India to Georgia Pharmaceutical Export
Bilateral Trade Intelligence · $78.6M Total Trade · 166 Exporters · 227 Buyers · DGFT Verified · Updated March 2026
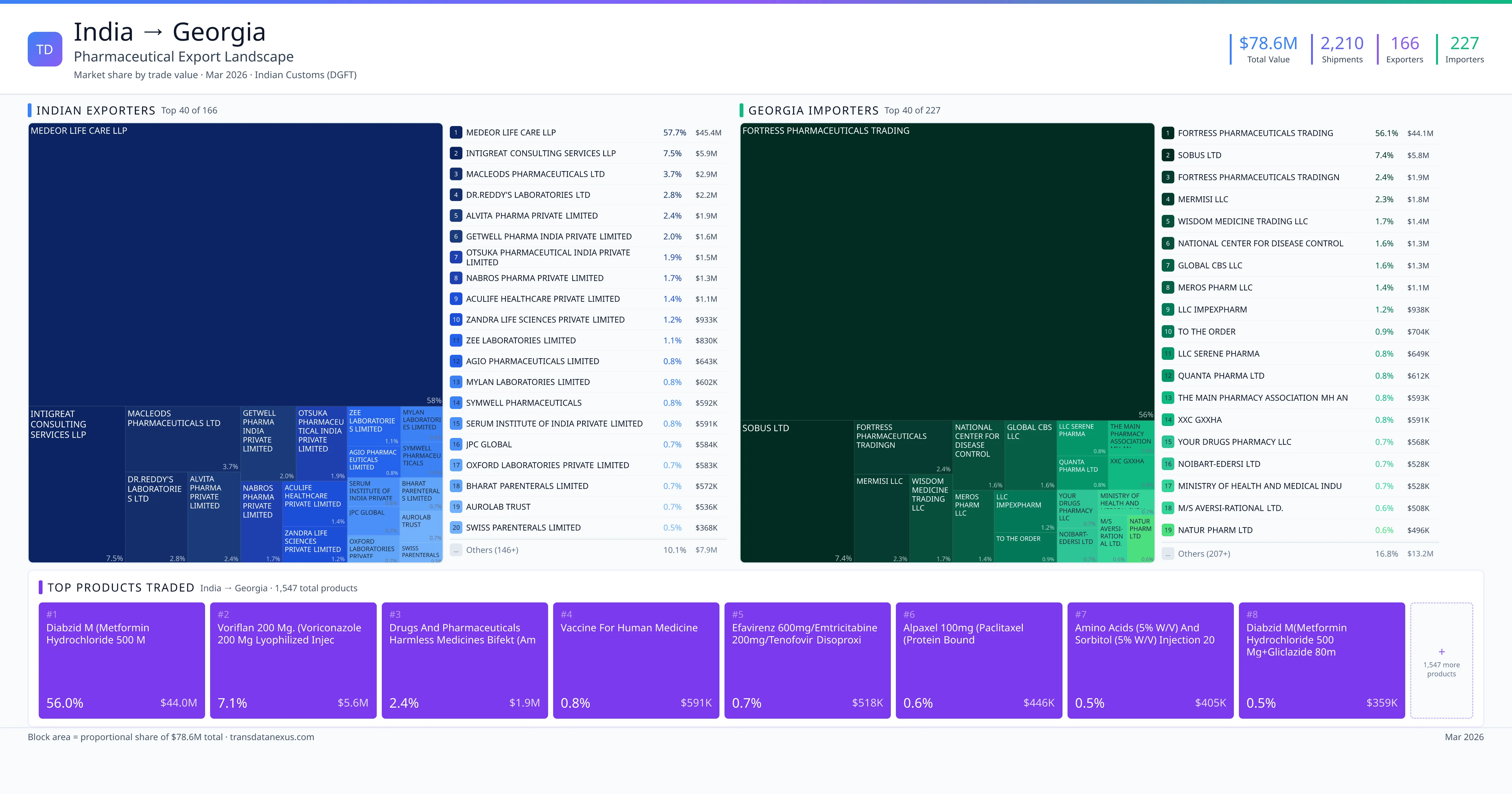
India exported $78.6M worth of pharmaceutical formulations to Georgia across 2,210 verified shipments, sourced from 166 Indian exporters supplying 227 Georgia buyers. The top exporters are MEDEOR LIFE CARE LLP ($45.4M) and INTIGREAT CONSULTING SERVICES LLP ($5.9M). The leading products are Metformin ($44.0M) and Voriconazole ($5.6M). Average shipment value: $35.6K.
Top Pharmaceutical Formulations — India to Georgia
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Diabzid M (metformin Hydrochloride 500 M | $44.0M | 56.0% |
| 2 | Voriflan 200 Mg. (voriconazole 200 MG Lyophilized Injection with 20ml Wfi (comb… | $5.6M | 7.1% |
| 3 | Drugs and Pharmaceuticals Harmless Medicines Bifekt (amlodipine and Lisinopril… | $1.9M | 2.4% |
| 4 | Vaccine for Human Medicine | $590.6K | 0.8% |
| 5 | Efavirenz 600mg/emtricitabine 200mg/tenofovir Disoproxil Fumarate 300mg Tablets… | $517.8K | 0.7% |
| 6 | Alpaxel 100mg (paclitaxel (protein Bound | $446.1K | 0.6% |
| 7 | Amino Acids (5% W/v) and Sorbitol (5% W/v) Injection 200ml Glass Bottle (celemi… | $405.4K | 0.5% |
| 8 | Diabzid M(metformin Hydrochloride 500 Mg+gliclazide 80mg Tablets) | $359.0K | 0.5% |
| 9 | Sodium Chloride Intravenous Infusion Bp(0.9% W/v) 500 ML | $317.5K | 0.4% |
| 10 | Lamivudine 300mg Tenofovir Disoproxil Fumarate 300mg Dolutegravir 50mg Tablets | $317.2K | 0.4% |
| 11 | Harmless Pharmaceutical Finished Product | $314.7K | 0.4% |
| 12 | Metronidazole Injection USP (0.5% W/v) 100ml Plastic Bottle (metrid) | $306.3K | 0.4% |
| 13 | Spasmanol Injection As Per Invoice | $273.8K | 0.3% |
| 14 | Ciprolet Tablets 500mg (n10) (156130 X 10 Pa | $260.9K | 0.3% |
| 15 | Diabzid M (metformin Hydrochloride 500 MG + Gliclazide 80 MG Tablets) | $260.4K | 0.3% |
India exports 20+ pharmaceutical formulations to Georgia with a combined trade value of $78.6M. Key products include Metformin ($44.0M), Voriconazole ($5.6M), Vaccine ($591K), Efavirenz ($518K), Paclitaxel ($446K). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 2,210 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Georgia Trade Routes
These are the top pharmaceutical products exported from India to Georgia, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Georgia buyers, regulatory requirements, and logistics for that specific product corridor. Products include Metformin ($44.0M), Voriconazole ($5.6M), Vaccine ($591K), Efavirenz ($518K), Paclitaxel ($446K) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Georgia
166 Indian pharmaceutical companies export finished formulations to Georgia. Leading exporters include Medeor Life Care Llp, Intigreat Consulting Services Llp, Macleods Pharmaceuticals Ltd, Dr.reddy's Laboratories Ltd. The top exporter accounts for 57.7% of total India–Georgia pharma exports. Source: Indian Customs (DGFT).
Top Georgia Buyers from India
227 companies in Georgia import pharmaceutical formulations from India. Top buyers include Fortress Pharmaceuticals Trading, Sobus Ltd, Fortress Pharmaceuticals Tradingn, Mermisi Llc. The largest buyer accounts for 56.1% of India–Georgia pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Georgia Pharmaceutical Shipments
Indian Export Ports
Georgia Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Georgia Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and Georgia has experienced significant growth over the past decade. In 2024, India exported pharmaceutical products valued at $24.75 million to Georgia, marking a substantial increase from previous years. This upward trajectory reflects the strengthening of bilateral trade relations and India's expanding presence in the Georgian pharmaceutical market.
Key milestones in this evolution include the establishment of direct shipping routes and the participation of Indian pharmaceutical companies in Georgian trade fairs, which have facilitated market penetration. Additionally, the recognition of India's pharmaceutical manufacturing standards by Georgian regulatory authorities has played a pivotal role in enhancing trade volumes.
2India's Market Position
India holds a significant share of Georgia's pharmaceutical imports, positioning itself as a primary supplier alongside other origins such as China and the European Union. The strategic importance of this relationship is underscored by India's ability to offer a diverse range of pharmaceutical formulations that cater to Georgia's healthcare needs. The competitive pricing and adherence to international quality standards further bolster India's position in the Georgian market.
3Recent Developments
Between 2024 and 2026, several developments have influenced the India-Georgia pharmaceutical trade. In January 2022, Georgia amended its Law on Medicines and Pharmaceutical Activities to require a certificate confirming that imported pharmaceutical products are manufactured in line with Good Manufacturing Practice (GMP) standards. (rustavi2.ge) This regulatory change has prompted Indian exporters to ensure compliance with GMP to maintain market access. Additionally, diplomatic engagements and trade agreements have been initiated to further enhance bilateral trade relations, with ongoing discussions aimed at reducing trade barriers and facilitating smoother market entry for Indian pharmaceutical products.
Georgia Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to register products in Georgia must navigate a structured process. The Georgian regulatory authority, the Regulation Agency for Medical and Pharmaceutical Activities, mandates that all pharmaceutical products be registered before importation. For products already approved by stringent regulatory authorities such as the FDA or EMA, Georgia offers a "Recognition Regime," allowing for a streamlined registration process that typically takes 7 working days. This regime is designed to expedite market entry for products with established international approvals. (legal.ge)
For other products, the "National Regime" applies, which involves a more comprehensive evaluation and can extend over several months. Regardless of the regime, a valid GMP certificate from the manufacturing site is mandatory for registration. This requirement ensures that imported pharmaceutical products meet the quality standards expected in the Georgian market. (legal.ge)
2GMP & Facility Requirements
Georgia's adherence to international standards necessitates that Indian manufacturing facilities comply with GMP and Good Distribution Practice (GDP) standards. Since 2022, compliance with these standards has been mandatory for manufacturers, importers, and distributors operating in Georgia. (legal.ge) The inspection process involves a thorough assessment of the manufacturing premises, equipment, and quality control measures to ensure adherence to GMP standards. Mutual recognition agreements between Georgia and countries with stringent regulatory frameworks, such as the EU and the USA, facilitate the acceptance of GMP certifications from these jurisdictions, streamlining the process for Indian exporters.
3Import Documentation
Importing pharmaceutical products into Georgia requires specific documentation to ensure compliance with regulatory standards. A certificate of pharmaceutical product (CPP) in the form recommended by the World Health Organization is essential. If such a certificate is unavailable, a document certifying that the pharmaceutical product has been manufactured according to GMP standards or a pharmaceutical product manufacturing license issued by an authorized body of the manufacturing country is acceptable. (wipo.int) Additionally, a standard packaging with standard labeling, or an electronic version, of the pharmaceutical product to be registered is required. In the case of registration of an imported pharmaceutical product, an authorized (attested) translation of a patient information leaflet into the Georgian language and an original patient information leaflet, under procedures established by the Ministry, must be provided. (wipo.int)
Product Categories & Therapeutic Trends — India to Georgia
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Georgia are predominantly composed of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. These products align with Georgia's healthcare needs, particularly in the management of chronic conditions such as diabetes and cardiovascular diseases. The dominance of these categories is attributed to the high prevalence of such diseases in Georgia and the efficacy of Indian formulations in addressing these health concerns.
2Emerging Opportunities
The expiration of patents for several blockbuster drugs presents opportunities for Indian exporters to introduce generic versions into the Georgian market. Additionally, the growing demand for biosimilars in Georgia offers a promising avenue for Indian pharmaceutical companies, given their expertise in biosimilar development. Exploring new therapeutic areas, such as oncology and immunology, could further diversify India's export portfolio to Georgia.
3Demand Drivers
Georgia's disease burden, characterized by a high incidence of chronic illnesses like diabetes and hypertension, drives the demand for pharmaceutical imports. The aging population further exacerbates this demand, as older individuals typically require more medical care and medications. Increased healthcare spending and government initiatives to improve healthcare infrastructure also contribute to the rising need for pharmaceutical products, creating a favorable environment for Indian exporters.
Trade Policy & Tariff Intelligence — India and Georgia
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Georgia's tariff structure for pharmaceutical products is designed to facilitate the importation of essential medicines while protecting domestic industries. Most-favored-nation (MFN) tariff rates apply to pharmaceutical imports, with preferential rates and duty exemptions available under specific trade agreements. These policies aim to balance the interests of domestic producers and international suppliers, ensuring the availability of quality medicines at competitive prices.
2Trade Agreements
Georgia has engaged in various trade agreements to enhance bilateral relations and promote economic cooperation. While there is no specific Free Trade Agreement (FTA) between India and Georgia, both countries have expressed interest in exploring such agreements to reduce trade barriers and facilitate smoother market access for pharmaceutical products. Ongoing negotiations focus on harmonizing regulatory standards and addressing tariff and non-tariff barriers to trade.
3IP & Patent Landscape
Georgia's intellectual property (IP) laws provide protection for pharmaceutical patents, data exclusivity, and compulsory licensing. These provisions impact the entry of Indian generics into the Georgian market, as patent protections may limit the ability to produce and sell generic versions of patented drugs. Understanding the nuances of Georgia's IP landscape is crucial for Indian exporters to navigate potential challenges and identify opportunities for market entry.
Supply Chain & Logistics — India to Georgia Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Major sea and air routes facilitate the transportation of pharmaceutical products from Indian ports to Georgia. Sea routes typically involve transit through the Suez Canal, with an estimated transit time of 20-25 days, depending on the port of departure in India. Air freight offers faster delivery, with transit times ranging from 5 to 7 days. However, geopolitical events, such as disruptions in the Red Sea, can impact these routes, necessitating alternative planning and potential delays.
2Port Infrastructure
In India, key export ports include Ahmedabad ICD (6.2%), Delhi Air Cargo Acc (INDEL4) (9.2%), and Nhava Sheva Sea (INNSA1) (8.7%). In Georgia, primary import ports are Poti (16.2%), Tbilisi (4.0%), and Tbilisi (51.3%). These ports are equipped to handle pharmaceutical shipments, ensuring efficient customs clearance and distribution within Georgia.
3Cold Chain & Compliance
Temperature-controlled logistics are essential for maintaining the efficacy of pharmaceutical products during transit. Compliance with Good Distribution Practice (GDP) standards is mandatory for pharmaceutical distributors in Georgia to ensure the quality of medicines during transport and storage. (legal.ge) Indian exporters must adhere to these standards, implementing robust cold chain management systems and ensuring packaging meets international compliance requirements to maintain product integrity upon arrival.
Market Opportunity Assessment — Georgia for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Georgia's pharmaceutical market has been experiencing steady growth, with an increasing reliance on imported medicines to meet domestic demand. The market's expansion is driven by factors such as an aging population, rising healthcare awareness, and government initiatives to improve healthcare infrastructure. The share of generics in the market is substantial, presenting opportunities for Indian exporters to introduce cost-effective alternatives to branded drugs.
2Healthcare System
Georgia's government healthcare programs aim to provide accessible and affordable medical services to its population. Insurance coverage is expanding, with more individuals gaining access to health insurance plans that cover a range of pharmaceutical products. The drug procurement system is centralized, with the government negotiating prices and managing the distribution of essential medicines, ensuring equitable access across the country.
3Opportunity for Indian Exporters
Indian pharmaceutical exporters have specific opportunities in Georgia, particularly in the supply of generic medications for chronic diseases, biosimil
FAQ — India to Georgia Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Georgia?
India exported pharmaceuticals worth $78.6M to Georgia across 2,210 verified shipments.
Who are the top Indian pharmaceutical exporters to Georgia?
1. MEDEOR LIFE CARE LLP — $45.4M. 2. INTIGREAT CONSULTING SERVICES LLP — $5.9M. 3. MACLEODS PHARMACEUTICALS LTD — $2.9M. Total: 166 suppliers.
Which companies in Georgia import pharmaceuticals from India?
1. FORTRESS PHARMACEUTICALS TRADING — $44.1M. 2. SOBUS LTD — $5.8M. 3. FORTRESS PHARMACEUTICALS TRADINGN — $1.9M. 227 buyers total.
What pharmaceutical products does India export most to Georgia?
1. Diabzid M (metformin Hydrochloride 500 M ($44.0M, 56.0%); 2. Voriflan 200 Mg. (voriconazole 200 MG Lyophilized Injection with 20ml Wfi (comb… ($5.6M, 7.1%); 3. Drugs and Pharmaceuticals Harmless Medicines Bifekt (amlodipine and Lisinopril… ($1.9M, 2.4%); 4. Vaccine for Human Medicine ($590.6K, 0.8%); 5. Efavirenz 600mg/emtricitabine 200mg/tenofovir Disoproxil Fumarate 300mg Tablets… ($517.8K, 0.7%)
Which ports handle pharmaceutical shipments from India to Georgia?
Export: AHEMDABAD ICD, DELHI AIR CARGO ACC (INDEL4), NHAVA SHEVA SEA (INNSA1), SAHAR AIR CARGO ACC (INBOM4), JOGBANI. Import: Poti, Tbilisi, TABILISI, POTI, TBILISI.
Why does Georgia import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $78.6M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Georgia?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Georgia pharmaceutical trade?
$35.6K per consignment across 2,210 shipments.
How many Indian pharmaceutical companies export to Georgia?
166 Indian companies. Largest: MEDEOR LIFE CARE LLP with $45.4M.
How can I find verified Indian pharmaceutical suppliers for Georgia?
TransData Nexus covers 166 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Georgia Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 2,210 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Georgia identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 2,210 verified shipments from 166 Indian exporters to 227 Georgia buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
166 Exporters
227 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists