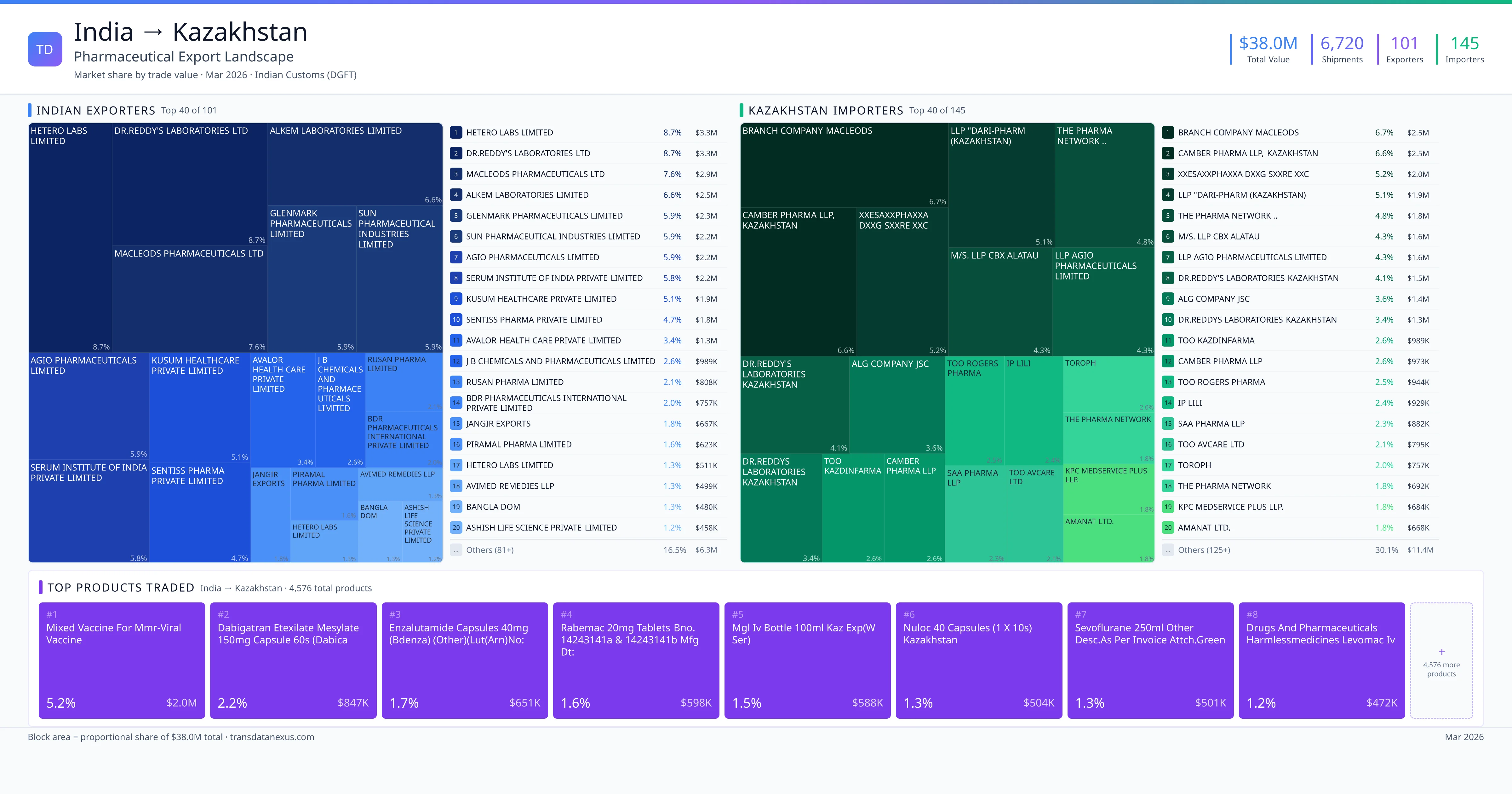
To export finished pharmaceutical formulations to Kazakhstan, Indian pharmaceutical companies must navigate a comprehensive registration process overseen by the National Center for Expertise of Medicines and Medical Devices (NCEMMD) under the Ministry of Health. This process ensures that imported medicines meet Kazakhstan's stringent standards for safety, efficacy, and quality.
Application Submission and Dossier Requirements
The registration process begins with the submission of an application accompanied by a detailed dossier. This dossier must include:
- An application form.
- Samples of labels and packaging.
- Certified copies of technical documentation, including the enterprise standard and instructions for use.
- Documents confirming the quality of the product and compliance with the claimed characteristics.
- Reports from accredited laboratories and research centers.
- Copies of documents detailing all essential attributes of the product, such as organoleptic, physical, microbiological, and chemical properties.
- Annotations with comprehensive information about the properties of the preparation, instructions for use, and precautions.
- Manufacturer’s founding documents.
For imported products, all documents in a foreign language must be translated into Russian, with translations certified accordingly. (medexpert.kz)
Expertise and Approval
Once the application and dossier are submitted, the NCEMMD conducts a thorough examination to assess the safety, efficacy, and quality of the medicinal product. This includes preclinical and clinical trials, as well as a pharmaceutical expert examination to identify all potential risks. The examination process is detailed in the Rules for Conducting the Examination of Medicines and Medical Devices, approved by the Minister of Health. (adalan.kz)
Pricing Registration
In addition to product registration, pharmaceutical companies must register the prices of their products. This involves submitting an application along with documents confirming the applicant's right to register the price under the statutory free medical assistance and/or in the compulsory social health insurance system. The application must also include information on the actual delivery price, supported by relevant documents such as purchase agreements and invoices from the last 12 months. (egov.kz)
Approval Timeline and Fees
The approval timeline for drug registration in Kazakhstan can vary depending on the complexity of the product and the completeness of the submitted dossier. While specific timelines are not explicitly stated, the process involves multiple stages, including application submission, dossier review, laboratory tests, and expert evaluations, which can extend over several months. The cost of obtaining a license for pharmaceutical activities is 10 monthly calculation indices (MCI), with the license issuance taking 15 working days. (egov.kz)