India to Latvia Pharmaceutical Export
Bilateral Trade Intelligence · $135.0M Total Trade · 69 Exporters · 116 Buyers · DGFT Verified · Updated March 2026
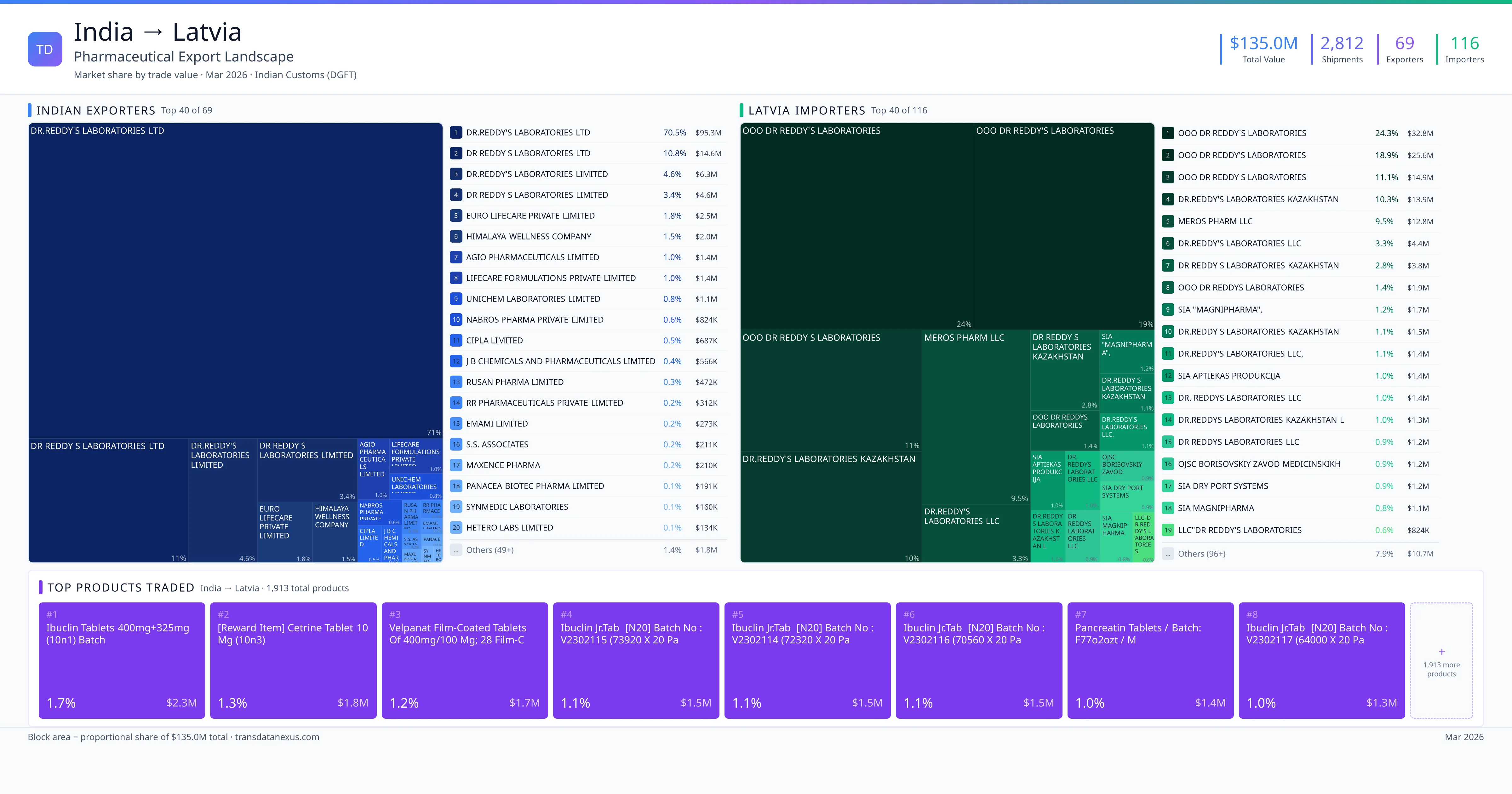
India exported $135.0M worth of pharmaceutical formulations to Latvia across 2,812 verified shipments, sourced from 69 Indian exporters supplying 116 Latvia buyers. The top exporters are DR.REDDY'S LABORATORIES LTD ($95.3M) and DR REDDY S LABORATORIES LTD ($14.6M). The leading products are Sofosbuvir ($1.7M). Average shipment value: $48.0K.
Top Pharmaceutical Formulations — India to Latvia
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Ibuclin Tablets 400mg+325mg (10n1) Batch | $2.3M | 1.7% |
| 2 | [reward Item] Cetrine Tablet 10 MG (10n3) | $1.8M | 1.3% |
| 3 | Velpanat Film-coated Tablets of 400mg/100 Mg; 28 Film-coated Tablets in a | $1.7M | 1.2% |
| 4 | Ibuclin Jr.tablets [n20] Batch No : V2302115 (73920 X 20 Packs = 1478400 ) | $1.5M | 1.1% |
| 5 | Ibuclin Jr.tablets [n20] Batch No : V2302114 (72320 X 20 Packs = 1446400 ) | $1.5M | 1.1% |
| 6 | Ibuclin Jr.tablets [n20] Batch No : V2302116 (70560 X 20 Packs = 1411200 ) | $1.5M | 1.1% |
| 7 | Pancreatin Tablets / Batch: F77o2ozt / M | $1.4M | 1.0% |
| 8 | Ibuclin Jr.tablets [n20] Batch No : V2302117 (64000 X 20 Packs = 1280000 ) | $1.3M | 1.0% |
| 9 | [reward Item] 300022733 Razo Enteric Coated Tablets 20 MG (15 No 2) | $1.3M | 1.0% |
| 10 | Nise Tablet 100 MG (10n2) Batch No.v2301 | $1.3M | 1.0% |
| 11 | Ibuclin Tablets (n20) Batch No : V2302100 (39616 X 20 Packs = 792320 ) | $1.2M | 0.9% |
| 12 | Nise Tablet 100 MG (10n3) ( 250500 Packsx 30 Tablets = 7515000 ) | $1.2M | 0.9% |
| 13 | Nise Tablet 100 MG (10n3) Batch No.v2500497 Mfg.dt.28.02.2025 Exp.dt.31.01.2028 | $976.7K | 0.7% |
| 14 | [reward Item]cetrine Tablet 10 MG (10n3) | $951.7K | 0.7% |
| 15 | 300000751 Cetrine Tablet 10 MG (10n2) (b | $876.2K | 0.6% |
India exports 20+ pharmaceutical formulations to Latvia with a combined trade value of $135.0M. Key products include Sofosbuvir ($1.7M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 2,812 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Latvia Trade Routes
These are the top pharmaceutical products exported from India to Latvia, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Latvia buyers, regulatory requirements, and logistics for that specific product corridor. Products include Sofosbuvir ($1.7M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Latvia
69 Indian pharmaceutical companies export finished formulations to Latvia. Leading exporters include Dr.reddy's Laboratories Ltd, Dr Reddy S Laboratories Ltd, Dr.reddy's Laboratories Limited, Dr Reddy S Laboratories Limited. The top exporter accounts for 70.5% of total India–Latvia pharma exports. Source: Indian Customs (DGFT).
Top Latvia Buyers from India
116 companies in Latvia import pharmaceutical formulations from India. Top buyers include Ooo Dr Reddy`s Laboratories, Ooo Dr Reddy's Laboratories, Ooo Dr Reddy S Laboratories, Dr.reddy's Laboratories Kazakhstan. The largest buyer accounts for 24.3% of India–Latvia pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Latvia Pharmaceutical Shipments
Indian Export Ports
Latvia Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Latvia Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The India-Latvia pharmaceutical trade corridor has experienced significant growth over the past decade. In 2024, India's pharmaceutical exports to Latvia reached approximately $139.71 million, marking a substantial increase from previous years. This upward trajectory reflects the strengthening economic ties and the expanding presence of Indian pharmaceutical products in the Latvian market.
Key milestones include the establishment of diplomatic relations and the opening of embassies, which have facilitated trade and economic cooperation. The Latvian pharmaceutical sector, known for its robust chemical and pharmaceutical industries, has been a significant contributor to this bilateral trade.
The growth trajectory indicates a promising future for the India-Latvia pharmaceutical trade corridor, with potential for further expansion and diversification of product offerings.
2India's Market Position
India holds a substantial share in Latvia's pharmaceutical imports, positioning itself as a key supplier in the Latvian market. In 2024, India's pharmaceutical exports to Latvia were valued at $139.71 million, indicating a strong presence.
This strategic importance is underscored by the complementary nature of the two countries' pharmaceutical sectors. India's extensive portfolio of generic medicines and cost-effective production aligns well with Latvia's demand for affordable and diverse pharmaceutical products.
The robust trade relationship highlights India's pivotal role in meeting Latvia's pharmaceutical needs, reinforcing its position as a preferred supplier.
3Recent Developments
Between 2024 and 2026, several developments have influenced the India-Latvia pharmaceutical trade. In October 2025, the opening of the Indian Embassy in Latvia marked a significant step in strengthening bilateral relations, potentially enhancing trade and economic cooperation.
Additionally, Latvia's pharmaceutical companies, such as Grindex, Olainfarm, and Kalceks, have been actively exporting to India, indicating a reciprocal interest in expanding trade ties.
These developments suggest a positive trajectory for the bilateral pharmaceutical trade, with potential for increased collaboration and market penetration in the coming years.
Latvia Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to export finished formulations to Latvia must navigate the regulatory framework established by the European Medicines Agency (EMA) and Latvia's National Competent Authority. The registration process involves submitting a comprehensive dossier, typically in the Common Technical Document (CTD) or electronic Common Technical Document (eCTD) format, which includes detailed information on the product's quality, safety, and efficacy.
A critical component of the registration is the demonstration of bioequivalence for generic products, ensuring therapeutic equivalence to the reference product. The evaluation timeline can vary, but applicants should anticipate a thorough review process, often extending several months, depending on the complexity of the submission and the workload of the regulatory authorities.
Engaging with local regulatory consultants or legal experts familiar with EMA procedures can facilitate a smoother registration process, ensuring compliance with all regulatory requirements.
2GMP & Facility Requirements
Latvia, adhering to EMA standards, requires that manufacturing facilities exporting pharmaceutical products meet Good Manufacturing Practice (GMP) standards. Indian manufacturers must ensure their facilities are compliant with these standards, which encompass quality management, personnel qualifications, premises and equipment, documentation, production, and quality control.
The inspection process involves a thorough assessment by Latvian or EMA inspectors to verify compliance with GMP standards. Mutual recognition agreements between Latvia and India may streamline this process, but manufacturers should be prepared for detailed inspections and potential corrective actions.
Maintaining up-to-date certifications and records, and proactively addressing any identified deficiencies, can facilitate smoother inspections and continued market access.
3Import Documentation
To export pharmaceutical formulations to Latvia, Indian companies must provide several key documents:
- Certificate of Pharmaceutical Product (CPP): Issued by the Central Drugs Standard Control Organisation (CDSCO), this certificate attests to the product's market authorization and compliance with Indian regulations.
- Certificate of Analysis (CoA): Demonstrates that the product meets specified quality standards.
- GMP Certificate: Confirms that the manufacturing facility adheres to GMP standards.
Customs procedures for pharmaceutical imports into Latvia require these documents to be submitted for clearance. Ensuring all documentation is accurate, complete, and up-to-date is crucial to prevent delays or rejections at customs.
Collaborating with experienced logistics partners familiar with Latvian import regulations can further streamline the process.
Product Categories & Therapeutic Trends — India to Latvia
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Latvia are predominantly composed of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. These products align with Latvia's healthcare needs, addressing prevalent conditions such as pain management, infectious diseases, and chronic illnesses.
The therapeutic areas dominating exports include analgesics, antivirals, and gastrointestinal medications. This focus reflects both India's manufacturing strengths and Latvia's demand for cost-effective, high-quality medicines to meet its healthcare requirements.
2Emerging Opportunities
The global pharmaceutical landscape is witnessing patent expirations and the rise of biosimilars, presenting new opportunities for Indian exporters. Latvia's interest in innovative therapies and cost-effective treatment options positions Indian pharmaceutical companies to expand their offerings in areas such as oncology, immunology, and biologics.
Additionally, Latvia's emphasis on sustainable and eco-friendly products opens avenues for Indian manufacturers to collaborate on green pharmaceutical initiatives, aligning with global sustainability goals.
3Demand Drivers
Latvia's aging population and increasing prevalence of chronic diseases are significant drivers of pharmaceutical import demand. The country's healthcare spending trends indicate a growing focus on quality healthcare services, further boosting the need for diverse pharmaceutical products.
India's ability to provide a wide range of affordable and high-quality medicines positions it as a key supplier to meet these evolving demands in Latvia's healthcare sector.
Trade Policy & Tariff Intelligence — India and Latvia
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Latvia, as an EU member state, applies the EU's Most Favored Nation (MFN) tariff rates to pharmaceutical imports. Pharmaceutical formulations entering Latvia are subject to these standard rates, with potential preferential rates available under specific trade agreements.
Duty exemptions may apply for certain products, particularly those deemed essential or for public health purposes. Staying informed about the latest tariff structures and any changes is essential for Indian exporters to optimize their pricing strategies.
2Trade Agreements
As of March 2026, there are no specific Free Trade Agreements (FTAs) between India and Latvia. However, both countries are part of broader international trade frameworks that facilitate pharmaceutical trade. Ongoing negotiations and potential future agreements could further enhance bilateral trade relations.
Monitoring developments in international trade policies and engaging in diplomatic channels can provide Indian exporters with insights into upcoming opportunities.
3IP & Patent Landscape
Latvia adheres to the European Union's intellectual property (IP) regulations, including patent provisions and data exclusivity periods. Indian generic manufacturers must navigate these IP landscapes, ensuring compliance with patent laws and respecting data exclusivity periods to avoid legal challenges.
Understanding Latvia's IP framework is crucial for Indian exporters to develop strategies that balance innovation with market access, ensuring sustainable growth in the Latvian market.
Supply Chain & Logistics — India to Latvia Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Major sea routes from Indian ports to Latvia involve transshipment hubs such as Rotterdam or Hamburg, with total transit times ranging from 25 to 35 days, depending on the specific ports and shipping lines used. Air freight offers faster delivery, with transit times of approximately 7 to 10 days, though at a higher cost.
Recent geopolitical events, such as disruptions in the Red Sea, have not significantly impacted these primary shipping routes, maintaining their reliability for pharmaceutical exports.
2Port Infrastructure
Key export ports in India include Hyderabad ICD, Baddi ICD, and other major ports handling pharmaceutical shipments. In Latvia, primary import ports are Riga and other Baltic Sea ports equipped to handle pharmaceutical imports.
These ports are equipped with facilities to manage pharmaceutical products, including temperature-controlled storage and handling, ensuring compliance with international shipping standards.
3Cold Chain & Compliance
Maintaining the integrity of the cold chain is critical for pharmaceutical exports. Indian exporters must ensure that logistics partners adhere to Good Distribution Practice (GDP) standards, utilizing temperature-controlled containers and monitoring systems throughout transit.
Proper packaging, including insulated materials and temperature indicators, is essential to prevent product degradation. Collaborating with experienced logistics providers familiar with both Indian and Latvian regulations can enhance compliance and ensure timely delivery.
Market Opportunity Assessment — Latvia for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Latvia's pharmaceutical
FAQ — India to Latvia Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Latvia?
India exported pharmaceuticals worth $135.0M to Latvia across 2,812 verified shipments.
Who are the top Indian pharmaceutical exporters to Latvia?
1. DR.REDDY'S LABORATORIES LTD — $95.3M. 2. DR REDDY S LABORATORIES LTD — $14.6M. 3. DR.REDDY'S LABORATORIES LIMITED — $6.3M. Total: 69 suppliers.
Which companies in Latvia import pharmaceuticals from India?
1. OOO DR REDDY`S LABORATORIES — $32.8M. 2. OOO DR REDDY'S LABORATORIES — $25.6M. 3. OOO DR REDDY S LABORATORIES — $14.9M. 116 buyers total.
What pharmaceutical products does India export most to Latvia?
1. Ibuclin Tablets 400mg+325mg (10n1) Batch ($2.3M, 1.7%); 2. [reward Item] Cetrine Tablet 10 MG (10n3) ($1.8M, 1.3%); 3. Velpanat Film-coated Tablets of 400mg/100 Mg; 28 Film-coated Tablets in a ($1.7M, 1.2%); 4. Ibuclin Jr.tablets [n20] Batch No : V2302115 (73920 X 20 Packs = 1478400 ) ($1.5M, 1.1%); 5. Ibuclin Jr.tablets [n20] Batch No : V2302114 (72320 X 20 Packs = 1446400 ) ($1.5M, 1.1%)
Which ports handle pharmaceutical shipments from India to Latvia?
Export: HYDERABAD ICD, BADDI ICD (INBDI6), HYDERABAD ICD (INSNF6), NHAVA SHEVA SEA, BADDI ICD. Import: Moskva, RIGA, Riga, Almaty, Kiev.
Why does Latvia import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $135.0M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Latvia?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Latvia pharmaceutical trade?
$48.0K per consignment across 2,812 shipments.
How many Indian pharmaceutical companies export to Latvia?
69 Indian companies. Largest: DR.REDDY'S LABORATORIES LTD with $95.3M.
How can I find verified Indian pharmaceutical suppliers for Latvia?
TransData Nexus covers 69 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Latvia Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 2,812 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Latvia identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 2,812 verified shipments from 69 Indian exporters to 116 Latvia buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
69 Exporters
116 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists