India to Oman Pharmaceutical Export
Bilateral Trade Intelligence · $58.3M Total Trade · 371 Exporters · 447 Buyers · DGFT Verified · Updated March 2026
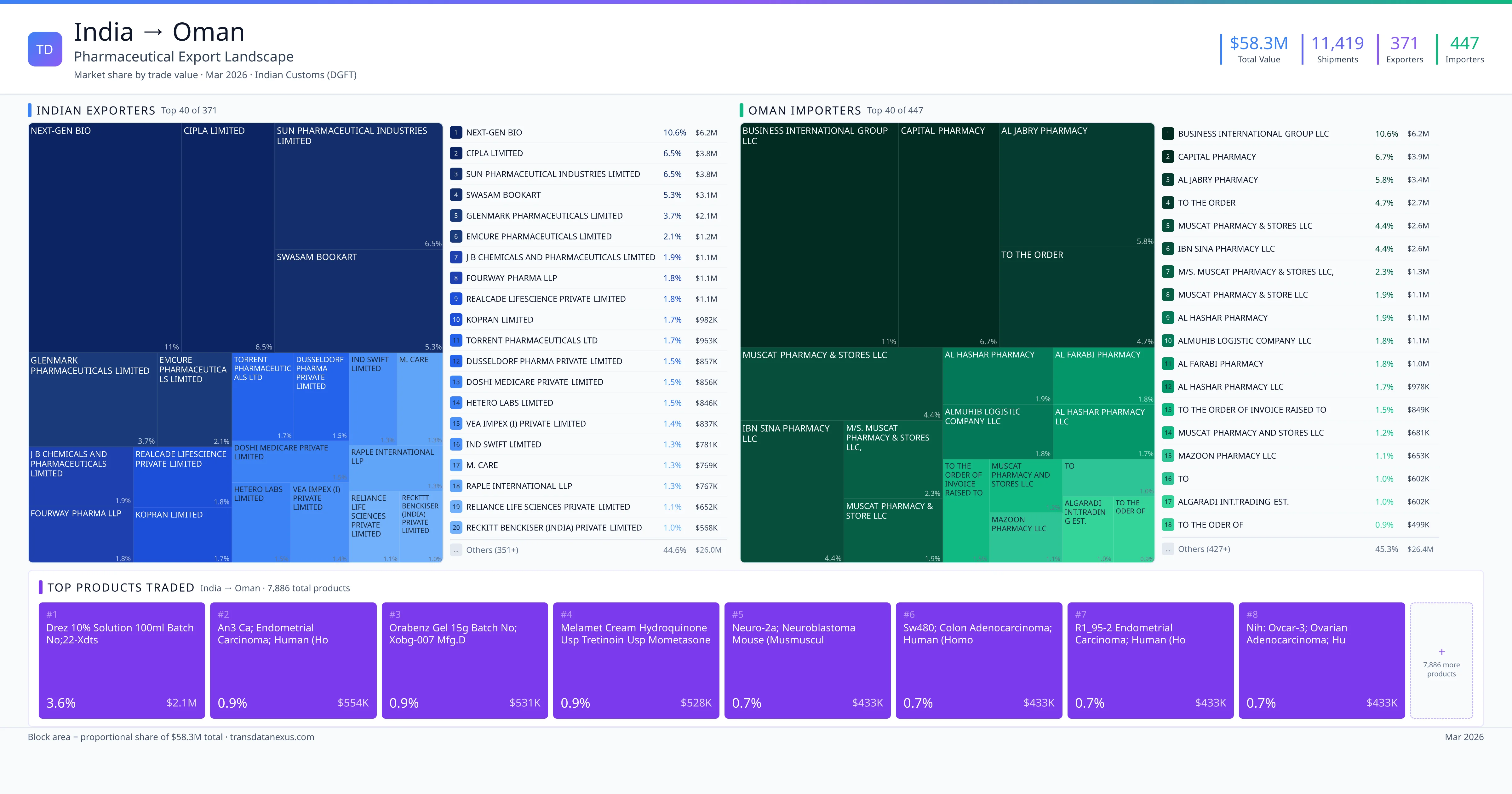
India exported $58.3M worth of pharmaceutical formulations to Oman across 11,419 verified shipments, sourced from 371 Indian exporters supplying 447 Oman buyers. The top exporters are NEXT-GEN BIO ($6.2M) and CIPLA LIMITED ($3.8M). The leading products are Drez 10% Solution 100ml Batch No;22-xdts, An3 Ca; Endometrial Carcinoma; Human (ho, Orabenz Gel 15g Batch No; Xobg-007 Mfg.d. Average shipment value: $5.1K.
Top Pharmaceutical Formulations — India to Oman
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Drez 10% Solution 100ml Batch No;22-xdts | $2.1M | 3.6% |
| 2 | An3 Ca; Endometrial Carcinoma; Human (ho | $553.5K | 0.9% |
| 3 | Orabenz Gel 15g Batch No; Xobg-007 Mfg.d | $531.0K | 0.9% |
| 4 | Melamet Cream Hydroquinone USP Tretinoin USP Mometasone Furoate IP in a Cream B… | $527.9K | 0.9% |
| 5 | Neuro-2a; Neuroblastoma Mouse (musmuscul | $433.0K | 0.7% |
| 6 | Sw480; Colon Adenocarcinoma; Human (homo | $433.0K | 0.7% |
| 7 | R1_95-2 Endometrial Carcinoma; Human (ho | $433.0K | 0.7% |
| 8 | Nih: Ovcar-3; Ovarian Adenocarcinoma; Hu | $433.0K | 0.7% |
| 9 | 293t; Embryonic Kidney Cells; Human (hom | $433.0K | 0.7% |
| 10 | Siha; Cervical Carcinoma; Human (homo Sa | $433.0K | 0.7% |
| 11 | Mcf7; Breast Adenocarcinoma; Human (homo | $433.0K | 0.7% |
| 12 | Hep G2 Hepatocellular; Carcinoma; Human | $433.0K | 0.7% |
| 13 | Panc-1; Pancreatic Carcinoma; Human (hom | $433.0K | 0.7% |
| 14 | Hela; Cervical Adenocarcinoma-ccl-2 (1ml | $433.0K | 0.7% |
| 15 | C-33 A; Cervical Carcinoma; Human (homo | $433.0K | 0.7% |
India exports 20+ pharmaceutical formulations to Oman with a combined trade value of $58.3M.. These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 11,419 verified Indian Customs (DGFT) shipment records.
Top Indian Exporters to Oman
371 Indian pharmaceutical companies export finished formulations to Oman. Leading exporters include Next-gen Bio, Cipla Limited, Sun Pharmaceutical Industries Limited, Swasam Bookart. The top exporter accounts for 10.6% of total India–Oman pharma exports. Source: Indian Customs (DGFT).
Top Oman Buyers from India
447 companies in Oman import pharmaceutical formulations from India. Top buyers include Business International Group Llc, Capital Pharmacy, Al Jabry Pharmacy, To The Order. The largest buyer accounts for 10.6% of India–Oman pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Oman Pharmaceutical Shipments
Indian Export Ports
Oman Import Ports
Trade Statistics
Other Destinations
India–Oman Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The India-Oman pharmaceutical trade corridor has experienced significant growth over the past decade. In 2013-14, India's pharmaceutical exports to Oman were valued at approximately $30 million. By 2024, this figure had increased to $52.44 million, reflecting a substantial rise in bilateral trade. This growth is attributed to India's competitive pricing, adherence to international quality standards, and Oman's increasing demand for affordable healthcare solutions.
A pivotal development occurred in December 2025 with the signing of the Comprehensive Economic Partnership Agreement (CEPA) between India and Oman. This agreement granted Indian pharmaceutical products duty-free access to the Omani market, including finished medicines and vaccines. Additionally, the CEPA introduced fast-track marketing authorizations for products approved by stringent regulatory authorities such as the USFDA, EMA, UK's MHRA, and Australia's TGA. These measures have streamlined the registration process, reducing approval timelines and compliance costs for Indian exporters.
2India's Market Position
India holds a dominant position in Oman's pharmaceutical imports, accounting for a significant share of the market. In 2024, India's pharmaceutical exports to Oman were valued at $52.44 million, making it one of the leading suppliers to the Omani market. This strong market presence is bolstered by India's extensive portfolio of generic medicines, competitive pricing, and compliance with international quality standards. The CEPA further enhances India's strategic importance, providing near-universal duty-free access to the Omani market and facilitating faster market entry for Indian pharmaceutical products.
3Recent Developments
Between 2024 and 2026, several key developments have influenced the India-Oman pharmaceutical trade corridor. The CEPA, signed in December 2025, has been instrumental in enhancing trade relations by providing duty-free access and expediting regulatory approvals for Indian pharmaceutical products. Additionally, in May 2022, Oman agreed to fast-track the approval process for Indian pharmaceutical products already registered by authorities in the US, UK, and EU, further facilitating market access for Indian exporters. These developments have collectively strengthened the bilateral trade relationship and opened new avenues for Indian pharmaceutical companies in the Omani market.
Oman Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to register products in Oman must adhere to the guidelines set by Oman's National Drug Regulatory Authority, which follows WHO-GMP standards. The registration process involves submitting a comprehensive dossier, typically in the Common Technical Document (CTD) or electronic CTD (eCTD) format, detailing the product's quality, safety, and efficacy. Oman accepts Good Manufacturing Practice (GMP) certificates and inspection outcomes from recognized stringent regulatory authorities, including the USFDA, EMA, UK's MHRA, and Australia's TGA. This mutual recognition reduces duplication and compliance costs for Indian exporters. The CEPA further facilitates the registration process by allowing products approved by these authorities to receive marketing authorization within 90 days, subject to submission of complete assessment dossiers.
2GMP & Facility Requirements
Oman requires Indian manufacturing sites to comply with WHO-GMP standards. The inspection process involves evaluating the manufacturing facilities to ensure they meet these international quality standards. Oman accepts GMP certificates and inspection outcomes from recognized stringent regulatory authorities, including the USFDA, EMA, UK's MHRA, and Australia's TGA. This mutual recognition reduces duplication and compliance costs for Indian exporters. The CEPA further facilitates the registration process by allowing products approved by these authorities to receive marketing authorization within 90 days, subject to submission of complete assessment dossiers.
3Import Documentation
To import pharmaceutical products into Oman, Indian exporters must provide several key documents:
- Certificate of Pharmaceutical Product (CPP): Confirms the product's registration and compliance with GMP standards.
- Certificate of Analysis (CoA): Details the product's quality and composition.
- GMP Certificate: Verifies that the manufacturing facility adheres to WHO-GMP standards.
These documents are essential for customs clearance and regulatory approval in Oman. The CEPA has streamlined the import process by providing duty-free access and expediting regulatory approvals for Indian pharmaceutical products.
Product Categories & Therapeutic Trends — India to Oman
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Oman are predominantly finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. These products align with Oman's healthcare needs, addressing prevalent conditions such as endometrial carcinoma, colon adenocarcinoma, and ovarian adenocarcinoma. The top products exported include DREZ 10% Solution, AN3 CA for endometrial carcinoma, and various gels and creams for dermatological conditions. This focus on finished formulations caters to the demand for ready-to-use medications in Oman's healthcare system.
2Emerging Opportunities
The CEPA has opened new avenues for Indian pharmaceutical exporters to supply a broader range of products to Oman. With duty-free access and expedited regulatory approvals, Indian companies can explore opportunities in biosimilars, novel drug formulations, and other therapeutic areas that align with Oman's evolving healthcare needs. The mutual recognition of GMP standards and fast-track marketing authorizations further enhance the attractiveness of the Omani market for Indian exporters.
3Demand Drivers
Oman's increasing disease burden, aging population, and rising healthcare expenditures are significant drivers of pharmaceutical import demand. The country's healthcare system relies heavily on imports to meet the needs of its population, creating a substantial market for foreign pharmaceutical products. The CEPA's provisions, including duty-free access and expedited regulatory processes, position Indian pharmaceutical companies to effectively meet this growing demand.
Trade Policy & Tariff Intelligence — India and Oman
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Under the CEPA, Indian pharmaceutical products, including finished medicines and vaccines, receive duty-free access to the Omani market. This preferential treatment eliminates tariffs that were previously up to 5%, enhancing the price competitiveness of Indian exports. The agreement also provides for fast-track marketing authorizations for products approved by stringent regulatory authorities, further facilitating market entry.
2Trade Agreements
The CEPA, signed in December 2025, is a significant bilateral trade agreement between India and Oman. It grants Indian pharmaceutical products duty-free access to the Omani market and introduces fast-track marketing authorizations for products approved by recognized stringent regulatory authorities. These provisions are expected to boost India's pharmaceutical exports to Oman and strengthen bilateral trade relations.
3IP & Patent Landscape
Oman's intellectual property and patent laws are aligned with international standards, providing protection for pharmaceutical innovations. The CEPA includes provisions for the protection of intellectual property rights, ensuring that Indian pharmaceutical companies' patents and data exclusivity are recognized and enforced in Oman. This legal framework supports the entry of Indian generic drugs into the Omani market, fostering a competitive environment.
Supply Chain & Logistics — India to Oman Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Major sea and air routes facilitate the export of pharmaceutical products from India to Oman. Sea shipments typically depart from ports such as Nhava Sheva and Chennai, while air shipments are often routed through airports like Sahar and Chennai. Transit times vary depending on the mode of transport and specific routes but are generally efficient, ensuring timely delivery of pharmaceutical products. The CEPA's provisions have further streamlined logistics by reducing customs procedures and facilitating faster clearance.
2Port Infrastructure
Key export ports in India include Chennai Air, Nhava Sheva Sea (INNSA1), and Sahar Air. In Oman, major import ports handling pharmaceutical shipments are Muscat, Sohar, and Salalah. These ports are equipped with facilities to handle pharmaceutical imports, ensuring compliance with international standards and efficient processing of shipments. The CEPA has enhanced port operations by simplifying customs procedures and reducing clearance times for pharmaceutical products.
3Cold Chain & Compliance
Temperature-controlled logistics are essential for maintaining the efficacy of pharmaceutical products during transit. Indian exporters adhere to Good Distribution Practice (GDP) compliance and packaging standards to ensure product integrity. The CEPA's provisions have facilitated compliance by standardizing requirements and reducing bureaucratic hurdles, allowing for more efficient cold chain management.
FAQ — India to Oman Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Oman?
India exported pharmaceuticals worth $58.3M to Oman across 11,419 verified shipments.
Who are the top Indian pharmaceutical exporters to Oman?
1. NEXT-GEN BIO — $6.2M. 2. CIPLA LIMITED — $3.8M. 3. SUN PHARMACEUTICAL INDUSTRIES LIMITED — $3.8M. Total: 371 suppliers.
Which companies in Oman import pharmaceuticals from India?
1. BUSINESS INTERNATIONAL GROUP LLC — $6.2M. 2. CAPITAL PHARMACY — $3.9M. 3. AL JABRY PHARMACY — $3.4M. 447 buyers total.
What pharmaceutical products does India export most to Oman?
1. Drez 10% Solution 100ml Batch No;22-xdts ($2.1M, 3.6%); 2. An3 Ca; Endometrial Carcinoma; Human (ho ($553.5K, 0.9%); 3. Orabenz Gel 15g Batch No; Xobg-007 Mfg.d ($531.0K, 0.9%); 4. Melamet Cream Hydroquinone USP Tretinoin USP Mometasone Furoate IP in a Cream B… ($527.9K, 0.9%); 5. Neuro-2a; Neuroblastoma Mouse (musmuscul ($433.0K, 0.7%)
Which ports handle pharmaceutical shipments from India to Oman?
Export: CHENNAI AIR, NHAVA SHEVA SEA (INNSA1), SAHAR AIR, SAHAR AIR CARGO ACC (INBOM4), JNPT/ NHAVA SHEVA SEA. Import: MUSCAT, Sohar, SALALAH, Salalah, SOHAR.
Why does Oman import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $58.3M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Oman?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Oman pharmaceutical trade?
$5.1K per consignment across 11,419 shipments.
How many Indian pharmaceutical companies export to Oman?
371 Indian companies. Largest: NEXT-GEN BIO with $6.2M.
How can I find verified Indian pharmaceutical suppliers for Oman?
TransData Nexus covers 371 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Oman Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 11,419 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Oman identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 11,419 verified shipments from 371 Indian exporters to 447 Oman buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
371 Exporters
447 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists