India to United Kingdom Pharmaceutical Export
Bilateral Trade Intelligence · $1.2B Total Trade · 610 Exporters · 1615 Buyers · DGFT Verified · Updated March 2026
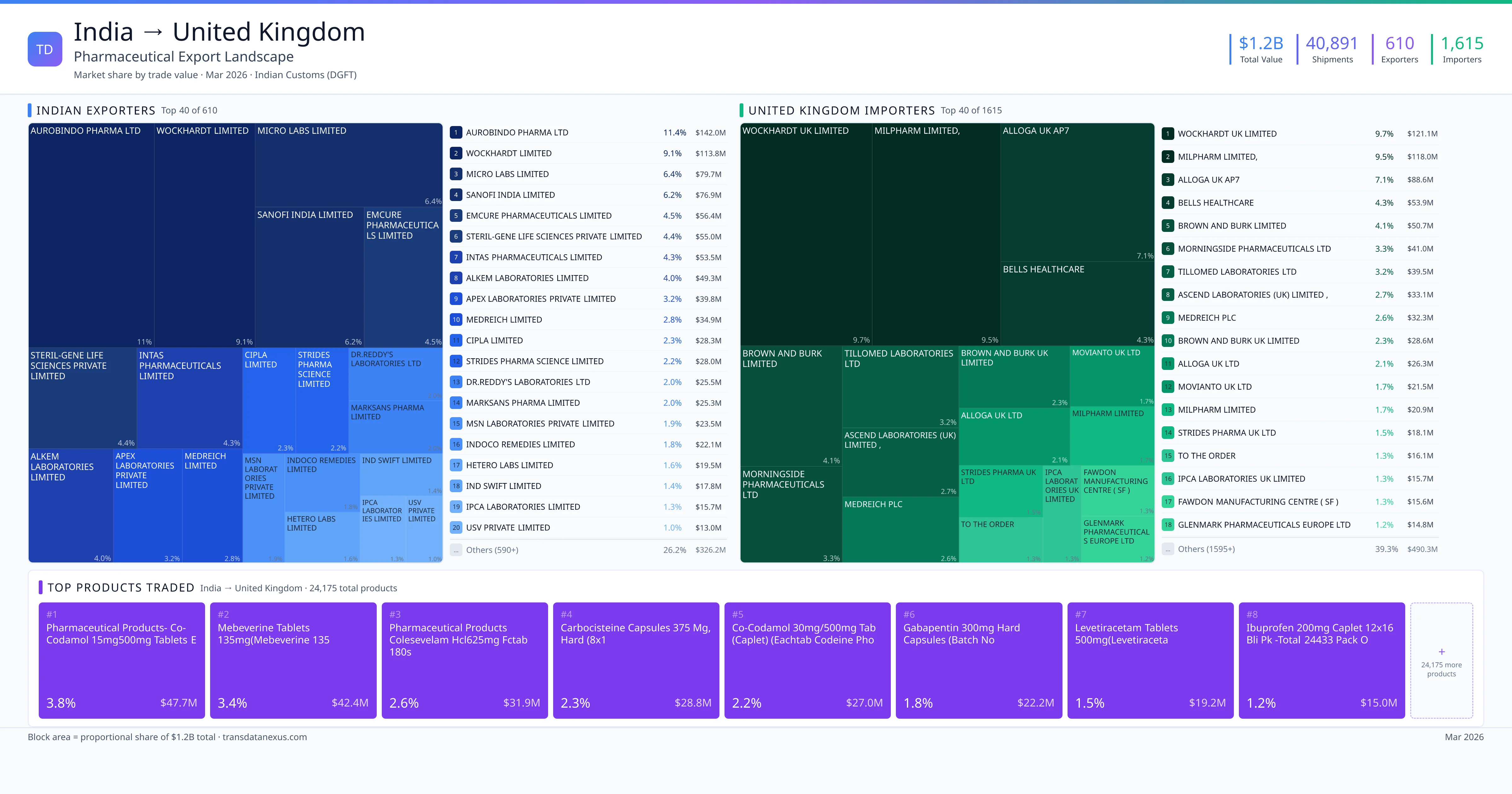
India exported $1.2B worth of pharmaceutical formulations to United Kingdom across 40,891 verified shipments, sourced from 610 Indian exporters supplying 1615 United Kingdom buyers. The top exporters are AUROBINDO PHARMA LTD ($142.0M) and WOCKHARDT LIMITED ($113.8M). The leading products are Gabapentin ($22.2M) and Ibuprofen ($15.0M). Average shipment value: $30.5K.
Top Pharmaceutical Formulations — India to United Kingdom
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Mebeverine Tablets 135mg(mebeverine 135 | $42.4M | 3.4% |
| 2 | Pharmaceutical Products Colesevelam Hcl625mg Fctab 180s Hdpe UK (inv.qty.5265x1… | $31.9M | 2.6% |
| 3 | Pharmaceutical Products- Co-codamol 15mg500mg Tablets Exempted Material(w/o Was… | $29.3M | 2.4% |
| 4 | Carbocisteine Capsules 375 Mg, Hard (8x1 | $28.8M | 2.3% |
| 5 | Co-codamol 30mg/500mg Tablets (caplet) (eachtab Codeine Phosphate Hemihydrate P… | $27.0M | 2.2% |
| 6 | Gabapentin 300mg Hard Capsules (batch No | $22.2M | 1.8% |
| 7 | Levetiracetam Tablets 500mg(levetiraceta | $19.2M | 1.5% |
| 8 | Pharmaceutical Products- Co-codamol 15mg500mg Tablets Exempted Material (w/o Wa… | $18.4M | 1.5% |
| 9 | Ibuprofen 200mg Caplet 12x16 Bli Pk -total 24433 Pack of 192 Tablets Total 4691… | $15.0M | 1.2% |
| 10 | Paracetamol 500 MG Tablets (2x4`s) | $13.6M | 1.1% |
| 11 | Paracetamol 500 MG Tablets (2x8`s) | $12.6M | 1.0% |
| 12 | Clarithromycin Film - Coated Tablets 250 | $11.3M | 0.9% |
| 13 | Fluoxetine 20mg Capsules (3x10s) - Tax I | $10.4M | 0.8% |
| 14 | Ibuprofen 200mg Caplet Tablets 12x16 Bli Pk--total 16419 Pack of 192 Tablets To… | $10.1M | 0.8% |
| 15 | Pregabalin Capsules 75 Mg(pregabalin Mil | $9.9M | 0.8% |
India exports 17+ pharmaceutical formulations to United Kingdom with a combined trade value of $1.2B. Key products include Gabapentin ($22.2M), Ibuprofen ($15.0M), Paracetamol ($13.6M), Clarithromycin ($11.3M), Fluoxetine ($10.4M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 40,891 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to United Kingdom Trade Routes
These are the top pharmaceutical products exported from India to United Kingdom, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, United Kingdom buyers, regulatory requirements, and logistics for that specific product corridor. Products include Gabapentin ($22.2M), Ibuprofen ($15.0M), Paracetamol ($13.6M), Clarithromycin ($11.3M), Fluoxetine ($10.4M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to United Kingdom
610 Indian pharmaceutical companies export finished formulations to United Kingdom. Leading exporters include Aurobindo Pharma Ltd, Wockhardt Limited, Micro Labs Limited, Sanofi India Limited. The top exporter accounts for 11.4% of total India–United Kingdom pharma exports. Source: Indian Customs (DGFT).
Top United Kingdom Buyers from India
1615 companies in United Kingdom import pharmaceutical formulations from India. Top buyers include Wockhardt Uk Limited, Milpharm Limited,, Alloga Uk Ap7, Bells Healthcare. The largest buyer accounts for 9.7% of India–United Kingdom pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to United Kingdom Pharmaceutical Shipments
Indian Export Ports
United Kingdom Import Ports
Trade Statistics
Other Destinations
Product Routes
India–United Kingdom Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and the United Kingdom has experienced significant growth over the past decades. In 2024, India exported pharmaceutical products valued at $760.17 million to the UK, marking a substantial increase from previous years. This upward trajectory reflects India's expanding role as a global supplier of finished pharmaceutical formulations, catering to the UK's diverse healthcare needs.
Key milestones in this bilateral trade include the establishment of robust supply chains and the adoption of international quality standards by Indian manufacturers. The UK's reliance on imported pharmaceuticals, particularly generics, has further bolstered this trade relationship. The Medicines and Healthcare products Regulatory Agency (MHRA) has played a pivotal role in facilitating the entry of Indian pharmaceutical products into the UK market by ensuring compliance with stringent regulatory requirements.
2India's Market Position
India holds a significant position in the UK's pharmaceutical import landscape. In 2024, pharmaceutical products accounted for $760.17 million of India's total exports to the UK, underscoring the sector's importance. This positions India as a key supplier, especially in the generic medicines segment, which aligns with the UK's strategic focus on cost-effective healthcare solutions.
3Recent Developments
Between 2024 and 2026, several developments have influenced the India-UK pharmaceutical trade. The MHRA has updated its registration processes to streamline approvals for foreign manufacturers, including those from India. These changes aim to expedite market access for compliant products, reflecting a commitment to maintaining high standards while facilitating trade. (gov.uk)
Additionally, the UK has engaged in discussions with India to enhance bilateral trade agreements, focusing on reducing tariffs and non-tariff barriers. These negotiations aim to create a more favorable environment for pharmaceutical exports, benefiting both nations by ensuring a steady supply of quality medicines.
EXPORT_REGULATORY
United Kingdom Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to export to the UK must navigate the MHRA's registration process. This involves submitting a comprehensive dossier, typically in the Common Technical Document (CTD) or electronic Common Technical Document (eCTD) format, which includes detailed information on the product's quality, safety, and efficacy. The MHRA assesses these submissions to ensure compliance with UK standards.
The registration timeline can vary. Standard applications may take up to 60 working days for processing, excluding any time required for additional information or data. If an inspection of the manufacturing facility is necessary, the process may extend to 90 days.
2GMP & Facility Requirements
The UK mandates that all pharmaceutical products imported into its market adhere to Good Manufacturing Practice (GMP) standards. Indian manufacturers must ensure their facilities comply with these standards, which are evaluated during the MHRA's inspection process. The MHRA conducts inspections to verify compliance with GMP, and mutual recognition agreements may facilitate the acceptance of inspections conducted by recognized authorities in India.
3Import Documentation
To import pharmaceutical products into the UK, Indian exporters must provide several key documents:
- Certificate of Pharmaceutical Product (CPP): Confirms that the product is authorized for sale in the exporting country.
- Certificate of Analysis (CoA): Details the product's composition and quality control measures.
- Good Manufacturing Practice (GMP) Certificate: Demonstrates that the manufacturing facility adheres to GMP standards.
These documents are essential for customs clearance and regulatory compliance. Additionally, adherence to the UK's customs procedures, including accurate labeling and packaging, is crucial for the smooth importation of pharmaceutical products.
EXPORT_TRENDS
Product Categories & Therapeutic Trends — India to United Kingdom
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to the UK are predominantly in the generic medicines category. This aligns with the UK's healthcare strategy, which emphasizes cost-effective treatments to manage public health expenditures. The demand for generics in the UK is substantial, driven by the need for affordable alternatives to branded medications.
2Emerging Opportunities
The UK's focus on biosimilars presents a growing opportunity for Indian exporters. As patents for biologic drugs expire, the demand for biosimilars is expected to rise, offering Indian manufacturers a chance to expand their market presence. Additionally, the UK's interest in personalized medicine and advanced therapies opens new avenues for Indian pharmaceutical companies to introduce innovative products.
3Demand Drivers
The UK's aging population and the increasing prevalence of chronic diseases are significant drivers of pharmaceutical demand. These demographic trends necessitate a steady supply of effective and affordable medications, creating a favorable environment for Indian pharmaceutical exports. Moreover, the UK's commitment to maintaining a robust healthcare system ensures sustained demand for imported pharmaceutical products.
EXPORT_POLICY
Trade Policy & Tariff Intelligence — India and United Kingdom
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
The UK applies Most Favored Nation (MFN) tariff rates to pharmaceutical imports, which are generally low to encourage the availability of essential medicines. While preferential rates and duty exemptions may exist under specific trade agreements, the UK's tariff structure is designed to facilitate the importation of pharmaceutical products, ensuring that cost-effective treatments are accessible to the population.
2Trade Agreements
The UK has engaged in trade negotiations with India to enhance bilateral relations, focusing on reducing tariffs and non-tariff barriers. These discussions aim to create a more favorable environment for pharmaceutical exports, benefiting both nations by ensuring a steady supply of quality medicines. The outcomes of these negotiations are expected to further strengthen the trade corridor between India and the UK.
3IP & Patent Landscape
The UK's intellectual property (IP) framework, including patent provisions and data exclusivity periods, influences the entry of Indian generic medicines into the market. While the UK upholds strong IP protections, it also allows for the entry of generics once patents expire, balancing innovation incentives with public health needs. Indian exporters must navigate this landscape to effectively introduce their products into the UK market.
EXPORT_LOGISTICS
Supply Chain & Logistics — India to United Kingdom Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to the UK primarily utilize sea and air routes. Sea freight is cost-effective for large volumes, with transit times typically ranging from 20 to 30 days, depending on the port of departure and arrival. Air freight offers faster delivery, usually within 7 to 10 days, and is preferred for high-value or time-sensitive products. Disruptions in major shipping routes, such as those through the Red Sea, can impact delivery schedules and require alternative routing.
2Port Infrastructure
In India, major export ports include Nhava Sheva Sea Port and Vizag Sea Port, which handle a significant portion of pharmaceutical exports. In the UK, key import ports are London Gateway Port, London Heathrow Airport, and Felixstowe Port. These ports are equipped to handle pharmaceutical shipments, ensuring compliance with international standards and facilitating efficient customs clearance processes.
3Cold Chain & Compliance
Maintaining the integrity of pharmaceutical products during transit is critical. Temperature-controlled logistics are essential to preserve product efficacy, especially for biologics and temperature-sensitive medications. Compliance with Good Distribution Practice (GDP) guidelines ensures that products are stored and transported under appropriate conditions. Packaging standards must also meet regulatory requirements to prevent contamination and ensure product safety upon arrival in the UK.
EXPORT_OPPORTUNITY
Market Opportunity Assessment — United Kingdom for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
The UK's pharmaceutical market is substantial, with a significant portion of its demand met through imports. The market's growth is driven by factors such as an aging population, increasing prevalence of chronic diseases, and a focus on innovative therapies. Generic medicines constitute a significant share of the market, offering cost-effective treatment options and presenting opportunities for Indian exporters.
2Healthcare System
The UK's National Health Service (NHS) is the primary healthcare provider, with a focus on providing comprehensive services to the population. Government healthcare programs and insurance coverage ensure that a wide range of medications, including imported pharmaceuticals, are accessible to patients. The NHS's procurement systems are designed to source medicines that meet quality standards at competitive prices, creating a favorable environment for Indian pharmaceutical exports.
3Opportunity for Indian Exporters
Indian pharmaceutical companies have specific opportunities in the UK's market, particularly in the generic medicines segment. The demand for affordable and effective treatments aligns with India's manufacturing capabilities and cost advantages. Additionally, the UK's interest in biosimilars and personalized medicine presents avenues for Indian exporters to introduce innovative products, leveraging their expertise in these areas.
EXPORT_COMPETITIVE
Competitive Landscape — India vs Other Pharmaceutical Suppliers to United Kingdom
Competing origins, India's edge, challenges and threats
1Competing Origins
The UK imports pharmaceutical products from various countries, including China, the European Union, and domestic manufacturers. While China and the EU are significant suppliers, India's share in the UK's pharmaceutical imports is notable, reflecting its competitive edge in the generics market. Domestic production in the UK also contributes to the supply but is complemented by imports to meet the diverse needs
FAQ — India to United Kingdom Pharmaceutical Trade
What is the total value of India's pharmaceutical export to United Kingdom?
India exported pharmaceuticals worth $1.2B to United Kingdom across 40,891 verified shipments.
Who are the top Indian pharmaceutical exporters to United Kingdom?
1. AUROBINDO PHARMA LTD — $142.0M. 2. WOCKHARDT LIMITED — $113.8M. 3. MICRO LABS LIMITED — $79.7M. Total: 610 suppliers.
Which companies in United Kingdom import pharmaceuticals from India?
1. WOCKHARDT UK LIMITED — $121.1M. 2. MILPHARM LIMITED, — $118.0M. 3. ALLOGA UK AP7 — $88.6M. 1615 buyers total.
What pharmaceutical products does India export most to United Kingdom?
1. Mebeverine Tablets 135mg(mebeverine 135 ($42.4M, 3.4%); 2. Pharmaceutical Products Colesevelam Hcl625mg Fctab 180s Hdpe UK (inv.qty.5265x1… ($31.9M, 2.6%); 3. Pharmaceutical Products- Co-codamol 15mg500mg Tablets Exempted Material(w/o Was… ($29.3M, 2.4%); 4. Carbocisteine Capsules 375 Mg, Hard (8x1 ($28.8M, 2.3%); 5. Co-codamol 30mg/500mg Tablets (caplet) (eachtab Codeine Phosphate Hemihydrate P… ($27.0M, 2.2%)
Which ports handle pharmaceutical shipments from India to United Kingdom?
Export: NHAVA SHEVA SEA, NHAVA SHEVA SEA (INNSA1), VIZAG SEA, CHENNAI AIR, KAMARAJAR PORT. Import: LONDON GATWAY PORT, LONDON - HEATHROW, Felixstowe, Southampton, FELIXSTOWE.
Why does United Kingdom import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $1.2B corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply United Kingdom?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to United Kingdom pharmaceutical trade?
$30.5K per consignment across 40,891 shipments.
How many Indian pharmaceutical companies export to United Kingdom?
610 Indian companies. Largest: AUROBINDO PHARMA LTD with $142.0M.
How can I find verified Indian pharmaceutical suppliers for United Kingdom?
TransData Nexus covers 610 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to United Kingdom Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 40,891 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to United Kingdom identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 40,891 verified shipments from 610 Indian exporters to 1615 United Kingdom buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
610 Exporters
1615 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists