Who Exports Instrument from India — 77 Suppliers Behind a $1.4M Market
India's instrument export market is supplied by 77 active exporters who collectively shipped $1.4M across 2,733 shipments. MERIL ENDO SURGERY PRIVATE LIMITED leads with a 16.0% market share, followed by EON MEDITECH PRIVATE LIMITED and SHIVA PRODUCTS. The top 5 suppliers together control 55.1% of total export value, reflecting a moderately competitive market structure.
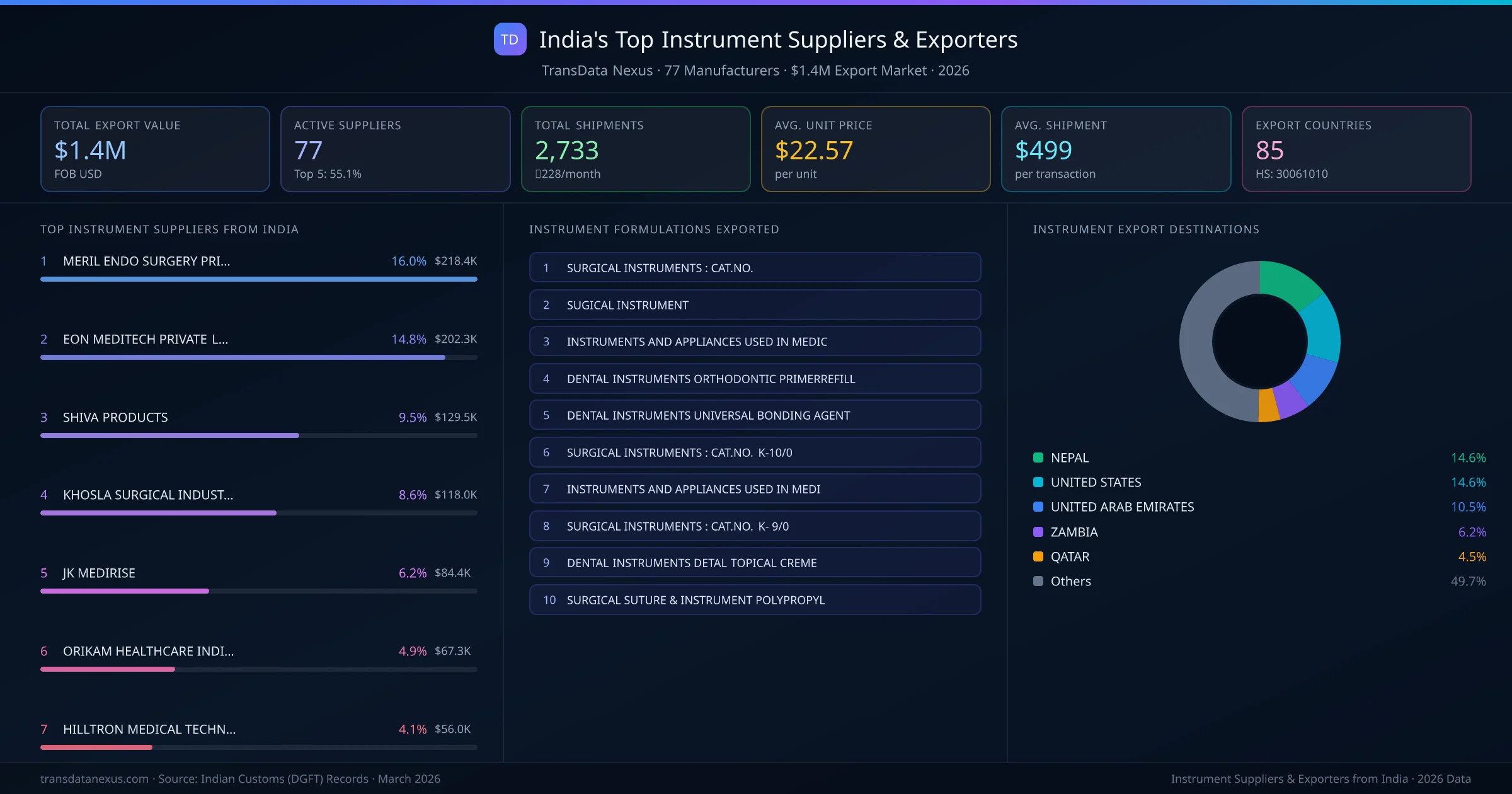
Top Instrument Exporters from India — Ranked by Export Value
MERIL ENDO SURGERY PRIVATE LIMITED is the leading instrument exporter from India, holding a 16.0% share of the $1.4M market across 2,733 shipments from 77 exporters. The top 5 suppliers — MERIL ENDO SURGERY PRIVATE LIMITED, EON MEDITECH PRIVATE LIMITED, SHIVA PRODUCTS, KHOSLA SURGICAL INDUSTRIES, JK MEDIRISE — collectively control 55.1% of total export value, indicating a moderately concentrated market. Individual shares are: MERIL ENDO SURGERY PRIVATE LIMITED (16.0%), EON MEDITECH PRIVATE LIMITED (14.8%), SHIVA PRODUCTS (9.5%), KHOSLA SURGICAL INDUSTRIES (8.6%), JK MEDIRISE (6.2%).
Top Instrument Exporters from India
Ranked by export value · 77 active suppliers · Indian Customs (DGFT) data
| # | Supplier & Formulations | Value | Ctry. | Share |
|---|---|---|---|---|
| 1 | MERIL ENDO SURGERY PRIVATE LIMITED INSTRUMENTS AND APPLIANCES USED IN MEDICINSTRUMENTS AND APPLIANCES USED IN MEDIDENTAL INSTRUMENTS DETAL TOPICAL CREME | $218.4K | 20 | 16.0% |
| 2 | EON MEDITECH PRIVATE LIMITED SURGICAL INSTRUMENT-: NASAL DRESSING WITHOUT STRING CMSURGICAL SUTURE & INSTRUMENT POLYPROPYLINSTRUMENTS AND APPLIANCES USED IN MEDIC | $202.3K | 4 | 14.8% |
| 3 | SHIVA PRODUCTS DENTAL INSTRUMENTS ORTHODONTIC PRIMERREFILLDENTAL INSTRUMENTS LIGHT- AND DUAL-CURE LUTING COMPOSITEDENTAL INSTRUMENTS DETAL TOPICAL CREME | $129.5K | 5 | 9.5% |
| 4 | KHOSLA SURGICAL INDUSTRIES SURGICAL INSTRUMENTS : CAT.NO.SURGICAL INSTRUMENTS : CAT.NO. K-10/0SURGICAL INSTRUMENTS : CAT.NO. K- 9/0 | $118.0K | 15 | 8.6% |
| 5 | JK MEDIRISE INSTRUMENTS AND APPLIANCES USED IN MEDIINSTRUMENTS AND APPLIANCES USED IN MEDICSURGICAL INSTRUMENTS : CAT.NO. | $84.4K | 5 | 6.2% |
| 6 | ORIKAM HEALTHCARE INDIA PRIVATE LIMITED SUGICAL INSTRUMENTDENTAL INSTRUMENTS AND APPARATUS - FILTEK Z350 XT A3 A3.5DENTAL INSTRUMENTS ORTHODONTIC PRIMERREFILL | $67.3K | 11 | 4.9% |
| 7 | HILLTRON MEDICAL TECHNOLOGY DENTAL INSTRUMENTS ORTHODONTIC PRIMERREFILLDENTAL INSTRUMENTS LIGHT- AND DUAL-CURE LUTING COMPOSITEDENTAL INSTRUMENTS DETAL TOPICAL CREME | $56.0K | 2 | 4.1% |
| 8 | ABM4 TRADES DENTAL INSTRUMENTS AND APPARATUS - FILTEK Z350 XT A3 A3.5DENTAL INSTRUMENTS AND APPARATUS - 56877RELYX U200 RF TRDENTAL MATERIALS & INSTRUMENTS: IPS E.MA | $39.2K | 1 | 2.9% |
| 9 | BEACON DIAGNOSTICS PVT LTD | $36.6K | 12 | 2.7% |
| 10 | AADPRI OPHTHALMIC COMPANY TRYPAN BLUE SOLUTION BRAND: TECNO INSTRUMENTS BLUE | $35.4K | 1 | 2.6% |
| 11 | VIRA CORPORATION SURGICAL INSTRUMENTS : CAT.NO.SURGICAL INSTRUMENTS : CAT.NO. K-10/0SURGICAL INSTRUMENTS : CAT.NO. K- 9/0 | $33.0K | 2 | 2.4% |
| 12 | MANARTI EXPORTS | $21.0K | 2 | 1.5% |
| 13 | PRECISION BIOMED PRIVATE LIMITED SURGICAL SUTURE & INSTRUMENT POLYPROPYLINSTRUMENTS AND APPLIANCES USED IN MEDICPGLA VIO 1 X 90-40MM HC RB TN / MITSU MATERIAL CODE | $19.9K | 5 | 1.5% |
Related Analysis
Supplier Certification & Compliance Matrix
FDA, WHO-GMP, and EU GMP status for top Instrument exporters
| Supplier | US FDA | WHO-GMP | EU GMP | ANDAs | Notes |
|---|---|---|---|---|---|
| Meril Endo Surgery Private Limited | Not Listed | Yes (as of December | No | Not verified | WHO-GMP certified for Levonorgestrel-containing hormonal intrauterine devices. |
| Meril Life Sciences Private Limited | Not Listed | No | No | Not verified | No specific regulatory certifications found. |
| EON Meditech Private Limited | Not Listed | No | No | Not verified | No specific regulatory certifications found. |
| Shiva Products | Not Listed | No | No | Not verified | No specific regulatory certifications found. |
| Khosla Surgical Industries | Not Listed | No | No | Not verified | No specific regulatory certifications found. |
| JK Medirise | Not Listed | No | No | Not verified | No specific regulatory certifications found. |
| Orikam Healthcare India Private Limited | Not Listed | No | No | Not verified | No specific regulatory certifications found. |
| Hilltron Medical Technology | Not Listed | No | No | Not verified | No specific regulatory certifications found. |
| ABM4 Trades | Not Listed | No | No | Not verified | No specific regulatory certifications found. |
| Beacon Diagnostics Pvt Ltd | Not Listed | No | No | Not verified | No specific regulatory certifications found. |
| Aadpri Ophthalmic Company | Not Listed | No | No | Not verified | No specific regulatory certifications found. |
| Vira Corporation | Not Listed | No | No | Not verified | No specific regulatory certifications found. |
| Manarti Exports | Not Listed | No | No | Not verified | No specific regulatory certifications found. |
| Precision Biomed Private Limited | Not Listed | No | No | Not verified | No specific regulatory certifications found. |
TransData Nexus reviewed the regulatory standing of 14 leading Instrument exporters from India. 0 hold US FDA facility approvals, 1 maintain WHO-GMP certification, and 0 are EU GMP compliant. For regulated markets (US, EU, Australia), prioritise suppliers with active FDA or EU GMP approvals. For semi-regulated markets (Africa, ASEAN, Latin America), WHO-GMP certification is the minimum recommended standard.
Certification status compiled from publicly available regulatory databases including FDA Orange Book, WHO Prequalification database, and EMA GMP registry. Buyers should independently verify compliance status with the relevant regulatory authority before placing orders.
TransData Nexus Research · Mar 2026
Manufacturing Hub Analysis — Vendor Proximity
India's pharmaceutical clusters relevant to Instrument sourcing
1Hyderabad — Bulk Drug Capital
Hyderabad, often referred to as the "Bulk Drug Capital of India," is renowned for its extensive production of active pharmaceutical ingredients (APIs). The city's pharmaceutical sector is bolstered by a robust infrastructure, including research institutions and manufacturing facilities. This strong foundation supports the production of medical instruments, contributing significantly to India's exports in this category.
2Ahmedabad-Vadodara — Formulations Hub
The Ahmedabad-Vadodara belt in Gujarat is a prominent hub for pharmaceutical formulations. This region hosts numerous companies specializing in the development and manufacturing of finished dosage forms. The presence of such facilities indicates a well-established ecosystem capable of producing high-quality medical instruments, aligning with the export data for HS code 30061010.
3Mumbai-Thane-Raigad — Export Gateway
The Mumbai-Thane-Raigad region serves as a critical export gateway for pharmaceutical products. Its strategic coastal location facilitates efficient international shipping, making it a preferred zone for companies aiming to export medical instruments. The region's advanced logistics and proximity to major ports enhance its role in the global supply chain for medical instruments.
4Baddi-Nalagarh — Tax Incentive Zone
Baddi-Nalagarh in Himachal Pradesh has emerged as a significant pharmaceutical manufacturing zone, largely due to favorable tax incentives. This area has attracted numerous pharmaceutical companies, contributing to the production and export of medical instruments. The region's growth underscores its importance in India's pharmaceutical landscape.
5Sourcing Recommendations
- Diversify Supplier Base: Engage with multiple suppliers across different clusters to mitigate risks associated with regional disruptions.
- Leverage Export Hubs: Utilize the Mumbai-Thane-Raigad region's export infrastructure to streamline international shipments of medical instruments.
- Monitor Regulatory Compliance: Ensure that suppliers adhere to the standards set by the Central Drugs Standard Control Organisation (CDSCO) to maintain product quality and compliance.
- Assess Incentive Benefits: Evaluate the financial advantages offered by manufacturing in tax incentive zones like Baddi-Nalagarh to optimize cost structures.
By strategically sourcing from these established pharmaceutical clusters, companies can enhance their supply chain resilience and capitalize on India's robust manufacturing capabilities in the medical instruments sector.
Recent M&A, Collaborations & Capacity Expansions
Industry developments among top Instrument exporters from India
MERIL ENDO SURGERY PRIVATE LIMITED — Meril Life Sciences receives $210 million from Warburg Pincus
In February 2022, Meril Life Sciences, the parent company of MERIL ENDO SURGERY PRIVATE LIMITED, secured a $210 million investment from private equity firm Warburg Pincus through its Dutch affiliate, South Elm Investments BV. IMPACT: This substantial investment is expected to enhance Meril's production capabilities and expand its global market reach, potentially increasing instrument exports.
Impact: This substantial investment is expected to enhance Meril's production capabilities and expand its global market reach, potentially increasing instrument exports.
MERIL ENDO SURGERY PRIVATE LIMITED — Meril grants Japan Lifeline exclusive rights for Myval Octacor in Japan
In November 2023, Meril Life Sciences entered into an agreement with Japan Lifeline, granting the latter exclusive rights to promote Meril's transcatheter heart valve, Myval Octacor, upon approval by Japan's Pharmaceuticals and Medical Devices Agency. IMPACT: This collaboration is likely to boost Meril's presence in the Japanese market, potentially leading to increased instrument exports.
Impact: This collaboration is likely to boost Meril's presence in the Japanese market, potentially leading to increased instrument exports.
MERIL ENDO SURGERY PRIVATE LIMITED — Meril introduces MISSO, a robotic-assisted surgical system
In 2024, Meril Life Sciences launched MISSO, an indigenously developed robotic-assisted surgical system designed to enhance precision and consistency in orthopedic joint replacement procedures. IMPACT: The introduction of MISSO may strengthen Meril's product portfolio, potentially increasing demand and exports of related instruments.
Impact: The introduction of MISSO may strengthen Meril's product portfolio, potentially increasing demand and exports of related instruments.
Common Questions — Instrument Suppliers from India
Answers based on Indian Customs (DGFT) shipment records compiled by TransData Nexus
Q Which instrument supplier from India is the most reliable for bulk orders?
Based on shipment frequency and export consistency, MERIL ENDO SURGERY PRIVATE LIMITED leads with 1,701 recorded shipments worth $218.4K. EON MEDITECH PRIVATE LIMITED (24 shipments) and SHIVA PRODUCTS (67 shipments) are also established high-volume exporters.
Q How many instrument manufacturers are there in India?
India has 77 active instrument exporters with a combined export market of $1.4M across 2,733 shipments to 85 countries. The top 5 suppliers hold 55.1% of total export value.
Q What certifications should I verify?
Verify: WHO-GMP certification (most markets), US FDA approval (United States), EU GMP certificate (EU/EEA), and Free Sale Certificate from CDSCO. Always request a Certificate of Analysis (CoA) and Certificate of Origin (CoO).
Q What is the typical price range for instrument from India?
Average FOB unit price: $22.57 per unit, ranging from $0.00 to $800.33. Average shipment value: $499.
Official References & Regulatory Resources
- CDSCO India
- Pharmexcil
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- India Trade Statistics (DGFT)
Verify manufacturer licensing and export certifications with the official agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Supplier Ranking: 77 verified Indian exporters of Instrument ranked by capped export value from DGFT shipping bill records.
- 2.Export Value Analysis: Total export value aggregated from 2,733 individual shipping bill records. Values are FOB in USD.
- 3.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 4.Market Concentration: Supplier market share and geographic reach analyzed across 85 destination countries.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2,733 Verified Shipments
77 exporters tracked
Expert-Reviewed
By pharmaceutical trade specialists