India to Iraq Pharmaceutical Export
Bilateral Trade Intelligence · $194.6M Total Trade · 260 Exporters · 379 Buyers · DGFT Verified · Updated March 2026
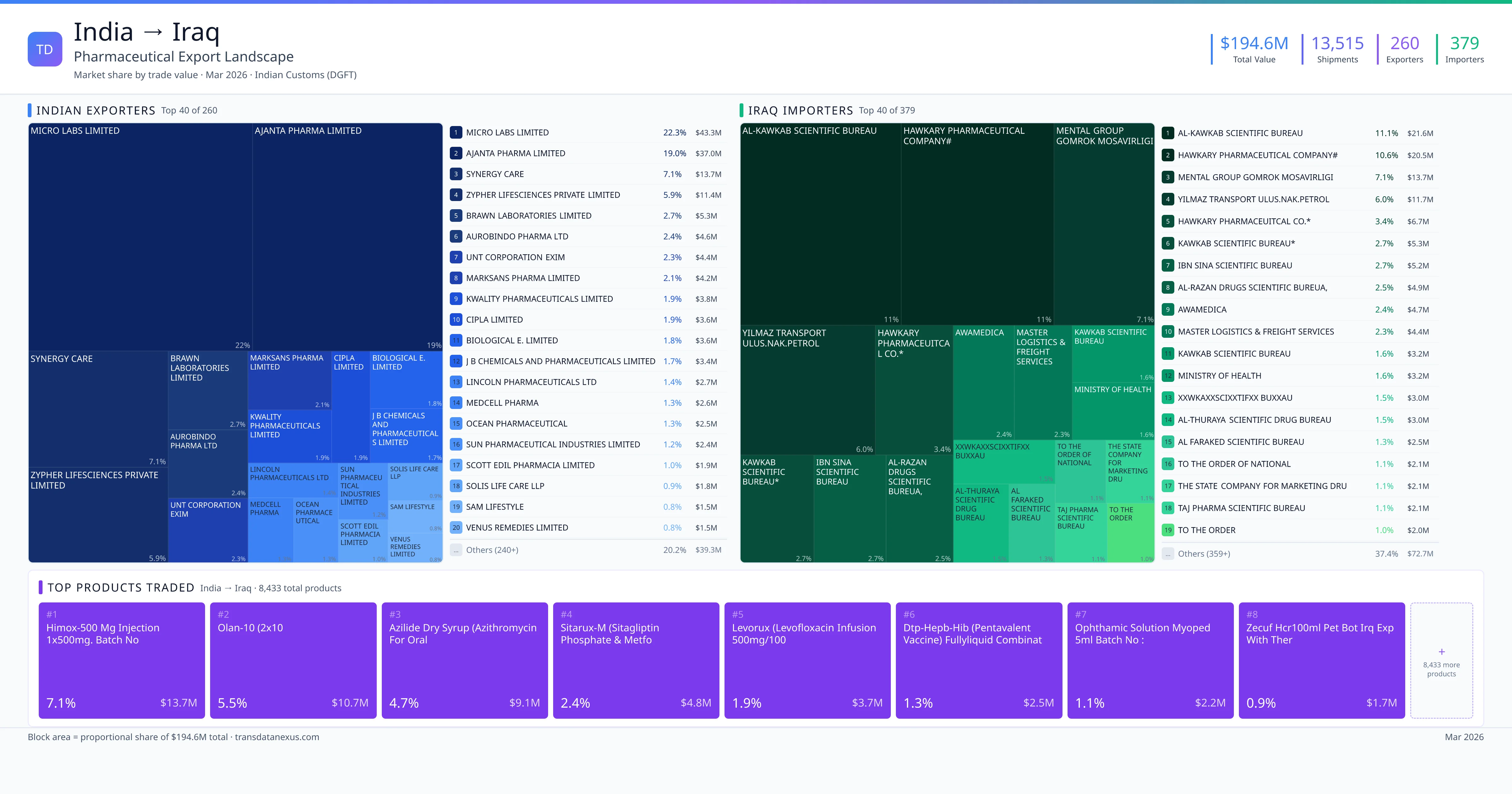
India exported $194.6M worth of pharmaceutical formulations to Iraq across 13,515 verified shipments, sourced from 260 Indian exporters supplying 379 Iraq buyers. The top exporters are MICRO LABS LIMITED ($43.3M) and AJANTA PHARMA LIMITED ($37.0M). The leading products are Sitagliptin ($4.8M) and Levofloxacin ($3.7M). Average shipment value: $14.4K.
Top Pharmaceutical Formulations — India to Iraq
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Himox-500 MG Injection 1x500mg. Batch No | $13.7M | 7.1% |
| 2 | Olan-10 (2x10 | $10.7M | 5.5% |
| 3 | Azilide Dry Syrup (azithromycin for Oral | $9.1M | 4.7% |
| 4 | Sitarux-m (sitagliptin Phosphate & Metfo | $4.8M | 2.4% |
| 5 | Levorux (levofloxacin Infusion 500mg/100 | $3.7M | 1.9% |
| 6 | Ophthamic Solution Myoped 5ml Batch No : | $2.2M | 1.1% |
| 7 | Zecuf Hcr100ml Pet Bot Irq Exp with Ther | $1.7M | 0.9% |
| 8 | Tamrux-d (tamsulosin Hydrochloride /duta | $1.6M | 0.8% |
| 9 | [reward Item] Dtp-hepb-hib (pentavalentvaccine) Fully Liquid Combination Vaccin… | $1.5M | 0.7% |
| 10 | Dtp-hepb-hib (pentavalent Vaccine) Fullyliquid Combination Vaccine 10dose (5ml)… | $1.5M | 0.7% |
| 11 | Isotroin 10 MG Capsules (isotretinoin) (inv.qty.33165 Pack 3x10s=994950 ) | $1.4M | 0.7% |
| 12 | Dtp-hepb-hib (pentavalent Vaccine) Fullyliquid Combination Vaccine 10 Dose (5ml… | $1.1M | 0.6% |
| 13 | Gepdil Tablets 4mg Cont As Per Inv | $1.0M | 0.5% |
| 14 | Other Single Vaccine-viral Vaccine | $803.1K | 0.4% |
| 15 | Bactoclav 625 (amoxicillin 500mg + Potassium Clavulanate 125mg BP Tablets)(2x10… | $787.0K | 0.4% |
India exports 19+ pharmaceutical formulations to Iraq with a combined trade value of $194.6M. Key products include Sitagliptin ($4.8M), Levofloxacin ($3.7M), Vaccine ($803K), Amoxicillin ($787K), Ceftriaxone ($764K). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 13,515 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Iraq Trade Routes
These are the top pharmaceutical products exported from India to Iraq, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Iraq buyers, regulatory requirements, and logistics for that specific product corridor. Products include Sitagliptin ($4.8M), Levofloxacin ($3.7M), Vaccine ($803K), Amoxicillin ($787K), Ceftriaxone ($764K) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Iraq
260 Indian pharmaceutical companies export finished formulations to Iraq. Leading exporters include Micro Labs Limited, Ajanta Pharma Limited, Synergy Care, Zypher Lifesciences Private Limited. The top exporter accounts for 22.3% of total India–Iraq pharma exports. Source: Indian Customs (DGFT).
Top Iraq Buyers from India
379 companies in Iraq import pharmaceutical formulations from India. Top buyers include Al-kawkab Scientific Bureau, Hawkary Pharmaceutical Company#, Mental Group Gomrok Mosavirligi, Yilmaz Transport Ulus.nak.petrol. The largest buyer accounts for 11.1% of India–Iraq pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Iraq Pharmaceutical Shipments
Indian Export Ports
Iraq Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Iraq Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The India-Iraq pharmaceutical trade corridor has experienced significant growth over the past decade. In 2014, Iraq imported pharmaceutical products valued at $803.91 million, with India contributing $10.1 million to this total. By 2024, India's pharmaceutical exports to Iraq had increased to $220.11 million, marking a substantial rise in bilateral trade. This growth trajectory reflects India's expanding role as a key supplier of pharmaceutical products to Iraq, driven by the quality and affordability of its offerings.
Key milestones in this evolution include the diversification of India's pharmaceutical exports to Iraq. In 2024, the top ten products exported encompassed a range of formulations, including HIMOX-500 MG INJECTION, OLAN-10 (Olanzapine 10mg Tablets), and AZILIDE Dry Syrup (Azithromycin for Oral Suspension), among others. This diversification indicates a deepening trust in Indian pharmaceutical products and a broadening of Iraq's healthcare needs being met by Indian exporters.
2India's Market Position
India's pharmaceutical exports to Iraq have grown from $10.1 million in 2014 to $220.11 million in 2024, indicating a substantial increase in market share. While specific data on Iraq's total pharmaceutical imports is not available, this growth suggests that India has become a significant supplier in the Iraqi pharmaceutical market. The strategic importance of this corridor is underscored by the presence of major Indian exporters such as Micro Labs Limited, Ajanta Pharma Limited, and Synergy Care, which have collectively contributed to the expansion of India's footprint in Iraq.
3Recent Developments
Between 2024 and 2026, the India-Iraq pharmaceutical trade corridor has been influenced by several developments. Notably, in February 2026, the United States and India announced an Interim Agreement framework aimed at enhancing bilateral trade, including tariff relief on medicines. While this agreement primarily focuses on U.S.-India trade, it may indirectly impact the India-Iraq corridor by influencing global pharmaceutical trade dynamics and potentially affecting the competitiveness of Indian pharmaceutical exports. Additionally, the Indian pharmaceutical industry has been targeting double-digit export growth by 2026-27, with discussions focusing on accelerating exports to various markets, including Iraq. These developments indicate a continued emphasis on strengthening India's pharmaceutical export capabilities and market presence in Iraq.
Iraq Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to export finished formulations to Iraq must navigate a structured registration process overseen by Iraq's Ministry of Health (MOH). The process involves submitting a comprehensive dossier that includes:
- Certificate of Pharmaceutical Product (CPP): Verifies the product's approval in the country of origin.
- Product Characteristics: Detailed information on the product's composition and intended use.
- Analytical Methods: Protocols for testing the product's quality.
- Stability Studies: Data demonstrating the product's shelf-life under various conditions.
- Price Certificate: Indicates the product's pricing in the country of origin.
- Samples: Physical samples of the product for evaluation.
The MOH has implemented a fast-track registration pathway to expedite the approval of urgently needed medications. This pathway allows products with approval from credible health organizations (e.g., FDA, EMA) to receive temporary registration and import licenses within one month. Conversely, the regular registration process typically takes about one year to process all required documents. (emro.who.int)
For Indian generic drugs, bioequivalence studies are generally not required for registration in Iraq. However, the MOH may prioritize the registration of new chemical entities (NCEs) to meet specific therapeutic needs.
2GMP & Facility Requirements
Iraq mandates that foreign pharmaceutical manufacturers adhere to Good Manufacturing Practices (GMP) to ensure product quality and safety. Manufacturing facilities must be inspected and approved by the MOH to confirm compliance with these standards. The MOH reserves the right to conduct inspections to ensure GMP compliance if deemed necessary.
While Iraq does not have a mutual recognition agreement with India regarding GMP certifications, Indian manufacturers can obtain GMP certification from recognized international bodies, such as the World Health Organization (WHO), to demonstrate compliance with global standards. This certification can facilitate the registration process in Iraq.
3Import Documentation
To import pharmaceutical products into Iraq, companies must secure an import license from the MOH. Required documentation includes:
- Certificate of Pharmaceutical Product (CPP): Confirms the product's approval in the country of origin.
- Certificate of Analysis (CoA): Provides detailed testing results to verify product quality.
- GMP Certificate: Demonstrates that the manufacturing facility complies with GMP standards.
Customs procedures for pharmaceutical imports into Iraq involve submitting these documents to the MOH for approval. Once approved, the products can be cleared through customs and distributed within the country.
Product Categories & Therapeutic Trends — India to Iraq
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Iraq are predominantly concentrated in therapeutic areas addressing infectious diseases, mental health, and chronic conditions. The leading products include HIMOX-500 MG INJECTION (13.7% of total exports), OLAN-10 (OLANZAPINE 10MG TABLETS) (10.7%), and AZILIDE DRY SYRUP (AZITHROMYCIN FOR ORAL USE) (9.1%). These products align with Iraq's healthcare needs, particularly in combating infections and managing mental health disorders.
The prevalence of infectious diseases in Iraq, coupled with a rising incidence of mental health issues, underscores the demand for such medications. The dominance of these therapeutic categories reflects India's capacity to meet Iraq's urgent healthcare requirements through effective and accessible pharmaceutical solutions.
2Emerging Opportunities
The Iraqi pharmaceutical market is witnessing a shift towards chronic disease management, creating opportunities for Indian exporters to introduce generics and biosimilars. The market is projected to reach US$2.84 billion by 2031, with a compound annual growth rate (CAGR) of 7.1% from 2025 to 2031. This growth is driven by the increasing prevalence of chronic diseases and the demand for cost-effective treatments.
Additionally, the global pharmaceutical market is projected to reach US$2.2 trillion by 2029, with a CAGR of 5.5% from 2024 to 2029. This expansion presents Indian pharmaceutical companies with opportunities to introduce biosimilars and generics, particularly as patents for key medications expire, creating a demand for affordable alternatives.
3Demand Drivers
Iraq's healthcare system faces challenges, including inadequate funding, shortages of essential medicines, and a reliance on imports. The country's healthcare spending per capita was US$254.62 in 2022, a 1.2% increase from 2021. Despite this increase, healthcare spending as a percentage of GDP was 4.29% in 2022, below the global average of 6.74%.
The rising prevalence of non-communicable diseases (NCDs), such as cardiovascular diseases and diabetes, contributes to the growing demand for pharmaceutical imports. NCDs accounted for an estimated 67% of all mortality in Iraq in 2019. This trend, combined with an aging population and increasing healthcare awareness, drives the need for diverse and affordable pharmaceutical products from countries like India.
Trade Policy & Tariff Intelligence — India and Iraq
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Iraq's tariff structure for pharmaceutical formulations, classified under HS codes 3003 and 3004, is not publicly detailed. The Most-Favored-Nation (MFN) tariff rates for these products are not readily accessible in public sources. Typically, MFN rates are applied to imports from countries without preferential trade agreements. However, without specific data, it's challenging to determine the exact duty rates applicable to Indian pharmaceutical exports to Iraq.
Regarding preferential duty rates, there is no evidence of a Free Trade Agreement (FTA) or any bilateral trade agreement between India and Iraq that would grant preferential tariffs on pharmaceutical products. Consequently, Indian pharmaceutical exports to Iraq are likely subject to standard MFN tariff rates.
2Trade Agreements
As of March 2026, there is no Free Trade Agreement (FTA) or bilateral trade agreement between India and Iraq that specifically addresses pharmaceutical products. This absence means that Indian pharmaceutical exports to Iraq are not eligible for preferential tariff rates and are instead subject to standard Most-Favored-Nation (MFN) tariffs. The lack of such agreements may impact the competitiveness of Indian pharmaceutical products in the Iraqi market compared to those from countries with preferential access.
3IP & Patent Landscape
Iraq's intellectual property (IP) and patent laws are not extensively documented in public sources. However, in the absence of specific data, it is reasonable to assume that Iraq's IP framework may not be as robust as those in countries with well-established pharmaceutical industries. This could potentially affect the protection of patents and data exclusivity for pharmaceutical products, including generic drugs. Consequently, Indian generic pharmaceutical companies may face challenges related to IP protection when exporting to Iraq. Additionally, the lack of comprehensive IP laws could lead to issues such as compulsory licensing or inadequate enforcement of patent rights, further complicating market entry for Indian pharmaceutical exporters.
Supply Chain & Logistics — India to Iraq Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to Iraq primarily utilize both sea and air freight, each offering distinct advantages. Sea freight is commonly employed for bulk shipments, while air freight is preferred for urgent deliveries.
Sea Freight: The journey typically involves transporting goods from major Indian ports such as Nhava Sheva (Jawaharlal Nehru Port) to Umm Qasr Port in Iraq. The transit time for sea freight from Nhava Sheva to Umm Qasr is approximately 2 days.
Air Freight: Air shipments depart from international airports in India, including Indira Gandhi International Airport (DEL), Chhatrapati Shivaji Maharaj International Airport (BOM), Chennai International Airport (MAA), and Kempegowda International Airport (BLR), arriving at Erbil International Airport (EBL) in Iraq.
Route Disruptions: The Red Sea, a critical maritime route connecting the Mediterranean Sea to the Indian Ocean, has experienced disruptions due to geopolitical tensions and piracy. Such disruptions can lead to increased transit times and costs for sea freight. Air freight, while generally unaffected by maritime issues, may face delays due to airspace restrictions or security concerns.
2Port Infrastructure
In India, key export ports handling pharmaceutical shipments include Nhava Sheva (Jawaharlal Nehru Port), Kamarajar Port, and the ICD at Sahnewal Ludhiana. Nhava Sheva, located in Maharashtra, is the largest container port in India and offers extensive facilities for handling pharmaceutical exports. Kamarajar Port, situated in Tamil Nadu, and Sahnewal Ludhiana ICD in Punjab, also serve as significant export hubs.
In Iraq, Umm Qasr Port is the primary import port for pharmaceutical products. Located in the Basra Governorate, Umm Qasr is Iraq's main seaport, handling a substantial volume of imports, including pharmaceuticals.
3Cold Chain & Compliance
Maintaining the cold chain is critical for pharmaceutical shipments, especially for temperature-sensitive formulations. The Indian pharmaceutical industry adheres to Good Distribution Practice (GDP) guidelines, ensuring that products are stored and transported within specified temperature ranges to maintain efficacy. Packaging solutions such as insulated boxes, gel packs, dry ice, and phase-change materials are commonly used to maintain the required temperature during transit.
In Iraq, the Ministry of Health has implemented stringent cold chain requirements for pharmaceutical distributors and logistics providers. These regulations mandate continuous digital temperature monitoring with automated data logging at 5-minute intervals, with data retention for 3 years. Additionally, pharmaceutical storage warehouses must undergo annual inspections, maintain backup power systems capable of 72-hour uninterrupted operation, and have calibrated temperature sensors with annual calibration certificates.
Compliance with these regulations is essential to ensure the safety and efficacy of pharmaceutical products throughout the supply chain.
Market Opportunity Assessment — Iraq for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Iraq's pharmaceutical market has experienced significant growth in recent years. The market is projected to expand at a Compound Annual Growth Rate (CAGR) of 7.2% from 2026 to 2032, indicating a robust upward trajectory. This growth is driven by factors such as increasing rates of chronic illnesses, the expansion of private hospitals, and a shift towards branded generics. The market is also transitioning from heavy import dependency towards gradual localization and structured procurement-led growth.
The share of generic drugs in Iraq's pharmaceutical market is substantial, with a growing preference for cost-effective treatment options. The global generic drugs market is projected to reach $711.1 billion by 2035, growing at a CAGR of 8.4% during the forecast period (2026-2035). This global trend is mirrored in Iraq, where the demand for generics is increasing due to their affordability and effectiveness.
2Healthcare System
Iraq's healthcare system has undergone significant reforms in recent years. In 2020, the Iraqi parliament ratified the National Health Insurance Law No. 22, aiming to introduce universal health insurance coverage with specific care access benefits. This law stipulates that insured beneficiaries contribute towards their healthcare insurance plans with affordable fees, providing subsidized coverage for healthcare expenses, including necessary medications and certain surgical procedures in both public and private hospitals.
The Ministry of Health in Iraq is responsible for providing health and medical services to every Iraqi citizen, both in normal times and during emergencies. The ministry's mission includes developing organizational structures that facilitate sustainability and decentralization, delivering and developing policies, strategies, regulations, laws, and frameworks to improve and reform the healthcare system, and establishing a healthy working environment that encourages continuous professional development.
3Opportunity for Indian Exporters
The Iraqi pharmaceutical market presents significant opportunities for Indian exporters, particularly in the generic drug segment. With a substantial portion of the market favoring generics due to their cost-effectiveness, Indian pharmaceutical companies can leverage their expertise in producing high-quality generics to meet the growing demand in Iraq. The recent implementation of the National Health Insurance Law in 2020 is expected to further drive the demand for pharmaceuticals, as more citizens gain access to subsidized healthcare services. This policy shift is likely to increase the volume of pharmaceutical imports, creating a favorable environment for Indian exporters to expand their market share in Iraq.
Competitive Landscape — India vs Other Pharmaceutical Suppliers to Iraq
Competing origins, India's edge, challenges and threats
1Competing Origins
Iraq's pharmaceutical market is supplied by a diverse array of international and domestic manufacturers. China and European Union (EU) countries are significant contributors, offering a range of generic and branded medications. Domestic production in Iraq has been expanding, with companies like Awamedica and Sanofi Iraq increasing their manufacturing capacities to meet local demand. Additionally, other emerging markets, including the United States and Japan, export pharmaceuticals to Iraq, often through partnerships with local distributors. However, precise market share data for these suppliers is limited.
2India's Competitive Edge
India holds a prominent position in Iraq's pharmaceutical sector, exporting a diverse portfolio of finished pharmaceutical formulations. In 2024, India's pharmaceutical exports to Iraq were valued at $220.11 million, reflecting a substantial contribution to Iraq's healthcare needs. Indian pharmaceutical companies, such as Micro Labs Limited and Ajanta Pharma Limited, have established a strong presence in the Iraqi market, offering a wide range of products. India's competitive advantages include cost-effectiveness, adherence to WHO-GMP standards, a large English-speaking workforce, and a broad product portfolio encompassing various therapeutic segments.
3Challenges & Threats
Indian pharmaceutical exporters face several challenges in the Iraqi market. Regulatory complexities, including compliance with the National Drug Regulatory Authority's standards, can pose obstacles. Competition from Chinese manufacturers, who often offer lower-priced alternatives, intensifies market pressures. Additionally, perceptions regarding the quality of Indian pharmaceuticals may affect their acceptance among Iraqi healthcare providers and patients. Addressing these challenges requires strategic partnerships, robust quality assurance measures, and effective market penetration strategies.
FAQ — India to Iraq Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Iraq?
India exported pharmaceuticals worth $194.6M to Iraq across 13,515 verified shipments.
Who are the top Indian pharmaceutical exporters to Iraq?
1. MICRO LABS LIMITED — $43.3M. 2. AJANTA PHARMA LIMITED — $37.0M. 3. SYNERGY CARE — $13.7M. Total: 260 suppliers.
Which companies in Iraq import pharmaceuticals from India?
1. AL-KAWKAB SCIENTIFIC BUREAU — $21.6M. 2. HAWKARY PHARMACEUTICAL COMPANY# — $20.5M. 3. MENTAL GROUP GOMROK MOSAVIRLIGI — $13.7M. 379 buyers total.
What pharmaceutical products does India export most to Iraq?
1. Himox-500 MG Injection 1x500mg. Batch No ($13.7M, 7.1%); 2. Olan-10 (2x10 ($10.7M, 5.5%); 3. Azilide Dry Syrup (azithromycin for Oral ($9.1M, 4.7%); 4. Sitarux-m (sitagliptin Phosphate & Metfo ($4.8M, 2.4%); 5. Levorux (levofloxacin Infusion 500mg/100 ($3.7M, 1.9%)
Which ports handle pharmaceutical shipments from India to Iraq?
Export: KAMARAJAR PORT, GRFL SAHNEWAL LUDHIANA ICD, NHAVA SHEVA SEA (INNSA1), JNPT/ NHAVA SHEVA SEA, SAHAR AIR. Import: Mersin, UMM QASR, Umm Qasr, Iskenderun, Baghdad.
Why does Iraq import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $194.6M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Iraq?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Iraq pharmaceutical trade?
$14.4K per consignment across 13,515 shipments.
How many Indian pharmaceutical companies export to Iraq?
260 Indian companies. Largest: MICRO LABS LIMITED with $43.3M.
How can I find verified Indian pharmaceutical suppliers for Iraq?
TransData Nexus covers 260 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Iraq Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 13,515 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Iraq identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 13,515 verified shipments from 260 Indian exporters to 379 Iraq buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
260 Exporters
379 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists