India to Guinea Pharmaceutical Export
Bilateral Trade Intelligence · $40.3M Total Trade · 124 Exporters · 139 Buyers · DGFT Verified · Updated March 2026
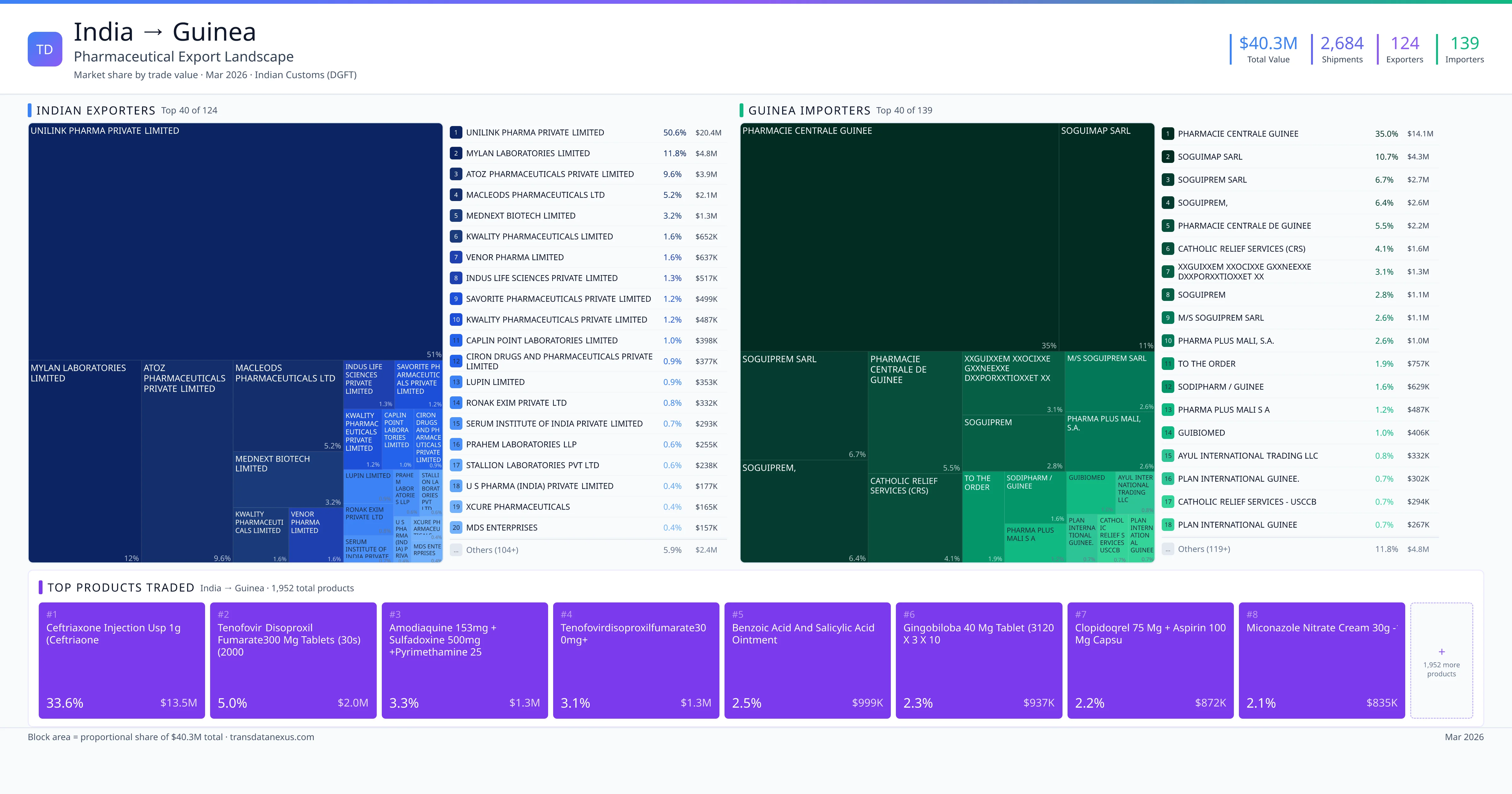
India exported $40.3M worth of pharmaceutical formulations to Guinea across 2,684 verified shipments, sourced from 124 Indian exporters supplying 139 Guinea buyers. The top exporters are UNILINK PHARMA PRIVATE LIMITED ($20.4M) and MYLAN LABORATORIES LIMITED ($4.8M). The leading products are Ceftriaxone ($13.5M) and Tenofovir ($2.0M). Average shipment value: $15.0K.
Top Pharmaceutical Formulations — India to Guinea
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Ceftriaxone Injection USP 1g (ceftriaone | $13.5M | 33.6% |
| 2 | Tenofovir Disoproxil Fumarate300 MG Tablets (30s) (20000packs X 30s=600000 ) | $2.0M | 5.0% |
| 3 | Amodiaquine 153mg + Sulfadoxine 500mg +pyrimethamine 25mg Dispersible Tablets (… | $1.3M | 3.3% |
| 4 | Tenofovirdisoproxilfumarate300mg+ | $1.3M | 3.1% |
| 5 | Benzoic Acid and Salicylic Acid Ointment | $998.8K | 2.5% |
| 6 | Gingobiloba 40 MG Tablet (3120 X 3 X 10 | $937.5K | 2.3% |
| 7 | Clopidoqrel 75 MG + Aspirin 100 MG Capsu | $872.1K | 2.2% |
| 8 | Miconazole Nitrate Cream 30g -19910 | $835.4K | 2.1% |
| 9 | Primaderm Cream - 9390 | $629.1K | 1.6% |
| 10 | Spironolactone 50 MG + Hydrochlorthiazid | $543.3K | 1.3% |
| 11 | Pharmaceuticals Brand. Verket Injection. Ketam | $458.2K | 1.1% |
| 12 | Artesunate Injection 60mg 7500 Aihi -inv | $414.6K | 1.0% |
| 13 | Silden -50 Sildenafil Citiate BP Eq to S | $412.6K | 1.0% |
| 14 | Multivitamin Syrup -4895 | $410.5K | 1.0% |
| 15 | Amoxicillin Capsules BP 500mg-5536000 | $357.2K | 0.9% |
India exports 20+ pharmaceutical formulations to Guinea with a combined trade value of $40.3M. Key products include Ceftriaxone ($13.5M), Tenofovir ($2.0M), Sulfadoxine ($1.3M), Aspirin ($872K), Miconazole ($835K). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 2,684 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Guinea Trade Routes
These are the top pharmaceutical products exported from India to Guinea, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Guinea buyers, regulatory requirements, and logistics for that specific product corridor. Products include Ceftriaxone ($13.5M), Tenofovir ($2.0M), Sulfadoxine ($1.3M), Aspirin ($872K), Miconazole ($835K) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Guinea
124 Indian pharmaceutical companies export finished formulations to Guinea. Leading exporters include Unilink Pharma Private Limited, Mylan Laboratories Limited, Atoz Pharmaceuticals Private Limited, Macleods Pharmaceuticals Ltd. The top exporter accounts for 50.6% of total India–Guinea pharma exports. Source: Indian Customs (DGFT).
Top Guinea Buyers from India
139 companies in Guinea import pharmaceutical formulations from India. Top buyers include Pharmacie Centrale Guinee, Soguimap Sarl, Soguiprem Sarl, Soguiprem,. The largest buyer accounts for 35.0% of India–Guinea pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Guinea Pharmaceutical Shipments
Indian Export Ports
Guinea Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Guinea Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and Guinea has experienced significant growth over the past decade. In 2017, Guinea imported pharmaceutical products worth $199.24 million, with $55.08 million sourced from India. By 2024, India's pharmaceutical exports to Guinea had increased to $17.04 million, contributing to a total bilateral trade of $812.73 million. This upward trajectory underscores the strengthening economic ties between the two nations.
Key milestones in this bilateral relationship include the signing of agreements in 2014 covering areas such as Foreign Office Consultations (FOC), agriculture, and health, followed by renewable energy in 2019. These agreements have facilitated increased cooperation and trade, particularly in the pharmaceutical sector.
2India's Market Position
India's pharmaceutical exports to Guinea have been a significant component of the country's overall export portfolio. In the fiscal year 2024-25, India's pharmaceutical exports reached $30.47 billion, marking a 9.4% increase from the previous year. While specific data on Guinea's total pharmaceutical imports is limited, India's consistent export growth indicates a strengthening market position in Guinea. The strategic importance of this corridor is highlighted by the presence of major Indian pharmaceutical exporters, such as Unilink Pharma Private Limited, Mylan Laboratories Limited, and Atoz Pharmaceuticals Private Limited, which have collectively contributed to the growth of this trade relationship.
3Recent Developments
Between 2024 and 2026, several developments have influenced the India-Guinea pharmaceutical trade corridor. In March 2024, India signed the Trade and Economic Partnership Agreement with the European Free Trade Association (EFTA), aiming to enhance trade relations. While this agreement primarily targets EFTA member states, it may indirectly affect India's pharmaceutical exports to other regions, including Guinea. Additionally, in December 2025, India's pharmaceutical exports grew by 9.4% over the previous year, reflecting the sector's robust performance. These developments suggest a positive outlook for the India-Guinea pharmaceutical trade corridor, with potential for continued growth and deeper economic integration.
Guinea Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
To export finished pharmaceutical formulations to Guinea, Indian pharmaceutical companies must navigate a structured registration process overseen by Guinea's National Directorate of Pharmacies and Medicines (DNPM). This process ensures that all health products meet the required quality standards before entering the market.
Dossier Format Requirements
The registration dossier must adhere to the Common Technical Document (CTD) format, which is internationally recognized and facilitates the evaluation process. The CTD is organized into five modules:
- Module 1: Administrative and prescribing information
- Module 2: Summaries of quality, non-clinical, and clinical data
- Module 3: Quality data
- Module 4: Non-clinical study reports
- Module 5: Clinical study reports
This structured approach ensures that all necessary information is presented comprehensively, aiding in the efficient assessment of the product's safety, efficacy, and quality.
Approval Timeline and Fees
While specific timelines and fees for the registration process in Guinea are not explicitly detailed in the available sources, it is common for such processes to involve several months of evaluation. Companies should anticipate potential delays and plan accordingly. Engaging with local regulatory consultants or the DNPM directly can provide more precise information regarding current timelines and associated costs.
2GMP & Facility Requirements
Guinea's regulatory framework emphasizes the importance of Good Manufacturing Practice (GMP) to ensure the quality and safety of pharmaceutical products. Manufacturing facilities exporting to Guinea must comply with GMP standards recognized by the World Health Organization (WHO). This includes maintaining proper documentation, conducting regular quality control tests, and ensuring that manufacturing processes meet international standards.
The DNPM is responsible for inspecting manufacturing sites to verify compliance with these standards. While specific details about the inspection process are not provided in the available sources, it is standard practice for regulatory authorities to conduct thorough evaluations of manufacturing facilities to ensure adherence to GMP.
3Import Documentation
To import pharmaceutical products into Guinea, companies must provide several key documents:
- Certificate of Pharmaceutical Product (CPP): This certificate attests to the product's registration status and compliance with Guinea's regulatory requirements.
- Certificate of Analysis (CoA): This document provides detailed information about the product's composition and quality, ensuring it meets the specified standards.
- GMP Certificate: This certificate confirms that the manufacturing facility adheres to GMP standards, ensuring the product's quality and safety.
These documents facilitate the customs clearance process and ensure that imported pharmaceutical products meet Guinea's regulatory standards.
Recent Regulatory Changes (2024-2026)
As of March 2026, there are no publicly available records of significant regulatory changes in Guinea's pharmaceutical sector between 2024 and 2026. However, regulatory environments can evolve, and it is advisable for companies to consult the DNPM or local regulatory experts for the most current information.
In summary, Indian pharmaceutical companies seeking to export finished formulations to Guinea must ensure compliance with the DNPM's registration process, adhere to GMP standards, and provide the necessary import documentation. Staying informed about regulatory developments and maintaining open communication with Guinea's regulatory authorities are essential for successful market entry.
Product Categories & Therapeutic Trends — India to Guinea
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Guinea are predominantly concentrated in the treatment of infectious diseases, particularly malaria and HIV/AIDS. The leading product, Ceftriaxone Injection USP 1G, accounts for 33.6% of the total export value, underscoring the critical need for effective antibiotics in Guinea's healthcare system. Additionally, Tenofovir Disoproxil Fumarate 300 mg Tablets, used in the management of HIV/AIDS, represent 5.0% of exports, highlighting the ongoing demand for antiretroviral therapies. This focus aligns with Guinea's high disease burden, where malaria, respiratory infections, and diarrheal diseases are the leading causes of morbidity, collectively accounting for 70% of the country's non-fatal disease burden.
The prominence of these therapeutic categories reflects Guinea's pressing healthcare challenges. The National Directorate of Pharmacies and Medicines (DNPM) has updated its pharmaceutical regulatory law to enhance the quality assurance and surveillance of medicines, aiming to address the prevalence of substandard and falsified drugs in the market. This regulatory strengthening is crucial for ensuring the efficacy and safety of imported pharmaceuticals, particularly in the treatment of widespread infectious diseases.
2Emerging Opportunities
The evolving pharmaceutical landscape in Guinea presents several opportunities for Indian exporters. The country's updated pharmaceutical regulatory law, signed into effect in July 2018, has enhanced the quality assurance and surveillance of medicines, aiming to eliminate substandard and falsified drugs from the market. This regulatory strengthening is crucial for ensuring the efficacy and safety of imported pharmaceuticals, particularly in the treatment of widespread infectious diseases.
Additionally, Guinea's growing demand for essential medicines, driven by its high disease burden, creates a favorable environment for Indian pharmaceutical companies to expand their presence. The government's efforts to improve healthcare infrastructure and regulatory frameworks further support the entry of quality-assured generic medicines into the market.
3Demand Drivers
Guinea's substantial disease burden, characterized by high rates of malaria, respiratory infections, and diarrheal diseases, significantly drives the demand for pharmaceuticals. The country's morbidity rate was 395 per 1,000 population in 2008, with malaria being the leading cause of non-fatal diseases. This high prevalence necessitates a continuous supply of effective medications to manage and treat these conditions.
Furthermore, Guinea's limited local pharmaceutical manufacturing capacity, coupled with a growing population and increasing healthcare needs, underscores the reliance on imports to meet the demand for essential medicines. The government's initiatives to strengthen the regulatory framework and improve healthcare infrastructure are expected to enhance the accessibility and affordability of imported pharmaceuticals, creating a conducive environment for Indian exporters to meet Guinea's healthcare demands.
Trade Policy & Tariff Intelligence — India and Guinea
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Guinea's tariff structure for pharmaceutical products, particularly those classified under HS codes 3003 and 3004, is not readily available in the provided sources. Typically, such information is detailed in Guinea's national customs tariff schedule or through official communications from Guinea's customs authority. For precise and up-to-date tariff rates, it is advisable to consult Guinea's customs department or official trade publications.
Regarding preferential duty rates, Guinea's participation in regional trade agreements, such as the Economic Community of West African States (ECOWAS), may influence tariff structures. However, specific preferential rates applicable to pharmaceutical imports from India under these agreements are not specified in the available data. Engaging with Guinea's customs authority or trade ministry would provide more clarity on any preferential duty rates or exemptions that may apply.
2Trade Agreements
As of March 2026, there is no publicly available information indicating a Free Trade Agreement (FTA) between India and Guinea. The bilateral trade volume of $40.3 million USD, encompassing 2,684 shipments and involving 124 Indian exporters and 139 Guinea buyers, suggests a robust trade relationship. However, the absence of an FTA may result in the application of standard Most-Favored-Nation (MFN) tariff rates on pharmaceutical imports. To explore potential trade agreements or negotiations that could impact pharmaceutical trade, it would be prudent to consult official trade bodies or diplomatic channels between India and Guinea.
3IP & Patent Landscape
Guinea's intellectual property (IP) framework, particularly concerning pharmaceutical patents, is not extensively detailed in the available sources. However, the National Drug Regulatory Authority (WHO-GMP) in Guinea plays a pivotal role in regulating pharmaceutical products, ensuring they meet international standards. For Indian generic drug manufacturers, understanding Guinea's patent laws, data exclusivity periods, and compulsory licensing provisions is crucial to navigate the market effectively. Engaging with Guinea's IP office or consulting with legal experts familiar with Guinean IP law would provide comprehensive insights into the patent landscape affecting generic drug imports.
FAQ — India to Guinea Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Guinea?
India exported pharmaceuticals worth $40.3M to Guinea across 2,684 verified shipments.
Who are the top Indian pharmaceutical exporters to Guinea?
1. UNILINK PHARMA PRIVATE LIMITED — $20.4M. 2. MYLAN LABORATORIES LIMITED — $4.8M. 3. ATOZ PHARMACEUTICALS PRIVATE LIMITED — $3.9M. Total: 124 suppliers.
Which companies in Guinea import pharmaceuticals from India?
1. PHARMACIE CENTRALE GUINEE — $14.1M. 2. SOGUIMAP SARL — $4.3M. 3. SOGUIPREM SARL — $2.7M. 139 buyers total.
What pharmaceutical products does India export most to Guinea?
1. Ceftriaxone Injection USP 1g (ceftriaone ($13.5M, 33.6%); 2. Tenofovir Disoproxil Fumarate300 MG Tablets (30s) (20000packs X 30s=600000 ) ($2.0M, 5.0%); 3. Amodiaquine 153mg + Sulfadoxine 500mg +pyrimethamine 25mg Dispersible Tablets (… ($1.3M, 3.3%); 4. Tenofovirdisoproxilfumarate300mg+ ($1.3M, 3.1%); 5. Benzoic Acid and Salicylic Acid Ointment ($998.8K, 2.5%)
Which ports handle pharmaceutical shipments from India to Guinea?
Export: KAMARAJAR PORT, NHAVA SHEVA SEA (INNSA1), CHENNAI AIR, MUNDRA SEA, KAMARAJAR PORT (INENR1). Import: Conakry, CONAKRY, Conakry Eco, Malabo, PORT MORESBY.
Why does Guinea import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $40.3M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Guinea?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Guinea pharmaceutical trade?
$15.0K per consignment across 2,684 shipments.
How many Indian pharmaceutical companies export to Guinea?
124 Indian companies. Largest: UNILINK PHARMA PRIVATE LIMITED with $20.4M.
How can I find verified Indian pharmaceutical suppliers for Guinea?
TransData Nexus covers 124 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Guinea Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 2,684 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Guinea identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 2,684 verified shipments from 124 Indian exporters to 139 Guinea buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
124 Exporters
139 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists