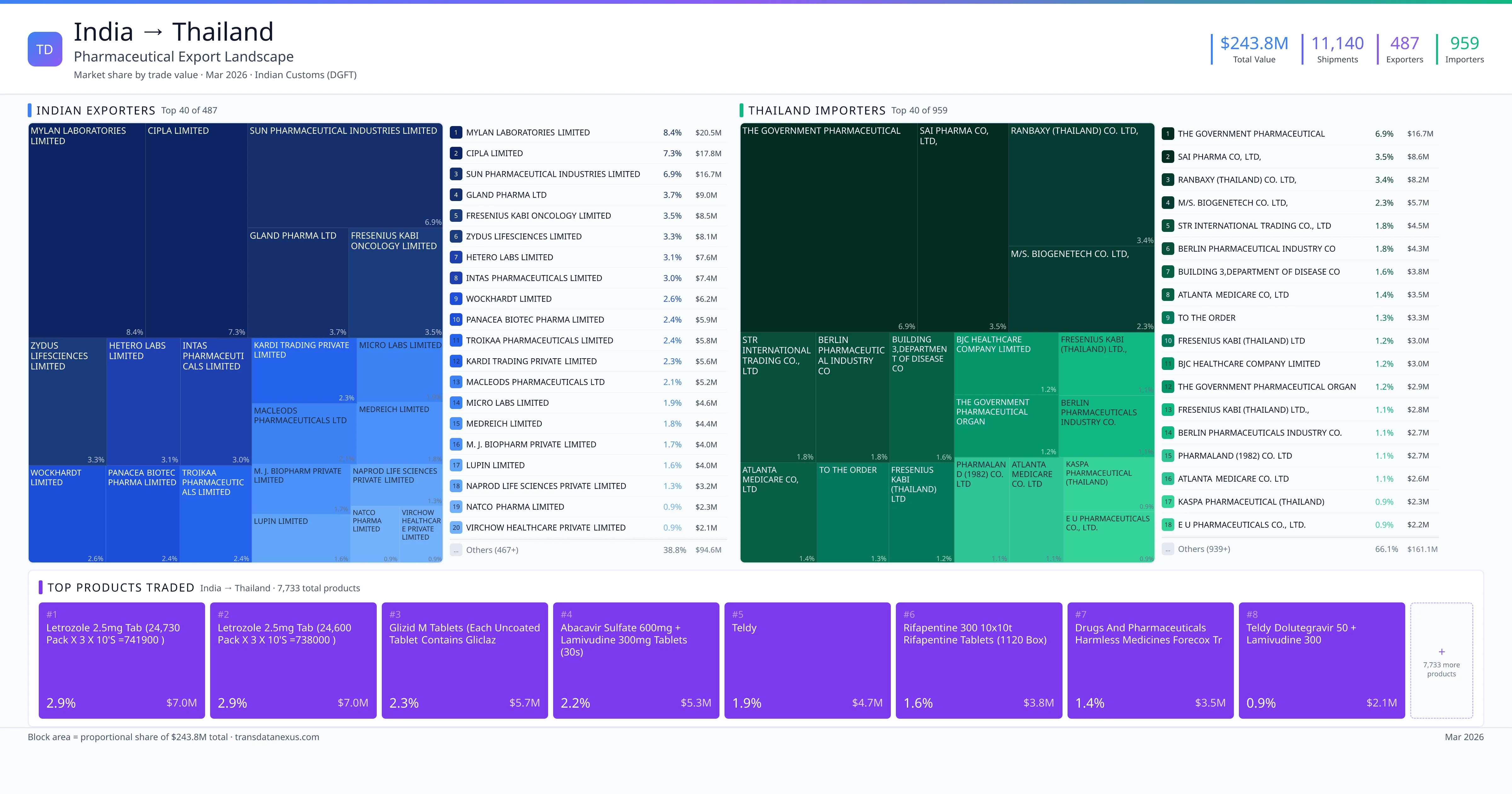
Indian pharmaceutical companies seeking to export finished pharmaceutical products to Thailand must navigate a structured registration process overseen by the Thai FDA. The process involves several key steps:
1. Drug Facility Licensing: Overseas manufacturing facilities must obtain GMP clearance from the Thai FDA. This clearance ensures that the manufacturing processes meet international quality standards. (en.fda.moph.go.th)
2. Product Registration: Companies must submit a comprehensive application that includes a completed application form, drug samples, and technical documents detailing the drug's quality, efficacy, and safety. (en.fda.moph.go.th)
3. Approval Process: The Thai FDA reviews the submitted dossier, which should adhere to the Common Technical Document (CTD) format. This standardized format facilitates the evaluation process and ensures consistency in submissions.
The entire registration process can be time-consuming, often taking several months, depending on the complexity of the product and the completeness of the submission. Therefore, Indian exporters are advised to engage with local regulatory consultants to navigate the process efficiently.