India to Ethiopia Pharmaceutical Export
Bilateral Trade Intelligence · $277.8M Total Trade · 185 Exporters · 500 Buyers · DGFT Verified · Updated March 2026
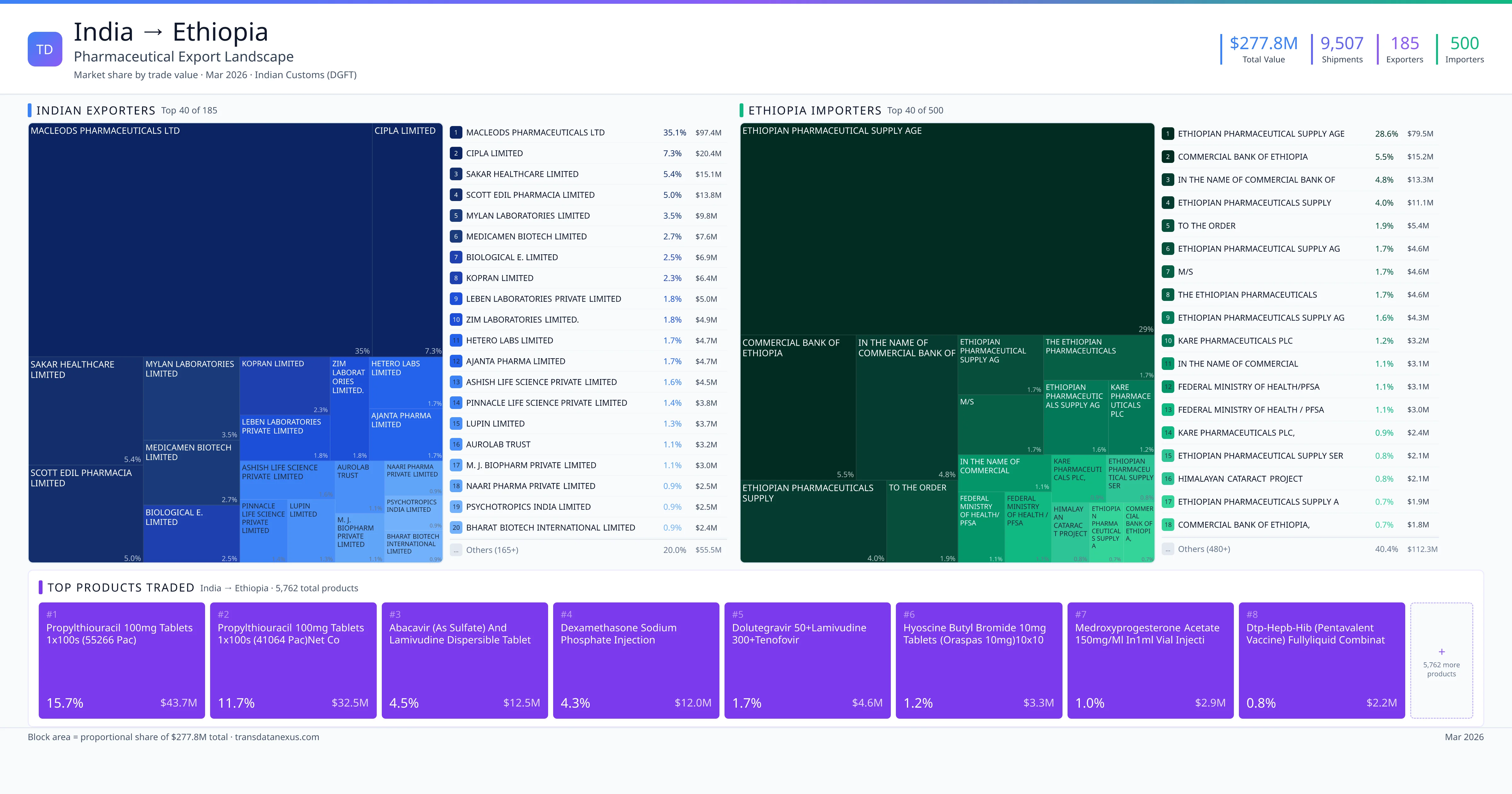
India exported $277.8M worth of pharmaceutical formulations to Ethiopia across 9,507 verified shipments, sourced from 185 Indian exporters supplying 500 Ethiopia buyers. The top exporters are MACLEODS PHARMACEUTICALS LTD ($97.4M) and CIPLA LIMITED ($20.4M). The leading products are Dexamethasone ($12.0M) and Dolutegravir ($4.6M). Average shipment value: $29.2K.
Top Pharmaceutical Formulations — India to Ethiopia
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Propylthiouracil 100mg Tablets 1x100s (55266 Pac) | $43.7M | 15.7% |
| 2 | Propylthiouracil 100mg Tablets 1x100s (41064 Pac)net Content | $32.5M | 11.7% |
| 3 | Dexamethasone Sodium Phosphate Injection | $12.0M | 4.3% |
| 4 | Abacavir (as Sulfate) and Lamivudine Dispersible Tablet 120mg/60mg (16240 Packx… | $5.2M | 1.9% |
| 5 | Abacavir (as Sulfate) and Lamivudine Dispersible Tablet 120mg/60mg (16100 Packx… | $5.2M | 1.9% |
| 6 | Dolutegravir 50+lamivudine 300+tenofovir | $4.6M | 1.7% |
| 7 | Hyoscine Butyl Bromide 10mg Tablets (oraspas 10mg)10x10s (13659 Pac)net Content | $3.3M | 1.2% |
| 8 | Medroxyprogesterone Acetate 150mg/ml In1ml Vial Injection Aqueous Suspension Co… | $2.9M | 1.0% |
| 9 | Dtp-hepb-hib (pentavalent Vaccine) Fullyliquid Combination Vaccine,single Dosev… | $2.2M | 0.8% |
| 10 | Abacavir (as Sulfate) and Lamivudine Dispersible Tablet 120mg/60mg (6230 Packx1… | $2.0M | 0.7% |
| 11 | Patient Pack Kit Rhze(rifampicin 150mg+i | $1.9M | 0.7% |
| 12 | Lumiter Ps: 30x24s Bno. Naa24440a to Naa24465a ** | $1.9M | 0.7% |
| 13 | Biopolio B1/3 1.0ml Bivalent Type 1 and | $1.9M | 0.7% |
| 14 | Aurovisc[hydroxypropyl Methylcellulose O | $1.7M | 0.6% |
| 15 | Lamivudine 300mg+tenofovir Disoproxil Fumarate 300mg+dolutegravir 50mg Tablets… | $1.5M | 0.6% |
India exports 20+ pharmaceutical formulations to Ethiopia with a combined trade value of $277.8M. Key products include Dexamethasone ($12.0M), Dolutegravir ($4.6M), Artemether ($1.9M), Tenofovir ($1.5M), Rifampicin ($1.5M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 9,507 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Ethiopia Trade Routes
These are the top pharmaceutical products exported from India to Ethiopia, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Ethiopia buyers, regulatory requirements, and logistics for that specific product corridor. Products include Dexamethasone ($12.0M), Dolutegravir ($4.6M), Artemether ($1.9M), Tenofovir ($1.5M), Rifampicin ($1.5M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Ethiopia
185 Indian pharmaceutical companies export finished formulations to Ethiopia. Leading exporters include Macleods Pharmaceuticals Ltd, Cipla Limited, Sakar Healthcare Limited, Scott Edil Pharmacia Limited. The top exporter accounts for 35.1% of total India–Ethiopia pharma exports. Source: Indian Customs (DGFT).
Top Ethiopia Buyers from India
500 companies in Ethiopia import pharmaceutical formulations from India. Top buyers include Ethiopian Pharmaceutical Supply Age, Commercial Bank Of Ethiopia, In The Name Of Commercial Bank Of, Ethiopian Pharmaceuticals Supply. The largest buyer accounts for 28.6% of India–Ethiopia pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Ethiopia Pharmaceutical Shipments
Indian Export Ports
Ethiopia Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Ethiopia Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and Ethiopia has experienced significant growth over the past decade. In 2023, Ethiopia imported pharmaceutical products valued at $244.72 million from India, marking a substantial increase from previous years. This upward trajectory underscores the strengthening bilateral trade relations and India's growing role as a key supplier of pharmaceutical products to Ethiopia.
The expansion is attributed to India's competitive pricing, diverse product offerings, and adherence to international quality standards. Ethiopia's increasing demand for quality medicines, driven by a rising population and healthcare needs, has further bolstered this trade corridor. The establishment of the Ethiopian Food and Drug Authority (EFDA) has also streamlined regulatory processes, facilitating smoother trade operations. (efda.gov.et)
2India's Market Position
India holds a dominant position in Ethiopia's pharmaceutical import market, accounting for a significant share of the total imports. This dominance is a result of India's robust pharmaceutical manufacturing capabilities, extensive product range, and cost-effectiveness. The strategic importance of this market is evident as Indian pharmaceutical exports to Africa grew by 19% in 2022-23, reflecting the continent's increasing reliance on Indian medicines.
3Recent Developments
Between 2024 and 2026, several key developments have influenced the India-Ethiopia pharmaceutical trade. In July 2025, the World Health Organization (WHO) emphasized the need for African countries, including Ethiopia, to reduce reliance on imported medicines by enhancing local production capabilities. (afro.who.int) This initiative may impact future import patterns, encouraging Ethiopia to bolster domestic pharmaceutical manufacturing. Additionally, the EFDA has been actively updating its Good Manufacturing Practices (GMP) guidelines to align with international standards, potentially affecting the regulatory landscape for pharmaceutical imports. (efda.gov.et)
Ethiopia Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to export to Ethiopia must navigate a structured registration process overseen by the EFDA. This process involves submitting a comprehensive dossier that includes product information, manufacturing details, and quality control measures. The EFDA requires adherence to the Common Technical Document (CTD) format for submissions, ensuring consistency and clarity in the evaluation process. Bioequivalence studies are mandatory for generics to demonstrate therapeutic equivalence to innovator products. The registration timeline varies but typically spans several months, depending on the completeness of the submission and the EFDA's workload.
2GMP & Facility Requirements
Ethiopia mandates that all pharmaceutical products imported into the country comply with current Good Manufacturing Practices (cGMP) as outlined by the EFDA. Indian manufacturing facilities must undergo inspections to verify adherence to these standards. The EFDA's GMP guidelines are detailed in their publication, which serves as a reference for both local and foreign manufacturers. (efda.gov.et) Mutual recognition agreements may expedite the inspection process for facilities already certified by recognized international bodies.
3Import Documentation
To import pharmaceutical products into Ethiopia, Indian exporters must provide several key documents:
- Certificate of Pharmaceutical Product (CPP): Confirms the product's registration and compliance with GMP standards.
- Certificate of Analysis (CoA): Demonstrates that the product meets specified quality standards.
- GMP Certificate: Indicates that the manufacturing facility adheres to GMP guidelines.
Additionally, importers must secure an import license and comply with customs procedures, which include submitting a commercial invoice, packing list, and other relevant documents. The EFDA's i-Import system facilitates the online submission and approval of import applications, streamlining the process for both importers and regulatory authorities. (efda.gov.et)
Product Categories & Therapeutic Trends — India to Ethiopia
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Ethiopia are predominantly in the form of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. These products align with Ethiopia's healthcare needs, addressing prevalent conditions such as infectious diseases, chronic illnesses, and maternal health. The demand for antiretroviral drugs, for instance, is significant due to the high HIV/AIDS prevalence in the region. The therapeutic areas of antithyroid medications, corticosteroids, antiretrovirals, and contraceptives are particularly prominent in the export portfolio.
2Emerging Opportunities
The global pharmaceutical industry is witnessing patent expirations, leading to the emergence of biosimilars and generic drugs. Ethiopia's growing healthcare infrastructure and increasing demand for affordable medicines present opportunities for Indian exporters to introduce these products. Additionally, expanding into new therapeutic areas such as oncology and diabetes care could cater to the evolving health needs of the Ethiopian population.
3Demand Drivers
Ethiopia's substantial disease burden, including high rates of infectious diseases like malaria and tuberculosis, drives the demand for pharmaceutical imports. The country's aging population and rising prevalence of non-communicable diseases such as diabetes and hypertension further contribute to this demand. Increased healthcare spending by the government and international donors also supports the importation of quality medicines to meet the population's needs.
Trade Policy & Tariff Intelligence — India and Ethiopia
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Ethiopia's tariff structure for pharmaceutical imports includes Most Favored Nation (MFN) rates, with potential preferential rates under trade agreements. Duty exemptions may apply to certain essential medicines to ensure affordability. Importers should consult the latest Ethiopian Customs Commission guidelines to understand applicable tariffs and exemptions.
2Trade Agreements
Ethiopia is a member of the Common Market for Eastern and Southern Africa (COMESA), which aims to promote regional integration and trade. While there is no specific Free Trade Agreement (FTA) between India and Ethiopia, the COMESA framework may offer preferential trade terms that benefit Indian pharmaceutical exporters. Ongoing negotiations and regional collaborations could further enhance trade relations in the future.
3IP & Patent Landscape
Ethiopia's intellectual property laws provide protection for pharmaceutical patents, including provisions for data exclusivity. Compulsory licensing is permitted under specific conditions, such as public health emergencies, which could impact the entry of Indian generics into the market. Indian exporters should stay informed about Ethiopia's IP regulations to navigate the market effectively.
Supply Chain & Logistics — India to Ethiopia Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to Ethiopia primarily utilize sea and air routes. Sea shipments depart from major Indian ports like Nhava Sheva and arrive at Djibouti, from where goods are transported overland to Ethiopia. Air shipments typically land at Addis Ababa Bole International Airport. Transit times vary, with sea shipments taking several weeks and air shipments approximately 3-5 days. Geopolitical factors, such as instability in the Red Sea region, can occasionally disrupt these routes.
2Port Infrastructure
In India, Nhava Sheva Sea Port and Sahar Air Cargo Terminal are key export points for pharmaceutical products. In Ethiopia, Addis Ababa Bole International Airport serves as the primary import hub for air shipments, while Djibouti Port handles a significant portion of sea freight. The efficiency of these ports is crucial for maintaining the timely delivery of pharmaceutical products.
3Cold Chain & Compliance
Maintaining the integrity of temperature-sensitive pharmaceutical products is essential. Both Indian exporters and Ethiopian importers must adhere to Good Distribution Practices (GDP) to ensure product quality. This includes using temperature-controlled logistics, proper packaging, and compliance with international standards for cold chain management. Regular audits and certifications are necessary to uphold these standards.
Market Opportunity Assessment — Ethiopia for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Ethiopia's pharmaceutical market is experiencing rapid growth, driven by an expanding population and increasing healthcare needs. The market's value is projected to continue rising, with a significant share of imports comprising generic medicines. This trend presents substantial opportunities for Indian pharmaceutical exporters to meet the demand for affordable and quality medicines.
2Healthcare System
Ethiopia's government has implemented various healthcare programs aimed at improving access to medicines and healthcare services. The establishment of the Pharmaceuticals Fund and Supply Agency (PFSA) has streamlined drug procurement and distribution. However, challenges remain in terms of infrastructure and resource allocation, which may impact the efficiency of drug delivery systems.
3Opportunity for Indian Exporters
Indian pharmaceutical exporters have opportunities to supply a wide range of products to Ethiopia, particularly in therapeutic areas such as infectious diseases, maternal health, and chronic conditions. The growing demand for generics and biosimilars aligns with India's manufacturing strengths. Collaborations with local distributors and adherence to EFDA's regulatory requirements will be key
FAQ — India to Ethiopia Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Ethiopia?
India exported pharmaceuticals worth $277.8M to Ethiopia across 9,507 verified shipments.
Who are the top Indian pharmaceutical exporters to Ethiopia?
1. MACLEODS PHARMACEUTICALS LTD — $97.4M. 2. CIPLA LIMITED — $20.4M. 3. SAKAR HEALTHCARE LIMITED — $15.1M. Total: 185 suppliers.
Which companies in Ethiopia import pharmaceuticals from India?
1. ETHIOPIAN PHARMACEUTICAL SUPPLY AGE — $79.5M. 2. COMMERCIAL BANK OF ETHIOPIA — $15.2M. 3. IN THE NAME OF COMMERCIAL BANK OF — $13.3M. 500 buyers total.
What pharmaceutical products does India export most to Ethiopia?
1. Propylthiouracil 100mg Tablets 1x100s (55266 Pac) ($43.7M, 15.7%); 2. Propylthiouracil 100mg Tablets 1x100s (41064 Pac)net Content ($32.5M, 11.7%); 3. Dexamethasone Sodium Phosphate Injection ($12.0M, 4.3%); 4. Abacavir (as Sulfate) and Lamivudine Dispersible Tablet 120mg/60mg (16240 Packx… ($5.2M, 1.9%); 5. Abacavir (as Sulfate) and Lamivudine Dispersible Tablet 120mg/60mg (16100 Packx… ($5.2M, 1.9%)
Which ports handle pharmaceutical shipments from India to Ethiopia?
Export: NHAVA SHEVA SEA, NHAVA SHEVA SEA (INNSA1), SAHAR AIR, SAHAR AIR CARGO ACC (INBOM4), AHEMDABAD ICD. Import: ADDIS ABABA, Djibouti, DJIBOUTI, MOJO ETHIOPIA, Addis Ababa.
Why does Ethiopia import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $277.8M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Ethiopia?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Ethiopia pharmaceutical trade?
$29.2K per consignment across 9,507 shipments.
How many Indian pharmaceutical companies export to Ethiopia?
185 Indian companies. Largest: MACLEODS PHARMACEUTICALS LTD with $97.4M.
How can I find verified Indian pharmaceutical suppliers for Ethiopia?
TransData Nexus covers 185 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Ethiopia Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 9,507 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Ethiopia identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 9,507 verified shipments from 185 Indian exporters to 500 Ethiopia buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
185 Exporters
500 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists