GROUP S.A DE CV (Mexico) may represent pharmacy chains that import insulin to stock their retail outlets, ensuring availability for consumers. Their order patterns are influenced by consumer demand and inventory turnover rates.
- Re-exporters: Some buyers might engage in re-exporting activities, purchasing insulin from India to supply neighboring countries or regions lacking direct access. This segment's purchasing behavior is opportunistic, capitalizing on market demands and regulatory environments.
- Contract Manufacturers: While not explicitly identified among the top buyers, contract manufacturers may import insulin for formulation, packaging, or further processing before distribution. Their procurement is typically project-based, aligned with contractual obligations and production schedules.
Understanding these segments is crucial for tailoring market strategies, forecasting demand, and ensuring compliance with diverse regulatory requirements across importing countries.
Country-Specific Import Regulations
TransData Nexus Insulin buyer market intelligence
1Country-Specific Import Regulations
Navigating the import regulations for insulin in the top importing countries is essential for market entry and compliance. Below is an overview for each:
Thailand
1. Drug Regulatory Authority: Food and Drug Administration (FDA) Thailand.
2. Import Registration Process: Importers must obtain an import permit from the FDA Thailand. This involves submitting detailed product information, including composition, manufacturing process, and quality control measures.
3. Estimated Timeline for Registration: The registration process can take several months, depending on the completeness of the application and the FDA's review workload.
4. Import Tariff/Duty for HS 30043110: Specific tariff rates should be confirmed with the Thai Customs Department, as they are subject to change and may vary based on trade agreements.
Mexico
1. Drug Regulatory Authority: Comisión Federal para la Protección contra Riesgos Sanitarios (COFEPRIS).
2. Import Registration Process: Importers are required to register the product with COFEPRIS, providing comprehensive documentation on the product's safety, efficacy, and quality.
3. Estimated Timeline for Registration: The registration process typically ranges from 6 to 12 months, contingent upon the complexity of the product and the regulatory body's assessment.
4. Import Tariff/Duty for HS 30043110: Tariff rates can vary; it is advisable to consult the Mexican Customs Authority for the most current information.
Ethiopia
1. Drug Regulatory Authority: Ethiopian Food and Drug Authority (EFDA).
2. Import Registration Process: Importers must register pharmaceutical products with the EFDA, submitting dossiers that include clinical data, manufacturing details, and labeling information.
3. Estimated Timeline for Registration: The process may take approximately 6 to 9 months, depending on the EFDA's review procedures and any additional information requests.
4. Import Tariff/Duty for HS 30043110: Import duties are subject to periodic revisions; consulting the Ethiopian Revenue and Customs Authority is recommended for accurate rates.
Pakistan
1. Drug Regulatory Authority: Drug Regulatory Authority of Pakistan (DRAP).
2. Import Registration Process: Registration with DRAP requires submission of a comprehensive dossier, including product specifications, clinical trial data, and manufacturing practices.
3. Estimated Timeline for Registration: The registration timeline can extend from 12 to 18 months, influenced by the product's complexity and DRAP's evaluation process.
4. Import Tariff/Duty for HS 30043110: Tariff rates are subject to change; it is advisable to verify current rates with the Pakistan Customs Department.
Dominican Republic
1. Drug Regulatory Authority: Dirección General de Medicamentos, Alimentos y Productos Sanitarios (DIGEMAPS).
2. Import Registration Process: Importers must obtain sanitary registration from DIGEMAPS, providing detailed information on the product's composition, manufacturing, and quality control.
3. Estimated Timeline for Registration: The process generally takes between 6 to 12 months, depending on the thoroughness of the application and the regulatory body's review process.
4. Import Tariff/Duty for HS 30043110: Import duties can vary; consulting the Dominican Republic Customs Authority is recommended for the latest rates.
Staying informed about these regulations and maintaining compliance is vital for successful market entry and sustained operations in these countries.
Demand Drivers & Market Opportunity
TransData Nexus Insulin buyer market intelligence
1Demand Drivers & Market Opportunity
The demand for insulin in importing countries is driven by several key factors:
- Disease Prevalence: The global rise in diabetes cases, particularly in low- and middle-income countries (LMICs), significantly increases the need for insulin. (who.int)
- Government Healthcare Programs: Many governments are implementing initiatives to improve diabetes care, including subsidizing insulin costs and enhancing healthcare infrastructure.
- Universal Health Coverage Expansion: Efforts to achieve universal health coverage are leading to increased access to essential medicines, including insulin, thereby boosting demand.
- WHO Essential Medicines List Procurement: The inclusion of insulin in the WHO Essential Medicines List encourages countries to prioritize its procurement, ensuring availability in public health systems.
- Tender-Based Purchasing: Many countries utilize tender systems for bulk purchasing of insulin, aiming to secure lower prices and ensure a consistent supply for their populations.
With 384 buyers across 111 countries, the market for insulin imported from India is extensive. This widespread demand underscores the importance of understanding and addressing the diverse needs and regulatory environments of each importing country to effectively capture market opportunities.
Official References & Regulatory Resources
- European Medicines Agency
- WHO Essential Medicines
- Invest India — Pharma Sector
- India Trade Statistics (DGFT)
Verify import regulations and drug registration requirements with the official agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Identification: 384 global importers of Insulin identified from consignee fields in DGFT shipping bill records.
- 2.Import Value Analysis: Total import value aggregated from 2,532 individual shipping bill records. Values are FOB in USD.
- 3.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 4.Geographic Distribution: Buyer imports distributed across 111 destination countries with market share per buyer calculated from capped values.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2,532 Verified Shipments
384 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists