India to Malta Pharmaceutical Export
Bilateral Trade Intelligence · $550.6M Total Trade · 84 Exporters · 174 Buyers · DGFT Verified · Updated March 2026
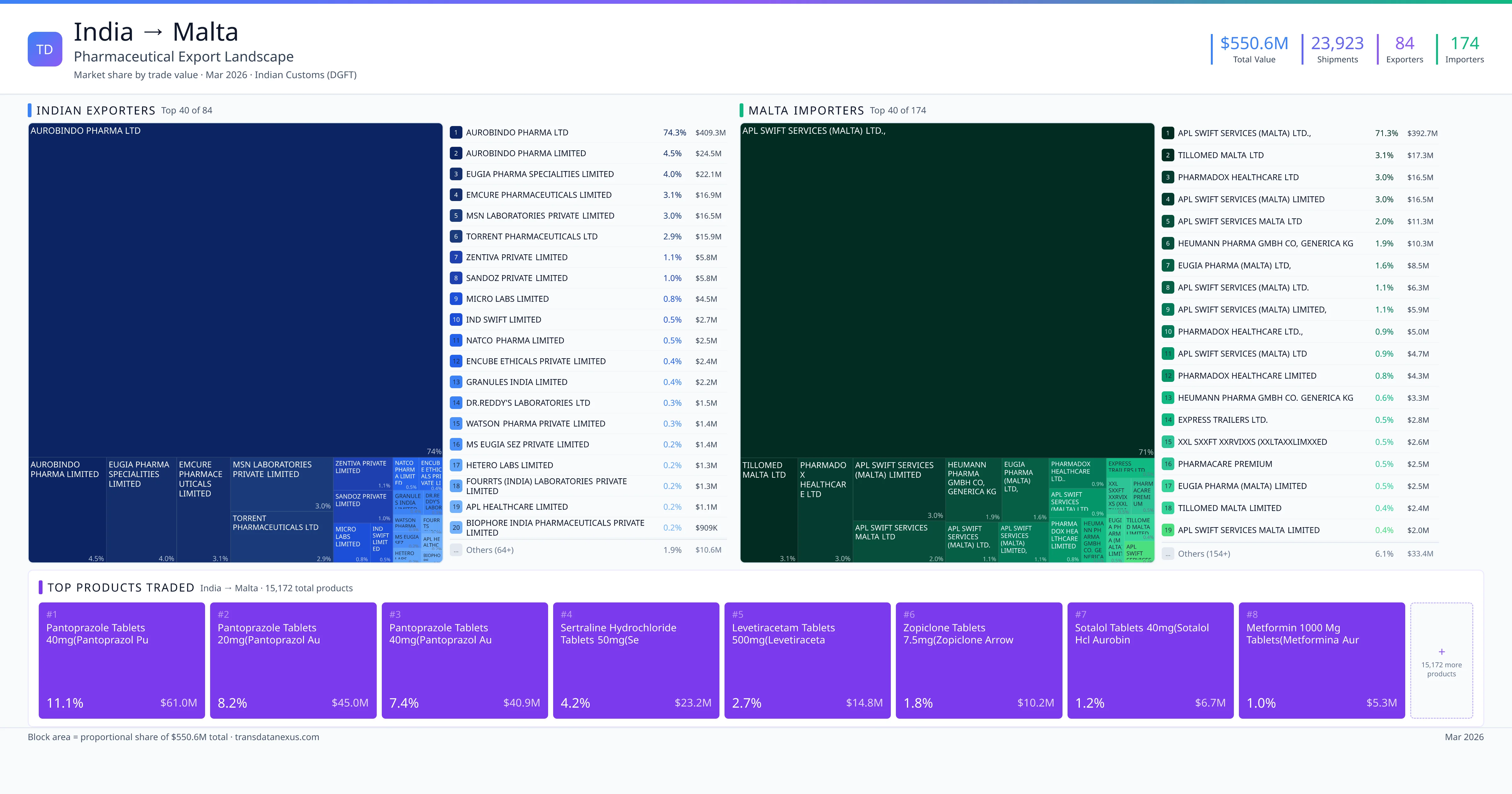
India exported $550.6M worth of pharmaceutical formulations to Malta across 23,923 verified shipments, sourced from 84 Indian exporters supplying 174 Malta buyers. The top exporters are AUROBINDO PHARMA LTD ($409.3M) and AUROBINDO PHARMA LIMITED ($24.5M). The leading products are Pantoprazole ($61.0M) and Sertraline ($23.2M). Average shipment value: $23.0K.
Top Pharmaceutical Formulations — India to Malta
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Pantoprazole Tablets 40mg(pantoprazol Pu | $61.0M | 11.1% |
| 2 | Pantoprazole Tablets 20mg(pantoprazol Au | $45.0M | 8.2% |
| 3 | Pantoprazole Tablets 40mg(pantoprazol Au | $40.9M | 7.4% |
| 4 | Sertraline Hydrochloride Tablets 50mg(se | $23.2M | 4.2% |
| 5 | Levetiracetam Tablets 500mg(levetiraceta | $14.8M | 2.7% |
| 6 | Zopiclone Tablets 7.5mg(zopiclone Arrow | $10.2M | 1.8% |
| 7 | Sotalol Tablets 40mg(sotalol Hcl Aurobin | $6.7M | 1.2% |
| 8 | Metformin 1000 MG Tablets(metformina Aur | $5.3M | 1.0% |
| 9 | Candesartan Heumann 8mg Tabletten (candesartan 8mg) (26435 Packs X 7x14s = 2590… | $5.0M | 0.9% |
| 10 | Sodium Valproate Tablets 500mg(natriumva | $4.7M | 0.9% |
| 11 | Sertraline Hydrochloride Tablets 100mg(s | $4.6M | 0.8% |
| 12 | Atorvastatin 80 MG Tablets(atorvastatina | $4.6M | 0.8% |
| 13 | Sodium Valproate Tablets 500mg(sodio Val | $4.4M | 0.8% |
| 14 | Macrogol 4 G Oral Solution(macrogol Auro | $4.0M | 0.7% |
| 15 | Macrogol 4 G Oral Solution(macrogol Ab 4 | $4.0M | 0.7% |
India exports 19+ pharmaceutical formulations to Malta with a combined trade value of $550.6M. Key products include Pantoprazole ($61.0M), Sertraline ($23.2M), Metformin ($5.3M), Sodium ($4.7M), Atorvastatin ($4.6M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 23,923 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Malta Trade Routes
These are the top pharmaceutical products exported from India to Malta, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Malta buyers, regulatory requirements, and logistics for that specific product corridor. Products include Pantoprazole ($61.0M), Sertraline ($23.2M), Metformin ($5.3M), Sodium ($4.7M), Atorvastatin ($4.6M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Malta
84 Indian pharmaceutical companies export finished formulations to Malta. Leading exporters include Aurobindo Pharma Ltd, Aurobindo Pharma Limited, Eugia Pharma Specialities Limited, Emcure Pharmaceuticals Limited. The top exporter accounts for 74.3% of total India–Malta pharma exports. Source: Indian Customs (DGFT).
Top Malta Buyers from India
174 companies in Malta import pharmaceutical formulations from India. Top buyers include Apl Swift Services (malta) Ltd.,, Tillomed Malta Ltd, Pharmadox Healthcare Ltd, Apl Swift Services (malta) Limited. The largest buyer accounts for 71.3% of India–Malta pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Malta Pharmaceutical Shipments
Indian Export Ports
Malta Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Malta Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The India-Malta pharmaceutical trade corridor has experienced significant growth over the past decade. In the fiscal year 2022–23, India's pharmaceutical exports to Malta were valued at $312.76 million, marking a substantial increase from $208.20 million in 2019–20. This upward trajectory continued into 2024, with exports reaching $422.69 million, according to the United Nations COMTRADE database. This growth underscores the strengthening bilateral trade relations and Malta's increasing reliance on Indian pharmaceutical products.
Key milestones include the establishment of the Malta Life Sciences Park and the Centre for Molecular Medicine and Biobanking, which have bolstered Malta's pharmaceutical sector and facilitated deeper collaboration with Indian exporters. (eurohealthobservatory.who.int) Additionally, the signing of a cultural agreement in 1992 laid the foundation for broader bilateral engagements, encompassing trade and investment in the pharmaceutical domain.
2India's Market Position
India holds a dominant position in Malta's pharmaceutical imports, accounting for approximately 80% of the total pharmaceutical imports by value. This substantial share highlights India's strategic importance as a supplier of high-quality, cost-effective pharmaceutical formulations. The dominance is further evidenced by the fact that in 2024, India's pharmaceutical exports to Malta were valued at $422.69 million, while Malta's total pharmaceutical imports stood at $518.85 million.
This strong market presence is attributed to India's robust manufacturing capabilities, adherence to international quality standards, and competitive pricing. The Indian pharmaceutical industry's ability to cater to diverse therapeutic needs aligns well with Malta's healthcare requirements, reinforcing India's strategic importance in the Maltese pharmaceutical market.
3Recent Developments
Between 2024 and 2026, several developments have influenced the India-Malta pharmaceutical trade. In February 2026, the Indian pharmaceutical industry reported a 9.4% growth in exports for the fiscal year 2024–25, reaching $30.47 billion. The industry aims for double-digit expansion in 2026–27, indicating a positive outlook for future exports to markets like Malta.
Additionally, in 2025, the Indian Ministry of External Affairs and Malta's Ministry of Foreign and European Affairs & Trade held discussions to review bilateral engagements, including trade and investments in the pharmaceutical sector. These talks focused on enhancing cooperation and addressing mutual interests, potentially leading to policy adjustments favorable to pharmaceutical trade.
Malta Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to export products to Malta must navigate the European Medicines Agency (EMA) regulatory framework. The Common Technical Document (CTD) format is mandatory for product dossiers, ensuring consistency and facilitating the evaluation process. Bioequivalence studies are required for generic products to demonstrate therapeutic equivalence to the reference listed drug. The registration timeline varies but typically ranges from 12 to 18 months, depending on the complexity of the product and the completeness of the submission.
The EMA's centralized procedure allows for a single marketing authorization valid across all EU member states, including Malta. This streamlined process is advantageous for Indian exporters aiming to access the broader European market. However, maintaining compliance with EMA guidelines and ensuring timely submission of all required documentation are critical to avoid delays in market entry.
2GMP & Facility Requirements
Malta, as an EU member state, adheres to the Good Manufacturing Practice (GMP) standards set by the European Union. Indian manufacturing sites must comply with these standards, which encompass quality management systems, personnel qualifications, premises and equipment, documentation, production, and quality control. The inspection process involves both pre-authorization inspections and periodic audits to ensure ongoing compliance.
Mutual recognition agreements between the EU and India facilitate the acceptance of GMP certificates issued by recognized authorities in India. This recognition streamlines the approval process for Indian manufacturers, reducing the need for duplicate inspections and expediting market access. However, maintaining GMP compliance is an ongoing requirement, and any deviations can lead to regulatory actions, including suspension of marketing authorizations.
3Import Documentation
To import pharmaceutical products into Malta, Indian exporters must provide several key documents:
- Certificate of Pharmaceutical Product (CPP): Issued by the Central Drugs Standard Control Organisation (CDSCO), this certificate attests to the product's compliance with Indian regulations and its suitability for export.
- Certificate of Analysis (CoA): Demonstrates that the product meets the specified quality standards.
- GMP Certificate: Confirms that the manufacturing facility adheres to GMP standards.
Customs procedures for pharmaceutical imports into Malta involve detailed documentation checks, including verification of the above certificates, to ensure compliance with EU regulations. Any discrepancies or incomplete documentation can result in delays or rejection of shipments. Therefore, meticulous attention to detail in preparing and submitting import documentation is essential for smooth market entry.
Product Categories & Therapeutic Trends — India to Malta
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Malta are predominantly in the form of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. These products cater to a wide range of therapeutic areas, with a notable emphasis on gastrointestinal, cardiovascular, and central nervous system disorders. The dominance of these categories aligns with Malta's healthcare needs, which prioritize the treatment of chronic conditions prevalent in its population.
The high demand for generic medications in Malta, driven by cost considerations and the need for accessible healthcare solutions, further supports the export of these formulations. Indian manufacturers' ability to produce high-quality generics at competitive prices makes them well-positioned to meet this demand.
2Emerging Opportunities
The expiration of patents for several blockbuster drugs presents opportunities for Indian exporters to introduce generic versions into the Maltese market. Additionally, the growing demand for biosimilars in Europe, including Malta, offers a promising avenue for Indian companies with the necessary expertise and regulatory approvals. Exploring new therapeutic areas, such as oncology and immunology, where Malta's healthcare system is expanding, can also provide growth prospects for Indian exporters.
To capitalize on these opportunities, Indian pharmaceutical companies should invest in research and development, ensure compliance with EMA regulations, and establish strong partnerships with Maltese distributors and healthcare providers.
3Demand Drivers
Malta's aging population and the increasing prevalence of chronic diseases, such as diabetes and hypertension, are significant drivers of pharmaceutical import demand. The country's healthcare system emphasizes the provision of affordable and effective treatments, creating a favorable environment for the import of generic pharmaceutical products. Additionally, Malta's integration into the EU market necessitates adherence to EU-wide health standards, further boosting the demand for high-quality pharmaceutical imports.
The Maltese government's commitment to enhancing healthcare infrastructure and expanding access to medical services also contributes to the growing demand for pharmaceutical products. This trend presents sustained opportunities for Indian exporters to supply a diverse range of medications to meet Malta's evolving healthcare needs.
Trade Policy & Tariff Intelligence — India and Malta
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Malta, as part of the European Union, applies the EU's Most Favored Nation (MFN) tariff rates to pharmaceutical imports. These rates are generally low for pharmaceutical products, reflecting the EU's commitment to ensuring affordable access to medicines. Additionally, the EU offers preferential rates and duty exemptions for pharmaceutical formulations under various trade agreements, including the Economic Partnership Agreements (EPAs) with certain countries.
For Indian pharmaceutical exports, the duty structure is favorable, with minimal tariffs and exemptions that facilitate competitive pricing in the Maltese market. However, exporters must stay informed about any changes in EU trade policies or tariff schedules to maintain compliance and optimize their market positioning.
2Trade Agreements
India and the European Union have engaged in discussions to enhance trade relations, including in the pharmaceutical sector. While there is no specific Free Trade Agreement (FTA) between India and Malta, the EU's trade policies and agreements with India influence the pharmaceutical trade dynamics. Ongoing negotiations aim to improve market access, streamline regulatory processes, and address non-tariff barriers, potentially leading to more favorable conditions for Indian pharmaceutical exporters.
These developments underscore the importance of staying abreast of policy changes and engaging in proactive dialogue to leverage opportunities arising from evolving trade agreements.
3IP & Patent Landscape
Malta, as part of the EU, adheres to the European Patent Convention, which provides robust protection for pharmaceutical patents. Indian generic manufacturers must navigate this landscape carefully, ensuring that their products do not infringe on existing patents. Data exclusivity periods and supplementary protection certificates can extend patent protection, affecting the timing of generic market entry.
Compulsory licensing provisions exist within the EU framework, allowing for the production of generic versions of patented drugs under specific conditions, such as public health emergencies. Indian exporters should monitor these provisions to identify potential opportunities for market entry.
FAQ — India to Malta Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Malta?
India exported pharmaceuticals worth $550.6M to Malta across 23,923 verified shipments.
Who are the top Indian pharmaceutical exporters to Malta?
1. AUROBINDO PHARMA LTD — $409.3M. 2. AUROBINDO PHARMA LIMITED — $24.5M. 3. EUGIA PHARMA SPECIALITIES LIMITED — $22.1M. Total: 84 suppliers.
Which companies in Malta import pharmaceuticals from India?
1. APL SWIFT SERVICES (MALTA) LTD., — $392.7M. 2. TILLOMED MALTA LTD — $17.3M. 3. PHARMADOX HEALTHCARE LTD — $16.5M. 174 buyers total.
What pharmaceutical products does India export most to Malta?
1. Pantoprazole Tablets 40mg(pantoprazol Pu ($61.0M, 11.1%); 2. Pantoprazole Tablets 20mg(pantoprazol Au ($45.0M, 8.2%); 3. Pantoprazole Tablets 40mg(pantoprazol Au ($40.9M, 7.4%); 4. Sertraline Hydrochloride Tablets 50mg(se ($23.2M, 4.2%); 5. Levetiracetam Tablets 500mg(levetiraceta ($14.8M, 2.7%)
Which ports handle pharmaceutical shipments from India to Malta?
Export: VIZAG SEA, HYDERABAD ICD, HYDERABAD ICD (INSNF6), NHAVA SHEVA SEA (INNSA1), SAHAR AIR. Import: Malta Freeport Distr, MALTA, Malta (Valetta), MALTA (VALETTA), MARSAXLOKK.
Why does Malta import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $550.6M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Malta?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Malta pharmaceutical trade?
$23.0K per consignment across 23,923 shipments.
How many Indian pharmaceutical companies export to Malta?
84 Indian companies. Largest: AUROBINDO PHARMA LTD with $409.3M.
How can I find verified Indian pharmaceutical suppliers for Malta?
TransData Nexus covers 84 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Malta Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 23,923 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Malta identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 23,923 verified shipments from 84 Indian exporters to 174 Malta buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
84 Exporters
174 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists