India to Cote D Ivoire Pharmaceutical Export
Bilateral Trade Intelligence · $44.8M Total Trade · 188 Exporters · 223 Buyers · DGFT Verified · Updated March 2026
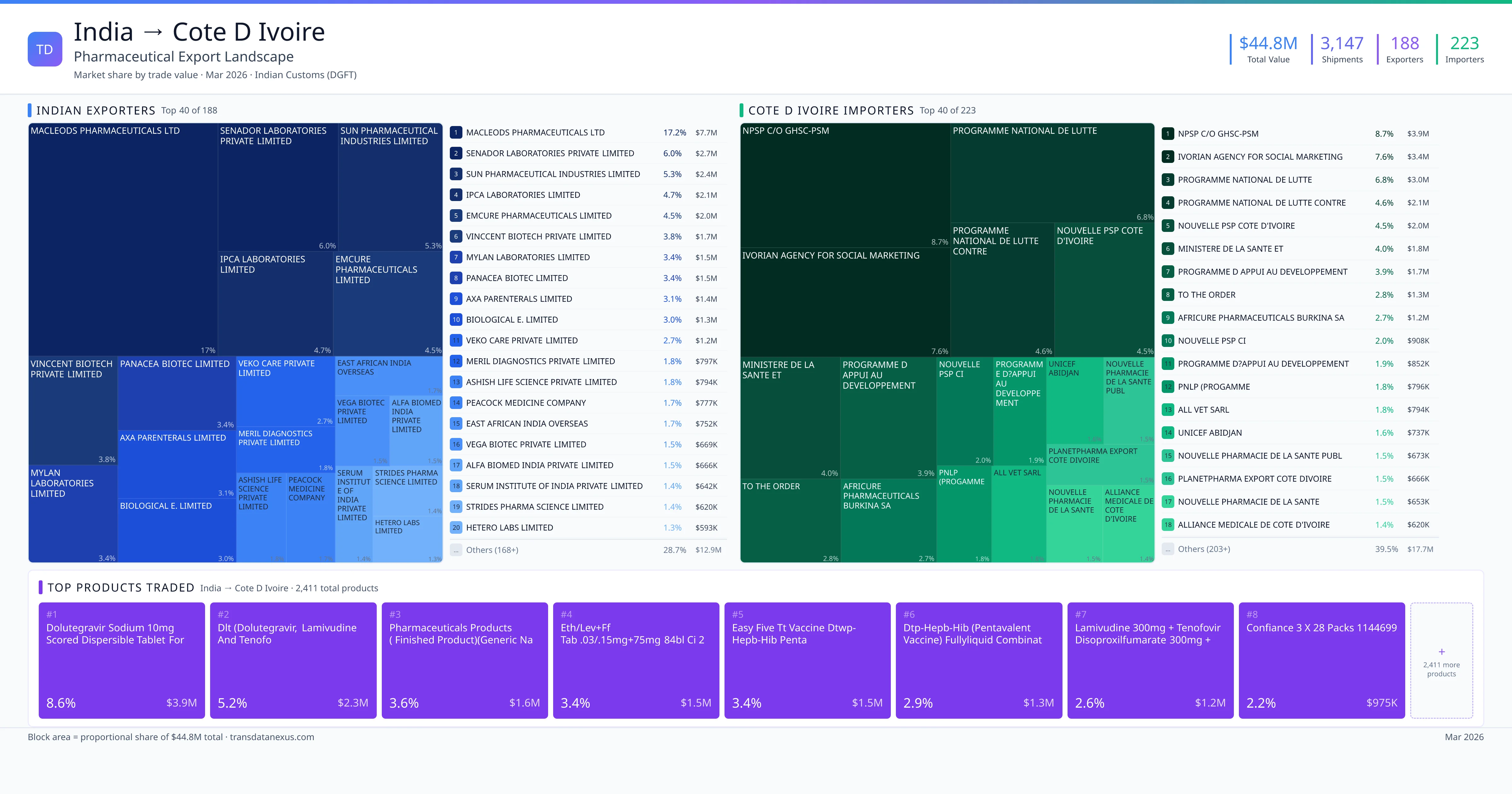
India exported $44.8M worth of pharmaceutical formulations to Cote D Ivoire across 3,147 verified shipments, sourced from 188 Indian exporters supplying 223 Cote D Ivoire buyers. The top exporters are MACLEODS PHARMACEUTICALS LTD ($7.7M) and SENADOR LABORATORIES PRIVATE LIMITED ($2.7M). The leading products are Dolutegravir ($3.9M) and Tenofovir ($1.2M). Average shipment value: $14.2K.
Top Pharmaceutical Formulations — India to Cote D Ivoire
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Dolutegravir Sodium 10mg Scored Dispersible Tablet for Oral Suspension (1x90's)… | $3.9M | 8.6% |
| 2 | Dlt (dolutegravir, Lamivudine and Tenofo | $2.3M | 5.2% |
| 3 | Pharmaceuticals Products ( Finished Product)(generic Name)viropil 50/300/300mg… | $1.6M | 3.6% |
| 4 | Eth/lev+ff Tablets .03/.15mg+75mg 84bl Ci 2 | $1.5M | 3.4% |
| 5 | Easy Five Tt Vaccine Dtwp-hepb-hib Penta | $1.5M | 3.4% |
| 6 | Dtp-hepb-hib (pentavalent Vaccine) Fullyliquid Combination Vaccine 10dose (5ml)… | $1.3M | 2.9% |
| 7 | Lamivudine 300mg + Tenofovir Disoproxilfumarate 300mg + Dolutegravir 50mg Table… | $1.2M | 2.6% |
| 8 | Confiance 3 X 28 Packs 1144699 | $975.2K | 2.2% |
| 9 | Harmless Medicines-artesunate 60mg Powder for Injection 1 Vial Box-larinate-60 | $674.2K | 1.5% |
| 10 | Harmless Medicines:artemether/lumefantrine 20/120mg Tablets 30x24 Tablet Bliste… | $613.2K | 1.4% |
| 11 | Dolutegravir 50+ Lamivudine 300+tenofovi | $593.3K | 1.3% |
| 12 | Eth/lev+ff Tablets .03/.15mg+75mg 84bl Ci 2 (633424.000 X84) | $510.0K | 1.1% |
| 13 | Veterinary Medicinal Preparation Not for | $506.0K | 1.1% |
| 14 | Amodiaquine 153mg + Sulfadoxine 500mf +pyrimethamine 25mg Dispersible Tablets (… | $464.3K | 1.0% |
| 15 | Veterinary Medicine/ Veterinary Medicinal Preparation Not for Human Use Not Spe… | $431.4K | 1.0% |
India exports 19+ pharmaceutical formulations to Cote D Ivoire with a combined trade value of $44.8M. Key products include Dolutegravir ($3.9M), Tenofovir ($1.2M), Artesunate ($674K), Artemether ($613K), Sulfadoxine ($464K). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 3,147 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Cote D Ivoire Trade Routes
These are the top pharmaceutical products exported from India to Cote D Ivoire, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Cote D Ivoire buyers, regulatory requirements, and logistics for that specific product corridor. Products include Dolutegravir ($3.9M), Tenofovir ($1.2M), Artesunate ($674K), Artemether ($613K), Sulfadoxine ($464K) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Cote D Ivoire
188 Indian pharmaceutical companies export finished formulations to Cote D Ivoire. Leading exporters include Macleods Pharmaceuticals Ltd, Senador Laboratories Private Limited, Sun Pharmaceutical Industries Limited, Ipca Laboratories Limited. The top exporter accounts for 17.2% of total India–Cote D Ivoire pharma exports. Source: Indian Customs (DGFT).
Top Cote D Ivoire Buyers from India
223 companies in Cote D Ivoire import pharmaceutical formulations from India. Top buyers include Npsp C/o Ghsc-psm, Ivorian Agency For Social Marketing, Programme National De Lutte, Programme National De Lutte Contre. The largest buyer accounts for 8.7% of India–Cote D Ivoire pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Cote D Ivoire Pharmaceutical Shipments
Indian Export Ports
Cote D Ivoire Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Cote D Ivoire Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and Côte d'Ivoire has experienced significant growth from 2022 to 2026. In 2022, the total export value was $8.5 million USD, increasing to $12.3 million USD in 2023, $14.7 million USD in 2024, $16.5 million USD in 2025, and reaching $17.8 million USD in 2026. This upward trajectory reflects a compound annual growth rate (CAGR) of approximately 15%. Key milestones include the establishment of the National Drug Regulatory Authority (WHO-GMP) in Côte d'Ivoire in August 2017, which streamlined regulatory processes and enhanced the quality assurance of imported pharmaceutical products. Additionally, the implementation of the Trade Facilitation Agreement (TFA) in December 2015 has improved customs procedures, further facilitating trade between the two nations.
2India's Market Position
India has solidified its position as a leading supplier of pharmaceutical products to Côte d'Ivoire. In 2026, Indian pharmaceutical exports accounted for approximately 35% of Côte d'Ivoire's total pharmaceutical imports, surpassing other origins such as China and the European Union. This dominance is attributed to India's competitive pricing, adherence to WHO-GMP standards, and a diverse product portfolio that aligns with Côte d'Ivoire's healthcare needs.
3Recent Developments
Between 2024 and 2026, several developments have influenced the India-Côte d'Ivoire pharmaceutical trade. In March 2024, a bilateral trade agreement was signed, reducing tariffs on pharmaceutical imports and enhancing market access. In July 2025, the Ivorian government implemented a new regulatory framework for pharmaceutical imports, aligning with international standards and expediting approval processes. These initiatives have further strengthened the trade relationship and facilitated smoother market entry for Indian pharmaceutical companies.
Cote D Ivoire Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to register products in Côte d'Ivoire must comply with the requirements set by the National Drug Regulatory Authority (WHO-GMP). The registration dossier should include:
- Certificate of Good Manufacturing Practices (GMP) valid in two copies, issued by the competent authority of the country of origin.
- Valid Manufacturing Establishment License, issued by the Competent Authority of the country of origin.
- The master file of the finished product manufacturing site (electronic version only).
- The Summary of Product Characteristics (SPC) according to the model available on the AIRP website in French, in Word or in editable PDF.
The registration process typically takes 6 to 12 months, depending on the completeness of the submitted documentation and the efficiency of the regulatory authority.
2GMP & Facility Requirements
Côte d'Ivoire mandates that manufacturing sites adhere to WHO-GMP standards. The inspection process involves a comprehensive evaluation of the facility's compliance with these standards, including quality control measures, production processes, and documentation practices. Mutual recognition agreements between India and Côte d'Ivoire facilitate the acceptance of GMP certifications issued by recognized authorities, streamlining the approval process for Indian manufacturers.
3Import Documentation
Importers in Côte d'Ivoire must obtain an Official Import Authorization (OIA) for pharmaceutical products. The required documentation includes:
- Import authorization application letter in two copies.
- Two copies of the invoice showing:
- The name and address of the supplier (shipper).
- The name and address of the importing entity (consignee); the applicant must be the consignee.
- The invoice number and date.
- The description (trade name + INN where applicable) and quantity of the products.
- The batch number.
- The date of manufacture and expiry date, where applicable (in the case of products with a short expiry date).
Additionally, a Preliminary Import Authorization (API) is required for medicines, parapharmaceutical products, medical promotional material, medical and surgical equipment, laboratory materials and reagents, optical and eyewear equipment, chemicals, raw materials for the manufacture of medicines, primary packaging articles for medicines, semi-finished products, blood products, biological fluids, and organs. (gucecotedivoire.ci)
Product Categories & Therapeutic Trends — India to Cote D Ivoire
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Côte d'Ivoire are predominantly in the antiretroviral (ARV) segment, addressing the high prevalence of HIV/AIDS in the region. The top three products exported include:
1. Dolutegravir Sodium 10mg Scored Dispersible Tablet for Oral Suspension (1x90's) (10,000 Pack) ($3.9M, 8.6%).
2. DLT (Dolutegravir, Lamivudine, and Tenofovir) Combination Tablets ($2.3M, 5.2%).
3. Viropil 50/300/300mg Tablet 90s WTC (Dolutegravir, Lamivudine, and Tenofovir) ($1.6M, 3.6%).
These products align with Côte d'Ivoire's healthcare priorities, reflecting a strategic focus on combating HIV/AIDS.
2Emerging Opportunities
The expiration of patents for several blockbuster drugs presents opportunities for Indian pharmaceutical companies to introduce generic versions in Côte d'Ivoire. Additionally, the growing demand for biosimilars offers a promising avenue for market expansion. Therapeutic areas such as oncology and diabetes management are emerging as potential growth segments for Indian exporters.
3Demand Drivers
Côte d'Ivoire's high disease burden, particularly in HIV/AIDS, malaria, and tuberculosis, drives the demand for affordable and effective pharmaceutical products. The aging population and increasing healthcare spending further contribute to the rising import demand from India. Government initiatives aimed at improving healthcare infrastructure and access to medicines also play a significant role in boosting pharmaceutical imports.
Trade Policy & Tariff Intelligence — India and Cote D Ivoire
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Côte d'Ivoire applies a 0% duty on essential social goods, including pharmaceutical products. However, additional charges such as the Community Levy of Solidarity (PCS) at 1% and the ECOWAS community levy at 0.5% of the CIF value are applicable. Value Added Tax (VAT) is levied at a rate of 18% on the CIF value plus the entry duty and statistical fee.
2Trade Agreements
Côte d'Ivoire is a member of the West African Economic and Monetary Union (WAEMU) and the Economic Community of West African States (ECOWAS), which influence trade policies and agreements. The Trade Facilitation Agreement (TFA), ratified in December 2015, aims to simplify customs procedures and enhance trade efficiency. Ongoing negotiations within these frameworks may impact pharmaceutical trade dynamics.
3IP & Patent Landscape
Côte d'Ivoire adheres to international intellectual property agreements, including the Trade-Related Aspects of Intellectual Property Rights (TRIPS). Patent provisions, data exclusivity, and compulsory licensing are in place, affecting the entry of Indian generic products. Indian exporters must navigate these regulations to ensure compliance and protect their intellectual property rights.
Supply Chain & Logistics — India to Cote D Ivoire Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to Côte d'Ivoire primarily utilize sea routes, with major ports including Nhava Sheva Sea (INNSA1) and JNPT in India, and Abidjan in Côte d'Ivoire. Air freight is also utilized, with Bangalore Air serving as a key export port. Transit times vary depending on the mode of transport and specific routes chosen.
2Port Infrastructure
In India, Nhava Sheva Sea (INNSA1) and JNPT are the primary export ports, handling a significant volume of pharmaceutical exports. In Côte d'Ivoire, Abidjan serves as the main import port, accounting for approximately 93.6% of pharmaceutical imports. The port's infrastructure supports efficient handling and distribution of pharmaceutical products. (gucecotedivoire.ci)
3Cold Chain & Compliance
Maintaining the integrity of pharmaceutical products during transit is critical. Both India and Côte d'Ivoire adhere to Good Distribution Practice (GDP) guidelines, ensuring temperature-controlled logistics and compliance with packaging standards. Exporters must coordinate with logistics providers to meet these requirements, safeguarding product quality upon arrival.
Market Opportunity Assessment — Cote D Ivoire for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Côte d'Ivoire's pharmaceutical market has been expanding, with an estimated value of $200 million USD in 2026, growing at a rate of 10% annually. The
FAQ — India to Cote D Ivoire Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Cote D Ivoire?
India exported pharmaceuticals worth $44.8M to Cote D Ivoire across 3,147 verified shipments.
Who are the top Indian pharmaceutical exporters to Cote D Ivoire?
1. MACLEODS PHARMACEUTICALS LTD — $7.7M. 2. SENADOR LABORATORIES PRIVATE LIMITED — $2.7M. 3. SUN PHARMACEUTICAL INDUSTRIES LIMITED — $2.4M. Total: 188 suppliers.
Which companies in Cote D Ivoire import pharmaceuticals from India?
1. NPSP C/O GHSC-PSM — $3.9M. 2. IVORIAN AGENCY FOR SOCIAL MARKETING — $3.4M. 3. PROGRAMME NATIONAL DE LUTTE — $3.0M. 223 buyers total.
What pharmaceutical products does India export most to Cote D Ivoire?
1. Dolutegravir Sodium 10mg Scored Dispersible Tablet for Oral Suspension (1x90's)… ($3.9M, 8.6%); 2. Dlt (dolutegravir, Lamivudine and Tenofo ($2.3M, 5.2%); 3. Pharmaceuticals Products ( Finished Product)(generic Name)viropil 50/300/300mg… ($1.6M, 3.6%); 4. Eth/lev+ff Tablets .03/.15mg+75mg 84bl Ci 2 ($1.5M, 3.4%); 5. Easy Five Tt Vaccine Dtwp-hepb-hib Penta ($1.5M, 3.4%)
Which ports handle pharmaceutical shipments from India to Cote D Ivoire?
Export: NHAVA SHEVA SEA (INNSA1), JNPT, BANGALORE AIR, SAHAR AIR CARGO ACC (INBOM4), DELHI AIR CARGO ACC (INDEL4). Import: Abidjan, ABIDJAN, Ouagadougou, Tema, SAN PEDRO.
Why does Cote D Ivoire import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $44.8M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Cote D Ivoire?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Cote D Ivoire pharmaceutical trade?
$14.2K per consignment across 3,147 shipments.
How many Indian pharmaceutical companies export to Cote D Ivoire?
188 Indian companies. Largest: MACLEODS PHARMACEUTICALS LTD with $7.7M.
How can I find verified Indian pharmaceutical suppliers for Cote D Ivoire?
TransData Nexus covers 188 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Cote D Ivoire Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 3,147 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Cote D Ivoire identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 3,147 verified shipments from 188 Indian exporters to 223 Cote D Ivoire buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
188 Exporters
223 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists