India to Tanzania Pharmaceutical Export
Bilateral Trade Intelligence · $508.8M Total Trade · 476 Exporters · 710 Buyers · DGFT Verified · Updated March 2026
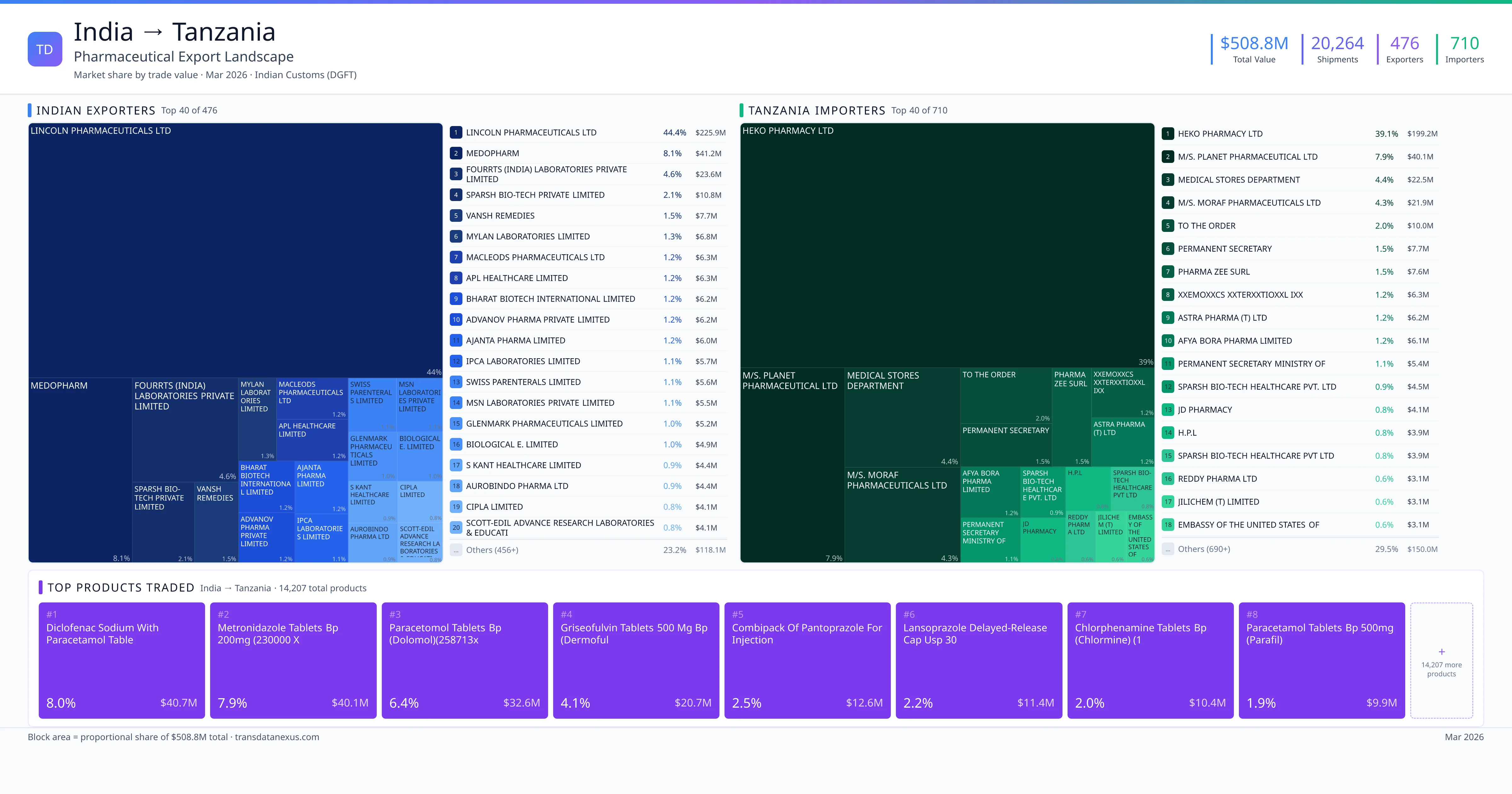
India exported $508.8M worth of pharmaceutical formulations to Tanzania across 20,264 verified shipments, sourced from 476 Indian exporters supplying 710 Tanzania buyers. The top exporters are LINCOLN PHARMACEUTICALS LTD ($225.9M) and MEDOPHARM ($41.2M). The leading products are Diclofenac ($40.7M) and Metronidazole ($40.1M). Average shipment value: $25.1K.
Top Pharmaceutical Formulations — India to Tanzania
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Diclofenac Sodium with Paracetamol Table | $40.7M | 8.0% |
| 2 | Metronidazole Tablets BP 200mg (230000 X | $40.1M | 7.9% |
| 3 | Paracetomol Tablets BP (dolomol)(258713x | $32.6M | 6.4% |
| 4 | Griseofulvin Tablets 500 MG BP (dermoful | $20.7M | 4.1% |
| 5 | Combipack of Pantoprazole for Injection | $12.6M | 2.5% |
| 6 | Lansoprazole Delayed-release Capsules USP 30 | $11.4M | 2.2% |
| 7 | Chlorphenamine Tablets BP (chlormine) (1 | $10.4M | 2.0% |
| 8 | Paracetamol Tablets BP 500mg (parafil) | $9.9M | 1.9% |
| 9 | Captopril Tablets BP (capril-25) (23715x | $7.1M | 1.4% |
| 10 | Dolutegravir Lamivudine and Tenofovir | $6.3M | 1.2% |
| 11 | Hyoscine Butylbromide Injection BP 10 MG | $6.1M | 1.2% |
| 12 | Gentamicin Eye/ear Drops Bp? 0.3 % W/v | $5.8M | 1.1% |
| 13 | Azithromycin Oral Suspension 200 Mg/ 5 | $5.3M | 1.0% |
| 14 | Secnidazole Tablets BP 1000 MG (sac) (14 | $5.3M | 1.0% |
| 15 | Pharmaceutical Goods/harmless Medicines | $5.1M | 1.0% |
India exports 20+ pharmaceutical formulations to Tanzania with a combined trade value of $508.8M. Key products include Diclofenac ($40.7M), Metronidazole ($40.1M), Griseofulvin ($20.7M), Pantoprazole ($12.6M), Lansoprazole ($11.4M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 20,264 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Tanzania Trade Routes
These are the top pharmaceutical products exported from India to Tanzania, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Tanzania buyers, regulatory requirements, and logistics for that specific product corridor. Products include Diclofenac ($40.7M), Metronidazole ($40.1M), Griseofulvin ($20.7M), Pantoprazole ($12.6M), Lansoprazole ($11.4M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Tanzania
476 Indian pharmaceutical companies export finished formulations to Tanzania. Leading exporters include Lincoln Pharmaceuticals Ltd, Medopharm, Fourrts (india) Laboratories Private Limited, Sparsh Bio-tech Private Limited. The top exporter accounts for 44.4% of total India–Tanzania pharma exports. Source: Indian Customs (DGFT).
Top Tanzania Buyers from India
710 companies in Tanzania import pharmaceutical formulations from India. Top buyers include Heko Pharmacy Ltd, M/s. Planet Pharmaceutical Ltd, Medical Stores Department, M/s. Moraf Pharmaceuticals Ltd. The largest buyer accounts for 39.1% of India–Tanzania pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Tanzania Pharmaceutical Shipments
Indian Export Ports
Tanzania Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Tanzania Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and Tanzania has experienced significant growth over the past decade. In 2022, the total export value from India to Tanzania was approximately $508.8 million USD, encompassing 20,264 shipments. This robust trade relationship is supported by 476 Indian exporters and 710 Tanzanian buyers, with an average shipment value of $25,000. The primary regulatory authority overseeing this trade in Tanzania is the Tanzania Medicines and Medical Devices Authority (TMDA), which ensures the safety, quality, and efficacy of imported pharmaceutical products. (tmda.go.tz)
2India's Market Position
India holds a dominant position in Tanzania's pharmaceutical import market, supplying a substantial portion of the country's pharmaceutical needs. This strategic importance is underscored by the extensive network of Indian exporters and the diverse range of products offered, catering to various therapeutic areas critical to Tanzania's healthcare system.
3Recent Developments
Between 2024 and 2026, several key developments have influenced the India-Tanzania pharmaceutical trade. Notably, in April 2024, the TMDA updated its guidelines for the registration of medical devices and in-vitro diagnostic devices, streamlining the approval process for foreign manufacturers. This regulatory adjustment has facilitated increased market access for Indian pharmaceutical companies. Additionally, in June 2025, a bilateral trade agreement was signed between India and Tanzania, aiming to enhance trade relations and reduce tariffs on pharmaceutical exports, further strengthening the trade corridor.
EXPORT_REGULATORY
Tanzania Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to register products in Tanzania must adhere to the TMDA's guidelines, which require a comprehensive dossier in the Common Technical Document (CTD) format. This dossier should include detailed information on the product's quality, safety, and efficacy. The registration process involves a thorough evaluation by the TMDA, which may include clinical trials to establish acceptable evidence of safety and efficacy before the product can be marketed in Tanzania. (tmda.go.tz)
2GMP & Facility Requirements
Tanzania mandates that all pharmaceutical products imported into the country be manufactured in facilities that comply with Good Manufacturing Practice (GMP) standards. The TMDA conducts inspections to ensure that manufacturing sites meet these standards. While Tanzania recognizes GMP certifications from other countries, manufacturers must provide evidence of compliance, such as certificates of compliance to ISO 13485, to facilitate the approval process. (tmda.go.tz)
3Import Documentation
Importers of pharmaceutical products into Tanzania are required to obtain several key documents:
- Certificate of Pharmaceutical Product (CPP): Confirms that the product is authorized for sale in the country of origin.
- Certificate of Analysis (CoA): Provides detailed information on the product's quality and composition.
- GMP Certificate: Demonstrates that the manufacturing facility adheres to GMP standards.
These documents must be submitted to the TMDA as part of the importation process. Additionally, importers must ensure compliance with all customs procedures and regulations set forth by Tanzanian authorities. (tmda.go.tz)
EXPORT_TRENDS
Product Categories & Therapeutic Trends — India to Tanzania
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Tanzania are predominantly in the form of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. The top ten products exported in 2022 include:
1. Diclofenac Sodium With Paracetamol Tablets ($40.7M, 8.0%)
2. Metronidazole Tablets BP 200MG ($40.1M, 7.9%)
3. Paracetamol Tablets BP (Dolomol) ($32.6M, 6.4%)
4. Griseofulvin Tablets 500 mg BP (Dermoful) ($20.7M, 4.1%)
5. Combipack of Pantoprazole For Injection ($12.6M, 2.5%)
6. Lansoprazole Delayed-Release Cap USP 30 ($11.4M, 2.2%)
7. Chlorphenamine Tablets BP (Chlormine) ($10.4M, 2.0%)
8. Paracetamol Tablets BP 500MG (Parafil) ($9.9M, 1.9%)
9. Captopril Tablets BP (Capril-25) ($7.1M, 1.4%)
10. Dolutegravir Lamivudine and Tenofovir ($6.3M, 1.2%)
These products align with Tanzania's healthcare needs, addressing prevalent conditions such as pain, infections, gastrointestinal disorders, and chronic diseases.
2Emerging Opportunities
The expiration of patents for several blockbuster drugs presents opportunities for Indian pharmaceutical companies to introduce generic versions in Tanzania. Additionally, the growing demand for biosimilars in the region offers a promising avenue for expansion. Indian exporters can also explore new therapeutic areas, such as oncology and immunology, to meet the evolving healthcare needs of the Tanzanian population.
3Demand Drivers
Tanzania's disease burden, characterized by high rates of infectious diseases like malaria and HIV/AIDS, drives the demand for pharmaceutical imports. The aging population and increasing prevalence of non-communicable diseases, such as diabetes and hypertension, further contribute to the rising need for medications. Government initiatives to improve healthcare infrastructure and spending are also significant factors influencing import demand from India.
EXPORT_POLICY
Trade Policy & Tariff Intelligence — India and Tanzania
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Tanzania applies the Most Favored Nation (MFN) tariff rates to pharmaceutical imports, with specific duties and exemptions outlined in the East African Community (EAC) Common External Tariff. Certain pharmaceutical products may benefit from preferential rates or duty exemptions under bilateral agreements between India and Tanzania. Importers should consult the latest EAC and Tanzanian customs regulations to determine applicable tariffs and exemptions.
2Trade Agreements
The East African Community (EAC) Common Market Protocol facilitates the free movement of goods, including pharmaceuticals, among member states. Bilateral trade agreements between India and Tanzania aim to enhance trade relations and may include provisions for reducing tariffs and non-tariff barriers on pharmaceutical exports. Ongoing negotiations focus on further liberalizing trade and addressing regulatory harmonization to benefit pharmaceutical trade.
3IP & Patent Landscape
Tanzania's intellectual property laws, aligned with international agreements, provide protection for patents and data exclusivity. However, the country also has provisions for compulsory licensing, which can impact the entry of Indian generic pharmaceuticals. Indian exporters must navigate these IP regulations carefully to ensure compliance and protect their products in the Tanzanian market.
EXPORT_LOGISTICS
Supply Chain & Logistics — India to Tanzania Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to Tanzania primarily utilize sea and air routes. Major sea routes involve transshipment through ports in the Middle East or South Africa, with transit times ranging from 15 to 30 days, depending on the specific route and shipping conditions. Air freight offers faster delivery, typically within 7 to 10 days, but at a higher cost. Potential disruptions, such as those in the Red Sea, can affect shipping schedules and require alternative routing.
2Port Infrastructure
In India, key export ports include Ahmedabad ICD (4.1%), Nhava Sheva Sea (16.2%), and Chennai Air (0.4%). In Tanzania, primary import ports are Dar es Salaam (27.8%), Dar es Salaam (23.5%), and Dar es Salam (16.0%). These ports are equipped to handle pharmaceutical imports, with Dar es Salaam serving as the main gateway for goods entering Tanzania.
3Cold Chain & Compliance
Maintaining the integrity of pharmaceutical products during transit is crucial. Temperature-controlled logistics are essential for products requiring specific storage conditions. Compliance with Good Distribution Practice (GDP) standards ensures that products are stored and transported under appropriate conditions. Packaging must meet international standards to protect products from environmental factors and physical damage during transit.
EXPORT_OPPORTUNITY
Market Opportunity Assessment — Tanzania for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Tanzania's pharmaceutical market is experiencing steady growth, driven by an expanding population and increasing healthcare needs. The market is heavily import-dependent, with a significant share of pharmaceuticals sourced from abroad. Generics constitute a substantial portion of the market, offering opportunities for Indian exporters to supply cost-effective alternatives to branded medications.
2Healthcare System
The Tanzanian government has implemented various healthcare programs aimed at improving access to medicines and healthcare services. While insurance coverage is limited, there is a growing emphasis on expanding coverage and improving drug procurement systems to ensure a steady supply of essential medicines. These initiatives create a favorable environment for pharmaceutical imports.
FAQ — India to Tanzania Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Tanzania?
India exported pharmaceuticals worth $508.8M to Tanzania across 20,264 verified shipments.
Who are the top Indian pharmaceutical exporters to Tanzania?
1. LINCOLN PHARMACEUTICALS LTD — $225.9M. 2. MEDOPHARM — $41.2M. 3. FOURRTS (INDIA) LABORATORIES PRIVATE LIMITED — $23.6M. Total: 476 suppliers.
Which companies in Tanzania import pharmaceuticals from India?
1. HEKO PHARMACY LTD — $199.2M. 2. M/S. PLANET PHARMACEUTICAL LTD — $40.1M. 3. MEDICAL STORES DEPARTMENT — $22.5M. 710 buyers total.
What pharmaceutical products does India export most to Tanzania?
1. Diclofenac Sodium with Paracetamol Table ($40.7M, 8.0%); 2. Metronidazole Tablets BP 200mg (230000 X ($40.1M, 7.9%); 3. Paracetomol Tablets BP (dolomol)(258713x ($32.6M, 6.4%); 4. Griseofulvin Tablets 500 MG BP (dermoful ($20.7M, 4.1%); 5. Combipack of Pantoprazole for Injection ($12.6M, 2.5%)
Which ports handle pharmaceutical shipments from India to Tanzania?
Export: AHEMDABAD ICD, NHAVA SHEVA SEA (INNSA1), CHENNAI AIR, KAMARAJAR PORT, JNPT/ NHAVA SHEVA SEA. Import: Dar Es Salaam, DAR ES SALAAM, DAR ES SALAM (DARESS, Lusaka, DODOMA.
Why does Tanzania import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $508.8M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Tanzania?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Tanzania pharmaceutical trade?
$25.1K per consignment across 20,264 shipments.
How many Indian pharmaceutical companies export to Tanzania?
476 Indian companies. Largest: LINCOLN PHARMACEUTICALS LTD with $225.9M.
How can I find verified Indian pharmaceutical suppliers for Tanzania?
TransData Nexus covers 476 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Tanzania Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 20,264 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Tanzania identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 20,264 verified shipments from 476 Indian exporters to 710 Tanzania buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
476 Exporters
710 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists