India to Dominican Republic Pharmaceutical Export
Bilateral Trade Intelligence · $25.8M Total Trade · 150 Exporters · 145 Buyers · DGFT Verified · Updated March 2026
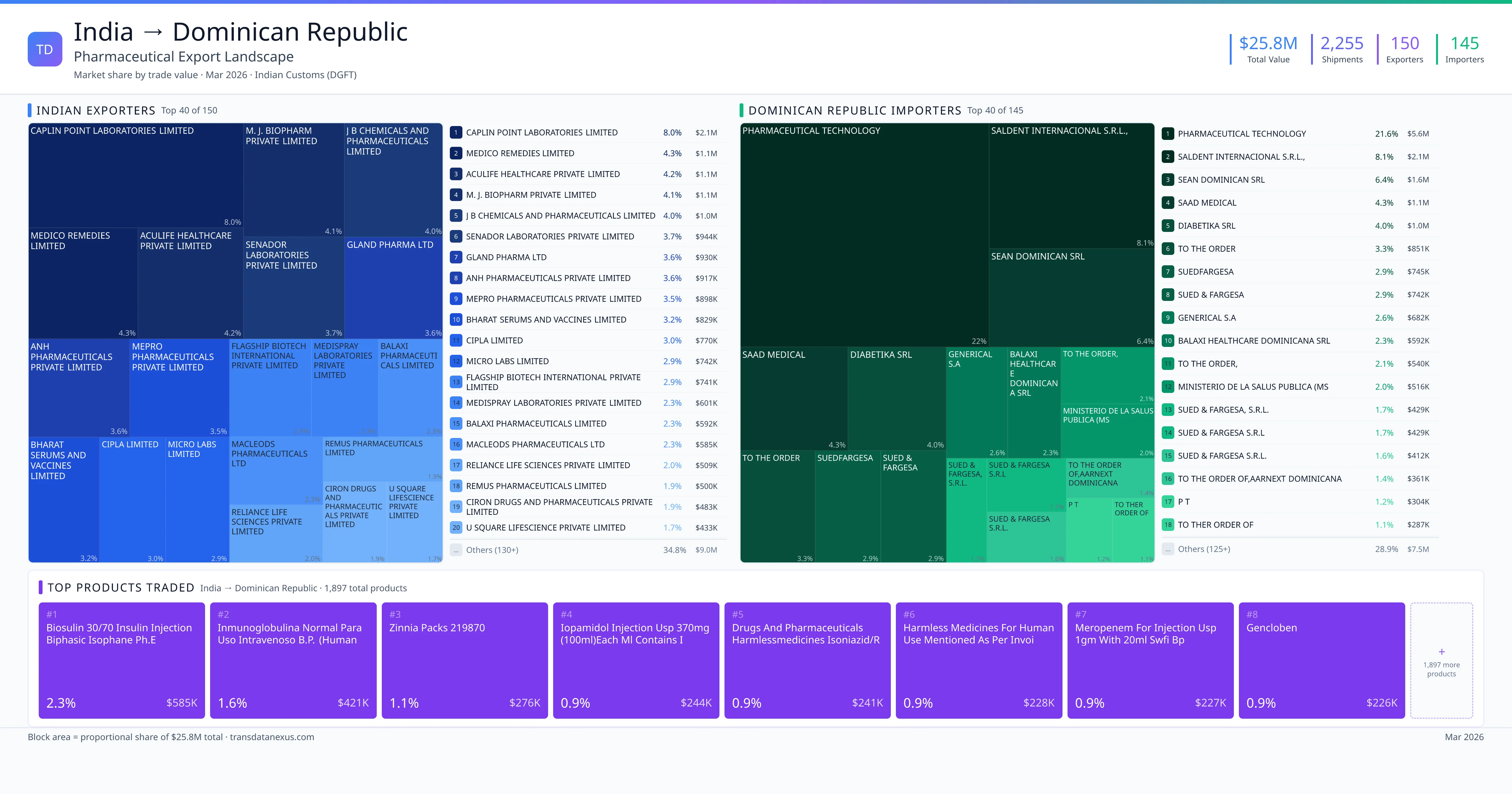
India exported $25.8M worth of pharmaceutical formulations to Dominican Republic across 2,255 verified shipments, sourced from 150 Indian exporters supplying 145 Dominican Republic buyers. The top exporters are CAPLIN POINT LABORATORIES LIMITED ($2.1M) and MEDICO REMEDIES LIMITED ($1.1M). The leading products are Insulin ($585K) and Meropenem ($227K). Average shipment value: $11.5K.
Top Pharmaceutical Formulations — India to Dominican Republic
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Biosulin 30/70 Insulin Injection Biphasic Isophane Ph.eur.100 Iu/ml,10 ML | $585.4K | 2.3% |
| 2 | Inmunoglobulina Normal Para Uso Intravenoso B.p. (humana) 5.0 G | $420.8K | 1.6% |
| 3 | Zinnia Packs 219870 | $275.9K | 1.1% |
| 4 | Iopamidol Injection USP 370mg (100ml)each ML Contains Iopamidol Usp-755.30 MG N… | $243.6K | 0.9% |
| 5 | Drugs and Pharmaceuticals Harmlessmedicines Isoniazid/rifapentine Coated Tablet… | $241.2K | 0.9% |
| 6 | Harmless Medicines for Human Use Mentioned As Per Invoice -icona London Triple… | $227.9K | 0.9% |
| 7 | Meropenem for Injection USP 1gm with 20ml Swfi BP | $226.8K | 0.9% |
| 8 | Gencloben | $226.1K | 0.9% |
| 9 | Tetglob 250.i.u | $215.9K | 0.8% |
| 10 | Low Molecular Weight Heparin Enoxaparinsodium Injection Usp, 40mg in 0.4ml Pre-… | $199.2K | 0.8% |
| 11 | Gencloben Gencloben 30g (gentamicina +betametasona + Clotrimazol Crema) | $197.3K | 0.8% |
| 12 | Lev/ee/fe 0,15-0,03-75 1x21+7 Do1 (5,537.000 X2800x1) | $192.0K | 0.7% |
| 13 | Sodium Chloride Intravenous Infusion Bp(0.9% W/v) (ns 1000ml) | $191.4K | 0.7% |
| 14 | Ampicillin Capsules BP 250mg ( =43,00,000 Capsules)(qty Ofampicillin Trihydrate… | $171.8K | 0.7% |
| 15 | Dapaglip 10 ( ) ( 3x10s ) ( Qty- 11717.000 ) | $165.3K | 0.6% |
India exports 20+ pharmaceutical formulations to Dominican Republic with a combined trade value of $25.8M. Key products include Insulin ($585K), Meropenem ($227K), Sodium ($191K), Ampicillin ($172K), Dapagliflozin ($165K). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 2,255 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Dominican Republic Trade Routes
These are the top pharmaceutical products exported from India to Dominican Republic, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Dominican Republic buyers, regulatory requirements, and logistics for that specific product corridor. Products include Insulin ($585K), Meropenem ($227K), Sodium ($191K), Ampicillin ($172K), Dapagliflozin ($165K) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Dominican Republic
150 Indian pharmaceutical companies export finished formulations to Dominican Republic. Leading exporters include Caplin Point Laboratories Limited, Medico Remedies Limited, Aculife Healthcare Private Limited, M. J. Biopharm Private Limited. The top exporter accounts for 8.0% of total India–Dominican Republic pharma exports. Source: Indian Customs (DGFT).
Top Dominican Republic Buyers from India
145 companies in Dominican Republic import pharmaceutical formulations from India. Top buyers include Pharmaceutical Technology, Saldent Internacional S.r.l.,, Sean Dominican Srl, Saad Medical. The largest buyer accounts for 21.6% of India–Dominican Republic pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Dominican Republic Pharmaceutical Shipments
Indian Export Ports
Dominican Republic Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Dominican Republic Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and the Dominican Republic has experienced significant growth over the past decade. In 2022, the total export value reached $25.8 million USD, with 2,255 shipments facilitated by 150 Indian exporters to 145 Dominican Republic buyers. This surge underscores the strengthening bilateral trade relations and India's expanding footprint in the Caribbean pharmaceutical market.
Key milestones include the participation of a Dominican delegation in the Global Conclave of Pharmaceuticals Regulatory Agencies in New Delhi from September 21 to 23, 2022. This event, organized by the Pharmaceuticals Export Promotion Council of India (Pharmexcil), aimed to enhance regulatory cooperation and market access between the two nations.
2India's Market Position
India holds a substantial share in the Dominican Republic's pharmaceutical imports, positioning itself as a primary supplier of finished pharmaceutical formulations. The country's competitive pricing, adherence to WHO-GMP standards, and a diverse product portfolio contribute to its strategic importance in meeting the Dominican Republic's healthcare needs.
3Recent Developments
Between 2024 and 2026, several developments have influenced the India-Dominican Republic pharmaceutical trade. The Dominican Republic's participation in international pharmaceutical forums has facilitated deeper regulatory alignment and market integration. Additionally, the establishment of direct shipping routes has improved logistics efficiency, reducing transit times and costs for pharmaceutical exports.
Dominican Republic Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to export to the Dominican Republic must navigate a structured registration process. Products must be registered with the Dirección General de Medicamentos, Alimentos y Productos Sanitarios (DIGEMAPS), the country's regulatory authority. The registration dossier should comply with the Common Technical Document (CTD) format, ensuring comprehensive data on quality, safety, and efficacy. Bioequivalence studies are mandatory for generics to demonstrate therapeutic equivalence to the reference product. The registration timeline typically spans 6 to 12 months, depending on the product's complexity and the completeness of the submitted documentation.
2GMP & Facility Requirements
The Dominican Republic mandates that manufacturing facilities adhere to Good Manufacturing Practice (GMP) standards recognized by the World Health Organization (WHO). Indian exporters must provide certificates of GMP compliance from recognized authorities, such as the Central Drugs Standard Control Organisation (CDSCO) in India. DIGEMAPS conducts inspections to verify compliance, and mutual recognition agreements may expedite the approval process for facilities with established GMP credentials.
3Import Documentation
To import pharmaceutical products into the Dominican Republic, Indian exporters must furnish several key documents:
- Certificate of Pharmaceutical Product (CPP): Issued by the CDSCO, confirming the product's registration and GMP compliance.
- Certificate of Analysis (CoA): Demonstrates that the product meets specified quality standards.
- GMP Certificate: Affirms that the manufacturing facility adheres to GMP standards.
Customs procedures require these documents to be in Spanish, and all product labeling must comply with Dominican regulations, including Spanish language requirements. Engaging a licensed Dominican customs broker is advisable to navigate the import process efficiently.
Product Categories & Therapeutic Trends — India to Dominican Republic
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to the Dominican Republic are predominantly in the form of finished formulations, including tablets, capsules, syrups, and injections. This aligns with the Dominican Republic's healthcare needs, particularly in managing chronic diseases such as diabetes and cardiovascular conditions. The demand for affordable and high-quality medications drives the preference for Indian pharmaceutical products.
2Emerging Opportunities
The expiration of patents for several blockbuster drugs presents opportunities for Indian exporters to introduce generic versions in the Dominican Republic. Additionally, the growing demand for biosimilars offers a niche market for Indian companies with the requisite expertise. Expanding into therapeutic areas like oncology and immunology could further diversify India's export portfolio.
3Demand Drivers
The Dominican Republic's increasing disease burden, coupled with an aging population, escalates the demand for pharmaceutical imports. Rising healthcare expenditures and a focus on improving healthcare infrastructure further drive the need for quality pharmaceutical products, positioning India as a key supplier to meet these demands.
Trade Policy & Tariff Intelligence — India and Dominican Republic
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
The Dominican Republic applies Most Favored Nation (MFN) tariff rates to pharmaceutical imports. While specific preferential rates or duty exemptions for pharmaceutical formulations are not detailed, engaging with local trade authorities or customs brokers can provide insights into potential incentives or duty structures applicable to Indian pharmaceutical exports.
2Trade Agreements
As of March 2026, there are no specific Free Trade Agreements (FTAs) between India and the Dominican Republic. However, both nations participate in broader regional trade agreements that may influence pharmaceutical trade dynamics. Ongoing diplomatic engagements and trade discussions aim to enhance bilateral trade relations, potentially leading to more favorable terms for pharmaceutical exports.
3IP & Patent Landscape
The Dominican Republic's intellectual property laws align with international standards, offering protection for patents and data exclusivity. Indian generic manufacturers must navigate these regulations, ensuring compliance to avoid potential legal challenges. Understanding the nuances of compulsory licensing provisions is crucial for Indian exporters to strategize market entry effectively.
Supply Chain & Logistics — India to Dominican Republic Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical exports from India to the Dominican Republic primarily utilize sea routes, with major ports including Nhava Sheva Sea (INNSA1) in India and Caucedo in the Dominican Republic. Air freight options are also available, particularly for temperature-sensitive products. Recent geopolitical events, such as disruptions in the Red Sea, have not significantly impacted these established shipping routes.
2Port Infrastructure
Key export ports in India include Nhava Sheva Sea (INNSA1), handling 34.8% of shipments, and Chennai Sea (INMAA1), accounting for 3.4%. In the Dominican Republic, Caucedo (50.6%) and Santo Domingo (38.0%) serve as primary import hubs. These ports are equipped to handle pharmaceutical imports, ensuring efficient customs clearance and distribution.
3Cold Chain & Compliance
Maintaining the integrity of temperature-sensitive pharmaceutical products is paramount. Indian exporters must implement validated thermal packaging and continuous temperature monitoring throughout transit. Compliance with Good Distribution Practice (GDP) standards and adherence to packaging regulations are essential to meet the Dominican Republic's import requirements.
Market Opportunity Assessment — Dominican Republic for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
The Dominican Republic's pharmaceutical market is valued at approximately $485.36 million USD, with a growth rate of 4.93% in 2022. The market exhibits a high dependency on imports, with generics comprising a significant share. This presents a substantial opportunity for Indian pharmaceutical exporters to expand their presence.
2Healthcare System
The Dominican Republic's healthcare system is characterized by a mix of public and private providers, with government programs aiming to increase insurance coverage and improve drug procurement systems. These initiatives enhance access to essential medications, creating a conducive environment for pharmaceutical imports.
3Opportunity for Indian Exporters
Indian exporters can capitalize on the growing demand for affordable and high-quality pharmaceutical products in the Dominican Republic. Focusing on therapeutic areas such as oncology, antivirals, and anti-HIV medications aligns with the country's healthcare priorities. Establishing partnerships with local distributors and understanding regulatory nuances will be key to successful market penetration.
Competitive Landscape — India vs Other Pharmaceutical Suppliers to Dominican Republic
Competing origins, India's edge, challenges and threats
1Competing Origins
The Dominican Republic's pharmaceutical imports are diversified, with the United States, China, and the European Union being notable suppliers. In 2023, the United States exported $957.83K worth of pharmaceutical glassware, China $182.62K, and the European Union $55.13K.
2India's Competitive Edge
India's competitive advantages include cost-effective manufacturing, adherence to WHO-GMP standards, and a broad portfolio of generic and branded formulations. These factors enable Indian exporters to offer high-quality products at competitive prices, appealing to the Dominican Republic's market needs.
3Challenges & Threats
Key challenges for Indian exporters include navigating complex regulatory requirements, potential competition from established suppliers like China and the European Union, and addressing quality perception issues. Staying abreast of regulatory changes and maintaining high-quality standards are essential to mitigate these threats.
FAQ — India to Dominican Republic Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Dominican Republic?
India exported pharmaceuticals worth $25.8M to Dominican Republic across 2,255 verified shipments.
Who are the top Indian pharmaceutical exporters to Dominican Republic?
1. CAPLIN POINT LABORATORIES LIMITED — $2.1M. 2. MEDICO REMEDIES LIMITED — $1.1M. 3. ACULIFE HEALTHCARE PRIVATE LIMITED — $1.1M. Total: 150 suppliers.
Which companies in Dominican Republic import pharmaceuticals from India?
1. PHARMACEUTICAL TECHNOLOGY — $5.6M. 2. SALDENT INTERNACIONAL S.R.L., — $2.1M. 3. SEAN DOMINICAN SRL — $1.6M. 145 buyers total.
What pharmaceutical products does India export most to Dominican Republic?
1. Biosulin 30/70 Insulin Injection Biphasic Isophane Ph.eur.100 Iu/ml,10 ML ($585.4K, 2.3%); 2. Inmunoglobulina Normal Para Uso Intravenoso B.p. (humana) 5.0 G ($420.8K, 1.6%); 3. Zinnia Packs 219870 ($275.9K, 1.1%); 4. Iopamidol Injection USP 370mg (100ml)each ML Contains Iopamidol Usp-755.30 MG N… ($243.6K, 0.9%); 5. Drugs and Pharmaceuticals Harmlessmedicines Isoniazid/rifapentine Coated Tablet… ($241.2K, 0.9%)
Which ports handle pharmaceutical shipments from India to Dominican Republic?
Export: NHAVA SHEVA SEA (INNSA1), SAHAR AIR CARGO ACC (INBOM4), CHENNAI SEA (INMAA1), DELHI AIR CARGO ACC (INDEL4), BANGALORE ACC (INBLR4). Import: CAUCEDO DOMINIC REP., SANTO DOMINGO, Santo Domingo, Rio Haina, Matadi.
Why does Dominican Republic import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $25.8M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Dominican Republic?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Dominican Republic pharmaceutical trade?
$11.5K per consignment across 2,255 shipments.
How many Indian pharmaceutical companies export to Dominican Republic?
150 Indian companies. Largest: CAPLIN POINT LABORATORIES LIMITED with $2.1M.
How can I find verified Indian pharmaceutical suppliers for Dominican Republic?
TransData Nexus covers 150 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Dominican Republic Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 2,255 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Dominican Republic identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 2,255 verified shipments from 150 Indian exporters to 145 Dominican Republic buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
150 Exporters
145 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists