India to El Salvador Pharmaceutical Export
Bilateral Trade Intelligence · $39.9M Total Trade · 101 Exporters · 189 Buyers · DGFT Verified · Updated March 2026
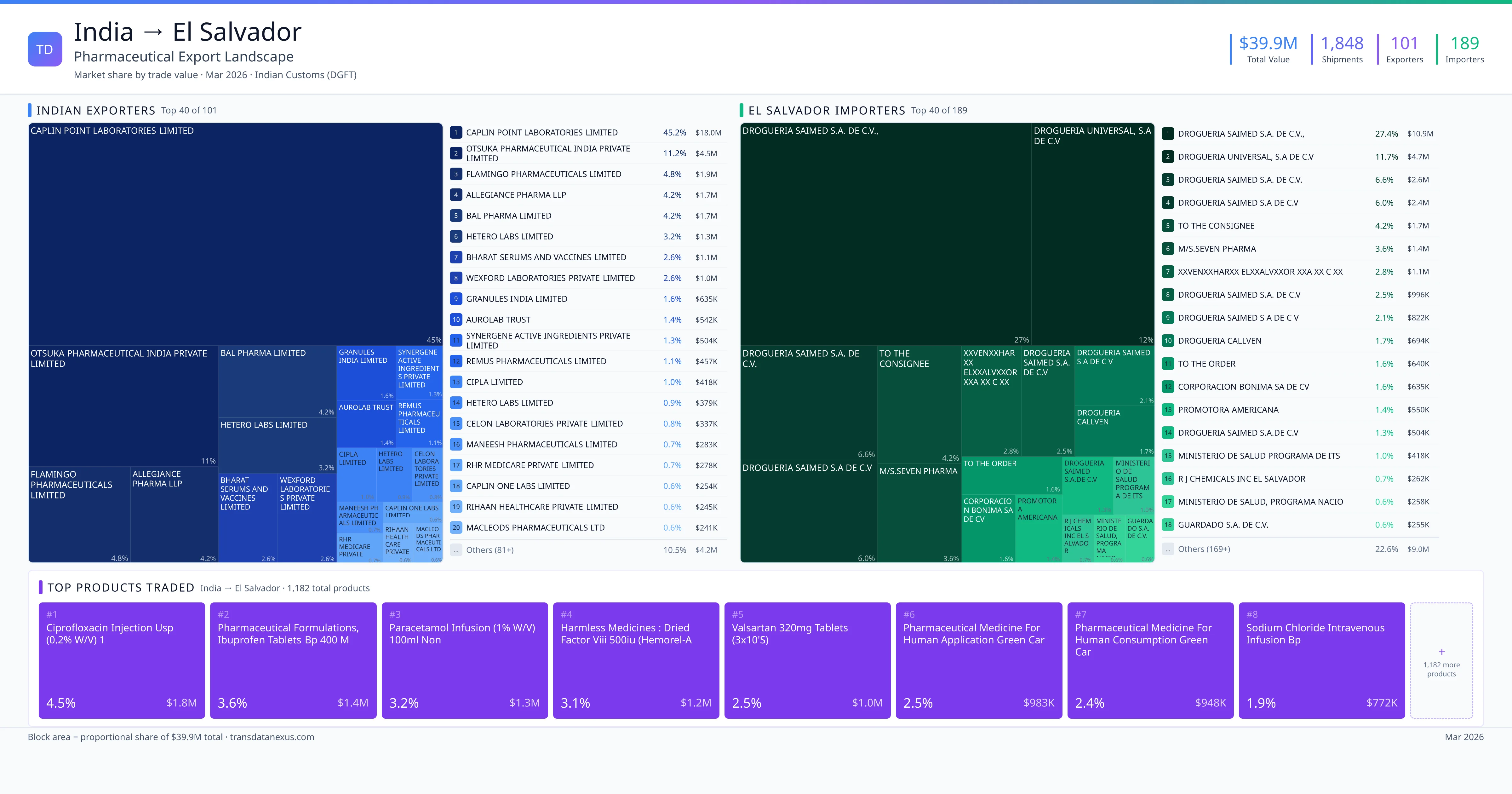
India exported $39.9M worth of pharmaceutical formulations to El Salvador across 1,848 verified shipments, sourced from 101 Indian exporters supplying 189 El Salvador buyers. The top exporters are CAPLIN POINT LABORATORIES LIMITED ($18.0M) and OTSUKA PHARMACEUTICAL INDIA PRIVATE LIMITED ($4.5M). The leading products are Ciprofloxacin ($1.8M) and Ibuprofen ($1.4M). Average shipment value: $21.6K.
Top Pharmaceutical Formulations — India to El Salvador
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Ciprofloxacin Injection USP (0.2% W/v) 1 | $1.8M | 4.5% |
| 2 | Pharmaceutical Formulations, Ibuprofen Tablets BP 400 Mg, Batch No-230815 to 23… | $1.4M | 3.6% |
| 3 | Paracetamol Infusion (1% W/v) 100ml Non | $1.3M | 3.2% |
| 4 | Harmless Medicines : Dried Factor Viii 500iu (hemorel-a 500) | $1.2M | 3.1% |
| 5 | Valsartan 320mg Tablets (3x10's) | $1.0M | 2.5% |
| 6 | Pharmaceutical Medicine for Human Consumption Green Card No.21168 Valid Upto 31… | $947.6K | 2.4% |
| 7 | Sodium Chloride Intravenous Infusion BP | $772.1K | 1.9% |
| 8 | Metronidazole Intravenous Infusion BP (0 | $604.6K | 1.5% |
| 9 | Pharmaceutical Medicine for Human Application Green Card No.21168 Valid Upto 31… | $597.7K | 1.5% |
| 10 | Gamma IV 5 Gm # | $549.1K | 1.4% |
| 11 | Aurocoat [sodium Hyaluronate 2% with Cho | $537.2K | 1.3% |
| 12 | Clarithromycin Tablets USP 500mg | $496.7K | 1.2% |
| 13 | Valsartan Tablets USP 160mg | $465.3K | 1.2% |
| 14 | Harmless Medicines: Dried Factor Viii 500iu (hemorel-a 500) | $420.8K | 1.1% |
| 15 | Hydralazine Hydrochloride Tablets USP 50mgnos | $400.9K | 1.0% |
India exports 20+ pharmaceutical formulations to El Salvador with a combined trade value of $39.9M. Key products include Ciprofloxacin ($1.8M), Ibuprofen ($1.4M), Paracetamol ($1.3M), Valsartan ($1.0M), Sodium ($772K). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 1,848 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to El Salvador Trade Routes
These are the top pharmaceutical products exported from India to El Salvador, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, El Salvador buyers, regulatory requirements, and logistics for that specific product corridor. Products include Ciprofloxacin ($1.8M), Ibuprofen ($1.4M), Paracetamol ($1.3M), Valsartan ($1.0M), Sodium ($772K) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to El Salvador
101 Indian pharmaceutical companies export finished formulations to El Salvador. Leading exporters include Caplin Point Laboratories Limited, Otsuka Pharmaceutical India Private Limited, Flamingo Pharmaceuticals Limited, Allegiance Pharma Llp. The top exporter accounts for 45.2% of total India–El Salvador pharma exports. Source: Indian Customs (DGFT).
Top El Salvador Buyers from India
189 companies in El Salvador import pharmaceutical formulations from India. Top buyers include Drogueria Saimed S.a. De C.v.,, Drogueria Universal, S.a De C.v, Drogueria Saimed S.a. De C.v., Drogueria Saimed S.a De C.v. The largest buyer accounts for 27.4% of India–El Salvador pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to El Salvador Pharmaceutical Shipments
Indian Export Ports
El Salvador Import Ports
Trade Statistics
Other Destinations
Product Routes
India–El Salvador Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and El Salvador has experienced significant growth over the past decade. In 2024, India exported pharmaceutical products valued at $32.38 million to El Salvador, marking a substantial increase from previous years. This upward trajectory underscores the strengthening trade relations and the growing demand for Indian pharmaceutical products in the Salvadoran market.
Key milestones include the establishment of the National Directorate of Medicines (DNM) in El Salvador in 2012, which streamlined the regulatory framework for pharmaceutical imports. This development facilitated smoother market entry for Indian exporters. Additionally, the implementation of the Law on Medicines in 2012 and subsequent regulations in 2014, such as the control of antimicrobial sales, have further shaped the trade dynamics.
2India's Market Position
India holds a significant position in El Salvador's pharmaceutical import landscape. In 2024, pharmaceutical imports from India amounted to $55.38 million, indicating a robust demand for Indian products. This positions India as a key supplier, contributing to over half of El Salvador's pharmaceutical imports.
The strategic importance of this trade is evident in the diversification of India's export portfolio, which includes a wide range of pharmaceutical formulations catering to various therapeutic areas. This diversity enhances India's competitiveness and appeal in the Salvadoran market.
3Recent Developments
Between 2024 and 2026, several developments have influenced the India-El Salvador pharmaceutical trade. In January 2025, El Salvador published the Technical Regulation RTS 11.02.04:24, which provides comprehensive guidelines for the storage, distribution, dispensing, and transportation of pharmaceuticals. This regulation aims to maintain product quality and safety, impacting how Indian exporters manage their supply chains to comply with Salvadoran standards.
Additionally, in November 2022, El Salvador's National Directorate of Medicines (DNM) implemented measures to control the sale of antimicrobials, reflecting a global trend towards regulating antimicrobial use. These measures may affect the types of pharmaceutical products in demand and influence the strategic decisions of Indian exporters.
El Salvador Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to register products in El Salvador must adhere to the requirements set by the National Directorate of Medicines (DNM). The registration process involves submitting a comprehensive dossier that includes:
- Power of Attorney: An authenticated document granting authorization for the registration process.
- Free Sale Certificate: A certificate indicating that the product is freely sold in the country of origin, authenticated by the Salvadoran consulate or via apostille.
- Good Manufacturing Practices (GMP) Certificate: A certificate confirming that the manufacturing facility complies with GMP standards, also authenticated as above.
- Quali-Quantitative Formula: A detailed formulation of the product, signed by the responsible party, expressed in the International System of Units.
These documents ensure that the pharmaceutical products meet the safety and efficacy standards required by Salvadoran authorities. (goldservice.com.sv)
2GMP & Facility Requirements
El Salvador requires that manufacturing facilities comply with Good Manufacturing Practices (GMP) to ensure the quality and safety of pharmaceutical products. The DNM conducts inspections to verify compliance with these standards. Notably, El Salvador accepts GMP certificates from recognized authorities, such as the U.S. FDA, which can streamline the approval process for Indian manufacturers with existing certifications.
3Import Documentation
To import pharmaceutical products into El Salvador, the following documentation is typically required:
- Commercial Invoice: A detailed invoice outlining the transaction specifics.
- Bill of Lading: A document issued by the carrier acknowledging receipt of the goods.
- Import License: An authorization from the relevant Salvadoran authorities, such as the DNM, especially for pharmaceutical products.
- Sanitary Certificate: For certain products, a certificate from the Ministry of Public Health may be necessary.
These documents facilitate the customs clearance process and ensure compliance with Salvadoran import regulations.
Product Categories & Therapeutic Trends — India to El Salvador
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
The dominant categories in India's pharmaceutical exports to El Salvador include:
- Antimicrobials: Reflecting global health priorities, antimicrobials are a significant export category.
- Analgesics and Anti-inflammatory Agents: Products like ibuprofen tablets and paracetamol infusions cater to common health concerns in El Salvador.
- Cardiovascular Drugs: Medications such as valsartan tablets address prevalent cardiovascular conditions in the region.
These categories align with El Salvador's healthcare needs, indicating a strategic focus by Indian exporters on high-demand therapeutic areas.
2Emerging Opportunities
The expiration of patents for certain blockbuster drugs presents opportunities for Indian exporters to introduce generic versions into the Salvadoran market. Additionally, the increasing demand for biosimilars and the expansion into new therapeutic areas, such as oncology and immunology, offer avenues for growth. Indian manufacturers can leverage their expertise to meet these emerging demands.
3Demand Drivers
El Salvador's disease burden, particularly concerning infectious diseases and chronic conditions, drives the demand for pharmaceutical imports. The aging population further contributes to the need for medications addressing age-related health issues. Moreover, trends in healthcare spending indicate a growing capacity and willingness to invest in quality pharmaceuticals, enhancing the market potential for Indian exporters.
Trade Policy & Tariff Intelligence — India and El Salvador
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
El Salvador's tariff structure for pharmaceutical imports is governed by the Central American Common Market (CACM) regulations. While specific Most Favored Nation (MFN) tariff rates and preferential rates are subject to change, pharmaceutical products often benefit from reduced tariffs under regional agreements. Duty exemptions may apply to certain essential medicines, facilitating smoother market access for Indian exporters.
2Trade Agreements
El Salvador is a member of the Central American Integration System (SICA), which includes trade agreements that may impact pharmaceutical trade. While there is no specific Free Trade Agreement (FTA) between India and El Salvador, regional agreements within SICA can influence trade dynamics. Ongoing negotiations and collaborations aim to enhance trade relations, potentially benefiting pharmaceutical exports.
3IP & Patent Landscape
El Salvador adheres to international intellectual property standards, including the Trade-Related Aspects of Intellectual Property Rights (TRIPS) Agreement. Patent provisions, data exclusivity, and compulsory licensing are in place, affecting the entry of Indian generics. Understanding these regulations is crucial for Indian exporters to navigate the Salvadoran market effectively.
Supply Chain & Logistics — India to El Salvador Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to El Salvador typically utilize major sea routes, with transit times varying based on the shipping method and specific ports used. Air freight offers faster delivery but at a higher cost. Recent geopolitical events, such as disruptions in the Red Sea, have not significantly impacted these routes, ensuring consistent delivery times.
2Port Infrastructure
In India, key export ports include:
- Chennai Sea Port: Handling a significant portion of pharmaceutical exports.
- Sahar Air Cargo Complex: Facilitating air shipments.
In El Salvador, primary import ports are:
- San Salvador: The capital city, serving as a central hub for imports.
- Acajutla: A major seaport handling a substantial volume of goods.
These ports are equipped to handle pharmaceutical imports, ensuring efficient logistics for Indian exporters.
3Cold Chain & Compliance
Maintaining the integrity of pharmaceutical products during transit is paramount. Temperature-controlled logistics are essential, with compliance to Good Distribution Practices (GDP) and packaging standards required to preserve product quality. Indian exporters must collaborate with logistics providers experienced in pharmaceutical shipments to meet these standards.
Market Opportunity Assessment — El Salvador for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
El Salvador's pharmaceutical market is experiencing growth, with imports reaching $55.38 million in 2024. The market is characterized by a high dependency on imports, with generics comprising a significant share. This presents a substantial opportunity for Indian exporters to expand their presence.
2Healthcare System
El Salvador's government healthcare programs and insurance coverage are evolving, with efforts to
FAQ — India to El Salvador Pharmaceutical Trade
What is the total value of India's pharmaceutical export to El Salvador?
India exported pharmaceuticals worth $39.9M to El Salvador across 1,848 verified shipments.
Who are the top Indian pharmaceutical exporters to El Salvador?
1. CAPLIN POINT LABORATORIES LIMITED — $18.0M. 2. OTSUKA PHARMACEUTICAL INDIA PRIVATE LIMITED — $4.5M. 3. FLAMINGO PHARMACEUTICALS LIMITED — $1.9M. Total: 101 suppliers.
Which companies in El Salvador import pharmaceuticals from India?
1. DROGUERIA SAIMED S.A. DE C.V., — $10.9M. 2. DROGUERIA UNIVERSAL, S.A DE C.V — $4.7M. 3. DROGUERIA SAIMED S.A. DE C.V. — $2.6M. 189 buyers total.
What pharmaceutical products does India export most to El Salvador?
1. Ciprofloxacin Injection USP (0.2% W/v) 1 ($1.8M, 4.5%); 2. Pharmaceutical Formulations, Ibuprofen Tablets BP 400 Mg, Batch No-230815 to 23… ($1.4M, 3.6%); 3. Paracetamol Infusion (1% W/v) 100ml Non ($1.3M, 3.2%); 4. Harmless Medicines : Dried Factor Viii 500iu (hemorel-a 500) ($1.2M, 3.1%); 5. Valsartan 320mg Tablets (3x10's) ($1.0M, 2.5%)
Which ports handle pharmaceutical shipments from India to El Salvador?
Export: AHEMDABAD ICD, CHENNAI SEA, SAHAR AIR, KAMARAJAR PORT (INENR1), KATTUPALLI PORT SEA. Import: SAN SALVADOR, Acajutla, ACAJUTLA, San Salvador, LA PAZ.
Why does El Salvador import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $39.9M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply El Salvador?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to El Salvador pharmaceutical trade?
$21.6K per consignment across 1,848 shipments.
How many Indian pharmaceutical companies export to El Salvador?
101 Indian companies. Largest: CAPLIN POINT LABORATORIES LIMITED with $18.0M.
How can I find verified Indian pharmaceutical suppliers for El Salvador?
TransData Nexus covers 101 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to El Salvador Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 1,848 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to El Salvador identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 1,848 verified shipments from 101 Indian exporters to 189 El Salvador buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
101 Exporters
189 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists