India to Uganda Pharmaceutical Export
Bilateral Trade Intelligence · $200.0M Total Trade · 298 Exporters · 469 Buyers · DGFT Verified · Updated March 2026
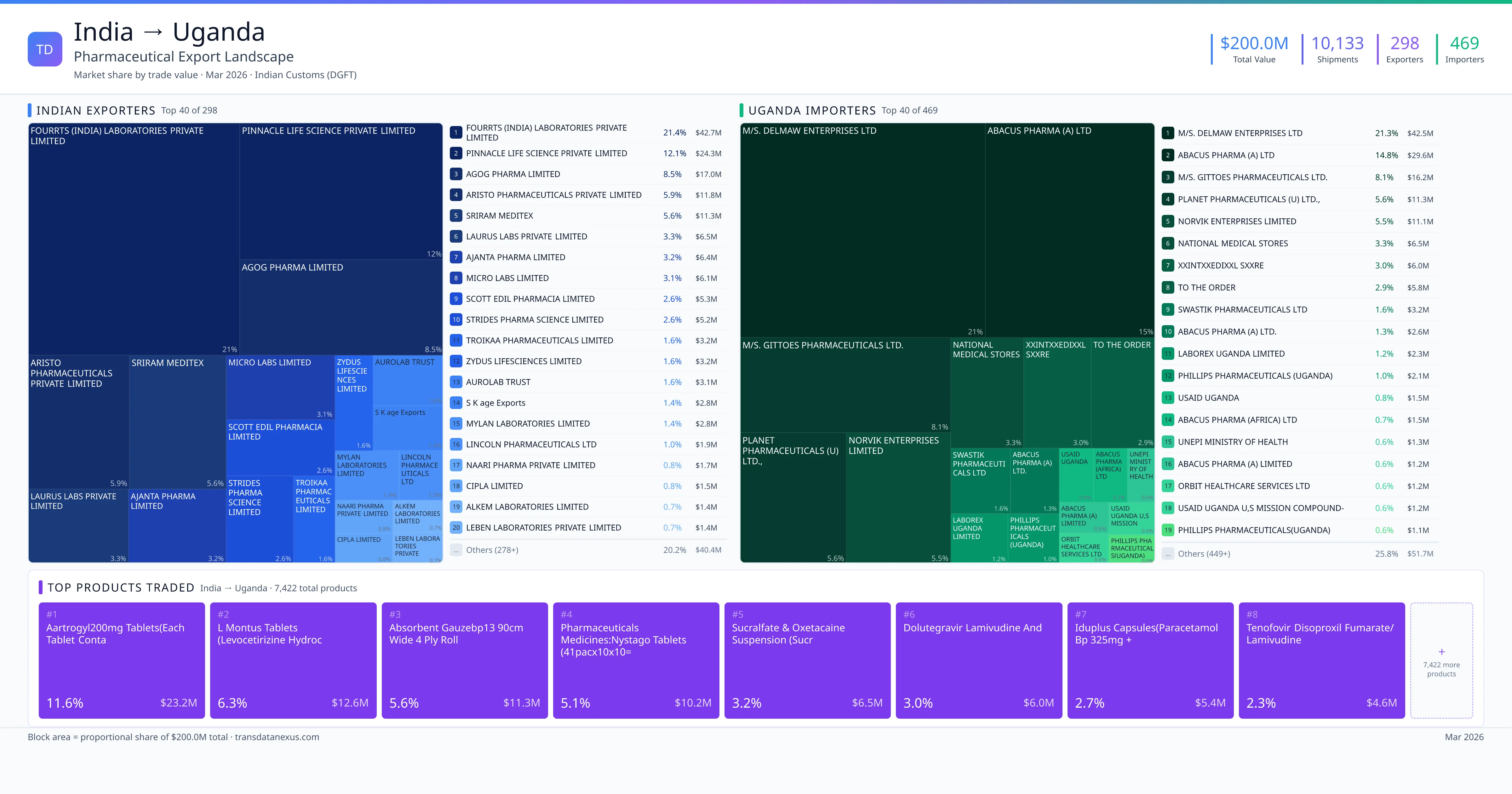
India exported $200.0M worth of pharmaceutical formulations to Uganda across 10,133 verified shipments, sourced from 298 Indian exporters supplying 469 Uganda buyers. The top exporters are FOURRTS (INDIA) LABORATORIES PRIVATE LIMITED ($42.7M) and PINNACLE LIFE SCIENCE PRIVATE LIMITED ($24.3M). The leading products are Dolutegravir ($6.0M) and Paracetamol ($5.4M). Average shipment value: $19.7K.
Top Pharmaceutical Formulations — India to Uganda
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Aartrogyl200mg Tablets(each Tablet Conta | $23.2M | 11.6% |
| 2 | L Montus Tablets (levocetirizine Hydroc | $12.6M | 6.3% |
| 3 | Absorbent Gauzebp13 90cm Wide 4 Ply Roll | $11.3M | 5.6% |
| 4 | Pharmaceuticals Medicines:nystago Tablets (41pacx10x10=4100 Tablets)(net Wt:0.3… | $10.2M | 5.1% |
| 5 | Sucralfate & Oxetacaine Suspension (sucr | $6.5M | 3.2% |
| 6 | Dolutegravir Lamivudine and | $6.0M | 3.0% |
| 7 | Iduplus Capsules(paracetamol BP 325mg + | $5.4M | 2.7% |
| 8 | Tenofovir Disoproxil Fumarate/lamivudine | $4.6M | 2.3% |
| 9 | Metformin Tablets BP 500mg (metfil) (198 | $4.2M | 2.1% |
| 10 | Aristo Telvas 80 (1640 X 10 X 10's= 164000 Tablets) | $3.4M | 1.7% |
| 11 | Paracetamol BP 500mg +chlorphenamine Mal | $3.0M | 1.5% |
| 12 | Aristo Telvas H 80/12.5 (1680 X 10 X 10's= 168000 Tablets) | $2.5M | 1.3% |
| 13 | Potassium Citrate BP 1100mg & Magnesium | $2.1M | 1.1% |
| 14 | Haematinic Capsules with Choline, Lysine | $1.9M | 1.0% |
| 15 | Quadrajel (6362 X 15g) | $1.9M | 0.9% |
India exports 20+ pharmaceutical formulations to Uganda with a combined trade value of $200.0M. Key products include Dolutegravir ($6.0M), Paracetamol ($5.4M), Tenofovir ($4.6M), Metformin ($4.2M), Telmisartan ($3.4M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 10,133 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Uganda Trade Routes
These are the top pharmaceutical products exported from India to Uganda, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Uganda buyers, regulatory requirements, and logistics for that specific product corridor. Products include Dolutegravir ($6.0M), Paracetamol ($5.4M), Tenofovir ($4.6M), Metformin ($4.2M), Telmisartan ($3.4M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Uganda
298 Indian pharmaceutical companies export finished formulations to Uganda. Leading exporters include Fourrts (india) Laboratories Private Limited, Pinnacle Life Science Private Limited, Agog Pharma Limited, Aristo Pharmaceuticals Private Limited. The top exporter accounts for 21.4% of total India–Uganda pharma exports. Source: Indian Customs (DGFT).
Top Uganda Buyers from India
469 companies in Uganda import pharmaceutical formulations from India. Top buyers include M/s. Delmaw Enterprises Ltd, Abacus Pharma (a) Ltd, M/s. Gittoes Pharmaceuticals Ltd., Planet Pharmaceuticals (u) Ltd.,. The largest buyer accounts for 21.3% of India–Uganda pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Uganda Pharmaceutical Shipments
Indian Export Ports
Uganda Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Uganda Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and Uganda has experienced significant growth over the past decade. In 2024, India exported pharmaceutical products worth approximately $102.75 million to Uganda, marking a substantial increase from previous years. This upward trajectory underscores the strengthening economic ties and the growing reliance of Uganda on India's pharmaceutical industry. The expansion is attributed to India's competitive pricing, diverse product offerings, and adherence to international quality standards. Uganda's increasing demand for affordable and quality medicines has further bolstered this trade relationship.
2India's Market Position
India holds a dominant position in Uganda's pharmaceutical import market. In 2024, pharmaceutical imports from India amounted to $180.82 million, making it the largest supplier of pharmaceutical products to Uganda. This dominance is indicative of India's strategic importance in meeting Uganda's healthcare needs. The extensive range of products, coupled with India's compliance with Good Manufacturing Practices (GMP), has solidified its standing as a preferred supplier.
3Recent Developments
Between 2024 and 2026, several developments have influenced the India-Uganda pharmaceutical trade. In September 2025, Uganda's Minister of Health presented the National Drug and Health Products Authority Bill to the Parliamentary Committee on Health. This bill aims to establish a more robust regulatory framework for drugs and health products, potentially impacting the registration and importation processes for pharmaceutical products. The proposed reforms emphasize stringent compliance with international standards, which could affect Indian exporters' operations in Uganda. (parliament.go.ug)
EXPORT_REGULATORY
Uganda Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to register products in Uganda must comply with the National Drug Authority's (NDA) guidelines, which align with World Health Organization (WHO) standards. The registration process involves submitting a comprehensive dossier, typically in the Common Technical Document (CTD) format, detailing the product's quality, safety, and efficacy. Bioequivalence studies are mandatory for generic products to demonstrate therapeutic equivalence to the innovator drug. The review and approval timeline can vary, but applicants should anticipate a thorough evaluation process to ensure compliance with NDA's stringent requirements. (nda.or.ug)
2GMP & Facility Requirements
Uganda mandates that all pharmaceutical products imported into the country be manufactured in facilities compliant with current Good Manufacturing Practice (cGMP) standards. The NDA conducts inspections of foreign manufacturing sites to assess compliance with these standards. Manufacturers must submit a Site Master File detailing their manufacturing processes and quality control measures. Upon approval, the NDA may conduct periodic inspections to ensure ongoing compliance. Non-compliance can result in sanctions, including suspension or revocation of marketing authorizations. (nda.or.ug)
3Import Documentation
To import pharmaceutical products into Uganda, Indian exporters must obtain an import permit from the NDA. The application process involves submitting necessary documentation, including Certificates of Pharmaceutical Product (CPP), Certificates of Analysis (CoA), and proof of GMP compliance. Upon arrival, shipments undergo inspection at designated ports of entry, such as Kampala, Mombasa, and Entebbe. The NDA, in collaboration with the Uganda Revenue Authority, oversees customs procedures to ensure that imported medicines meet the required standards before they are released into the market. (nda.or.ug)
EXPORT_TRENDS
Product Categories & Therapeutic Trends — India to Uganda
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Uganda are predominantly composed of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. These products address a wide range of therapeutic areas, with a notable emphasis on treatments for infectious diseases, gastrointestinal disorders, and chronic conditions such as diabetes and hypertension. The prevalence of these health issues in Uganda drives the demand for such medications, aligning with the country's healthcare priorities.
2Emerging Opportunities
The global pharmaceutical industry is witnessing patent expirations, leading to increased opportunities for generic drugs. Uganda's growing healthcare needs present a favorable market for Indian exporters to introduce biosimilars and generic versions of off-patent drugs. Additionally, emerging therapeutic areas such as oncology and immunology offer potential avenues for expansion, provided that Indian manufacturers can meet the regulatory and quality standards set by the NDA.
3Demand Drivers
Uganda's disease burden, characterized by high rates of infectious diseases, non-communicable diseases, and an aging population, significantly drives the demand for pharmaceutical imports. The government's increased healthcare spending and initiatives to improve access to essential medicines further contribute to this demand. Indian pharmaceutical products, known for their affordability and quality, are well-positioned to meet these needs, reinforcing India's role as a key supplier in Uganda's pharmaceutical market.
EXPORT_POLICY
Trade Policy & Tariff Intelligence — India and Uganda
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Uganda's tariff structure for pharmaceutical imports includes Most Favored Nation (MFN) rates, with potential preferential rates under specific trade agreements. While there are no blanket duty exemptions for pharmaceutical formulations, certain products may benefit from reduced tariffs based on their classification and the prevailing trade policies. Importers should consult the Uganda Revenue Authority for the most current tariff schedules and any applicable exemptions.
2Trade Agreements
Uganda is a member of the East African Community (EAC), which has established a Common Market Protocol facilitating the free movement of goods, including pharmaceuticals, within member states. However, there are no specific Free Trade Agreements (FTAs) between India and Uganda. The proposed National Drug and Health Products Authority Bill, presented in September 2025, aims to strengthen Uganda's regulatory framework, potentially impacting trade dynamics and requiring Indian exporters to adapt to new compliance standards. (parliament.go.ug)
3IP & Patent Landscape
Uganda's intellectual property laws, including provisions for patents and data exclusivity, influence the entry of generic drugs into the market. The proposed National Drug and Health Products Authority Bill seeks to enhance the regulatory environment, which may include stricter enforcement of IP rights. Indian exporters should stay informed about these developments to navigate the evolving landscape effectively. (parliament.go.ug)
EXPORT_LOGISTICS
Supply Chain & Logistics — India to Uganda Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to Uganda primarily utilize sea and air routes. Major Indian ports involved include Chennai Air and Nhava Sheva Sea (INNSA1), with a significant portion of exports departing from these locations. Upon arrival, goods are typically cleared through ports in Kampala, Mombasa, and Entebbe. Transit times vary based on the mode of transport and specific routes, with sea shipments generally taking longer than air freight. Exporters should monitor potential disruptions, such as those in the Red Sea region, which can affect shipping schedules.
2Port Infrastructure
In India, key export ports include Chennai Air (2.4% of shipments), GRFL Sahnewal Ludhiana ICD (0.2%), and Nhava Sheva Sea (INNSA1) (13.3%). In Uganda, primary import ports are Kampala (14.8%), Mombasa (11.6%), and Entebbe (45.9%). These ports are equipped to handle pharmaceutical imports, with facilities for inspection and clearance by the National Drug Authority and the Uganda Revenue Authority.
3Cold Chain & Compliance
Maintaining the integrity of pharmaceutical products during transit is crucial. Temperature-controlled logistics are essential for products requiring specific storage conditions. Compliance with Good Distribution Practice (GDP) standards ensures that products are stored, handled, and transported under conditions that preserve their quality. Indian exporters must adhere to these standards and ensure that packaging meets the requirements to prevent damage and contamination during transit.
EXPORT_OPPORTUNITY
Market Opportunity Assessment — Uganda for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Uganda's pharmaceutical market is experiencing growth, driven by an increasing population and rising healthcare needs. In 2024, Uganda imported pharmaceutical products worth $180.82 million from India, indicating a substantial market size. The demand for generic medicines, in particular, is on the rise, presenting opportunities for Indian exporters to expand their market share.
2Healthcare System
The Ugandan government has been investing in healthcare infrastructure and services, aiming to improve access to essential medicines and healthcare services. Initiatives include expanding health facilities, training healthcare professionals, and implementing programs to combat prevalent diseases. These efforts create a conducive environment for pharmaceutical imports, especially from countries like India that offer cost-effective and quality products.
3Opportunity for Indian Exporters
Indian pharmaceutical exporters have significant opportunities in Uganda, particularly in the generic medicines segment. Products such as antimalarials, antibiotics, and treatments for non-communicable diseases align with Uganda's healthcare priorities. The proposed regulatory reforms in Uganda may present challenges but also opportunities for exporters who can navigate the evolving compliance landscape effectively. (parliament.go.ug)
EXPORT_COMPETITIVE
Competitive Landscape — India vs Other Pharmaceutical Suppliers to Uganda
Competing origins, India's edge, challenges and threats
1Competing Origins
Uganda's pharmaceutical imports are diversified, with suppliers from various countries, including China
FAQ — India to Uganda Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Uganda?
India exported pharmaceuticals worth $200.0M to Uganda across 10,133 verified shipments.
Who are the top Indian pharmaceutical exporters to Uganda?
1. FOURRTS (INDIA) LABORATORIES PRIVATE LIMITED — $42.7M. 2. PINNACLE LIFE SCIENCE PRIVATE LIMITED — $24.3M. 3. AGOG PHARMA LIMITED — $17.0M. Total: 298 suppliers.
Which companies in Uganda import pharmaceuticals from India?
1. M/S. DELMAW ENTERPRISES LTD — $42.5M. 2. ABACUS PHARMA (A) LTD — $29.6M. 3. M/S. GITTOES PHARMACEUTICALS LTD. — $16.2M. 469 buyers total.
What pharmaceutical products does India export most to Uganda?
1. Aartrogyl200mg Tablets(each Tablet Conta ($23.2M, 11.6%); 2. L Montus Tablets (levocetirizine Hydroc ($12.6M, 6.3%); 3. Absorbent Gauzebp13 90cm Wide 4 Ply Roll ($11.3M, 5.6%); 4. Pharmaceuticals Medicines:nystago Tablets (41pacx10x10=4100 Tablets)(net Wt:0.3… ($10.2M, 5.1%); 5. Sucralfate & Oxetacaine Suspension (sucr ($6.5M, 3.2%)
Which ports handle pharmaceutical shipments from India to Uganda?
Export: CHENNAI AIR, GRFL SAHNEWAL LUDHIANA ICD, NHAVA SHEVA SEA (INNSA1), KAMARDWISA, SAHAR AIR. Import: Kampala, Mombasa, ENTEBBE, KAMPALA, MOMBASA.
Why does Uganda import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $200.0M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Uganda?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Uganda pharmaceutical trade?
$19.7K per consignment across 10,133 shipments.
How many Indian pharmaceutical companies export to Uganda?
298 Indian companies. Largest: FOURRTS (INDIA) LABORATORIES PRIVATE LIMITED with $42.7M.
How can I find verified Indian pharmaceutical suppliers for Uganda?
TransData Nexus covers 298 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Uganda Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 10,133 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Uganda identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 10,133 verified shipments from 298 Indian exporters to 469 Uganda buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
298 Exporters
469 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists