Maintaining the integrity of temperature-sensitive pharmaceutical products is paramount throughout the supply chain.
Temperature-Controlled Logistics: Pharmaceuticals, especially biologics and certain medications, require strict temperature control to preserve their efficacy. Typically, these products must be maintained within a temperature range of 2°C to 8°C during transit. To achieve this, insulated packaging solutions, such as thermal boxes equipped with phase-change materials (PCMs), are employed to regulate temperature fluctuations.
Good Distribution Practice (GDP) Compliance: Adherence to GDP guidelines is essential to ensure the quality and safety of pharmaceutical products during distribution. These guidelines emphasize the importance of temperature control, qualified equipment, continuous monitoring, and comprehensive documentation. Regular audits and staff training are integral to maintaining compliance and ensuring the integrity of the cold chain.
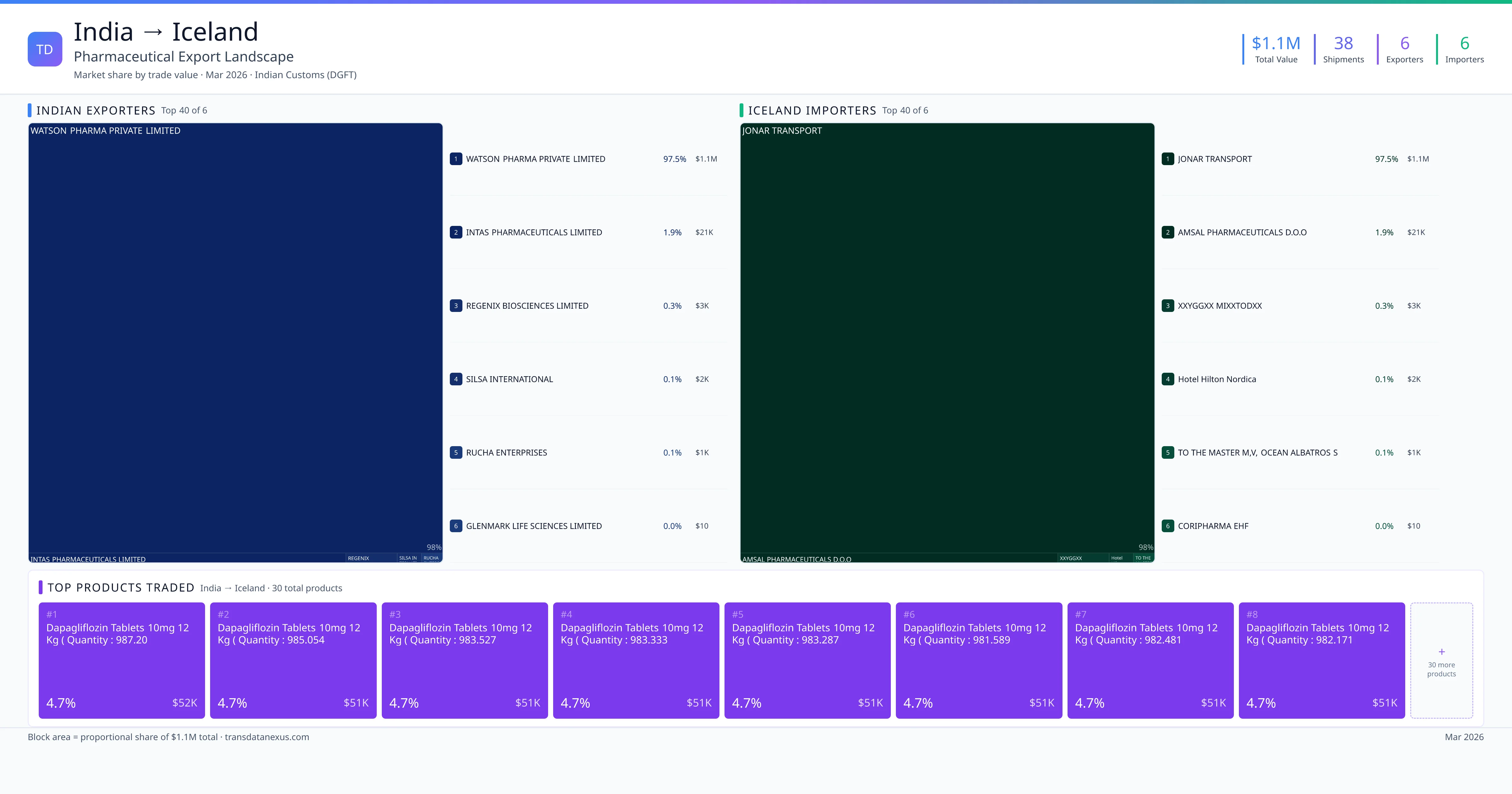
By meticulously managing shipping routes, port operations, and cold chain logistics, pharmaceutical shipments from India to Iceland can be conducted efficiently, ensuring the delivery of safe and effective products to the Icelandic market.