India to Congo Dr Pharmaceutical Export
Bilateral Trade Intelligence · $14.1M Total Trade · 95 Exporters · 80 Buyers · DGFT Verified · Updated March 2026
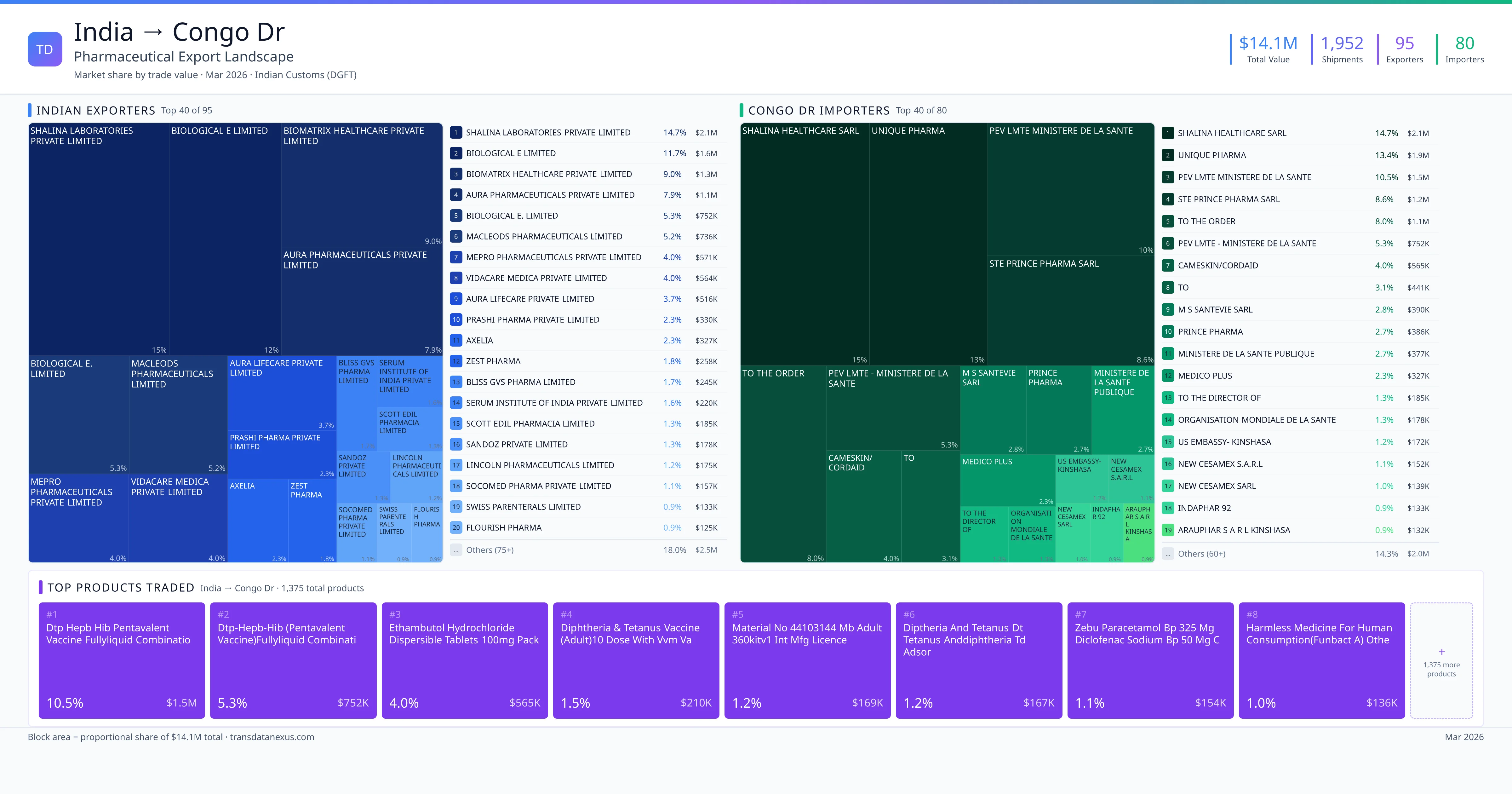
India exported $14.1M worth of pharmaceutical formulations to Congo Dr across 1,952 verified shipments, sourced from 95 Indian exporters supplying 80 Congo Dr buyers. The top exporters are SHALINA LABORATORIES PRIVATE LIMITED ($2.1M) and BIOLOGICAL E LIMITED ($1.6M). The leading products are Ethambutol ($565K) and Paracetamol ($154K). Average shipment value: $7.2K.
Top Pharmaceutical Formulations — India to Congo Dr
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Dtp Hepb Hib Pentavalent Vaccine Fullyliquid Combination Vaccine 10 Dose Vialpa… | $1.5M | 10.5% |
| 2 | Dtp-hepb-hib (pentavalent Vaccine)fullyliquid Combination Vaccine 10 Dose (5ml)… | $752.4K | 5.3% |
| 3 | Ethambutol Hydrochloride Dispersible Tablets 100mg Pack28160 | $564.6K | 4.0% |
| 4 | Diphtheria & Tetanus Vaccine (adult)10 Dose with Vvm Vaccine for Human Medvls | $210.0K | 1.5% |
| 5 | Material No 44103144 Mb Adult 360kitv1 Int Mfg Licence No Kd 390 | $168.8K | 1.2% |
| 6 | Diptheria and Tetanus Dt Tetanus Anddiphtheria Td Adsorbed for Adults Andadoles… | $167.1K | 1.2% |
| 7 | Zebu Paracetamol BP 325 MG Diclofenac Sodium BP 50 MG Caffeine Anhy BP 30 MG Ta… | $153.7K | 1.1% |
| 8 | Harmless Medicine for Human Consumption(funbact A) Other Details As Per Invoice… | $136.2K | 1.0% |
| 9 | Harmless Pharmaceutical Medicines Finished Formulation Cefixime Dispersible 200… | $120.1K | 0.9% |
| 10 | Artesunate for Injection 180 MG Start 180 As Per Inv | $114.5K | 0.8% |
| 11 | Ketazol Cream 15g E/f/p (s/r) | $112.8K | 0.8% |
| 12 | Dolutegravir Sodium 10mg Tablet for Oral Suspension (dispersible Tablets) - Dru… | $107.6K | 0.8% |
| 13 | Tramadol Hydrochloride Injection (dozem)( Ml) | $102.9K | 0.7% |
| 14 | Cypro 500 Tablets Each Film Coated Tablets Contains Ciprofloxacin Hcl USP Eq to… | $93.9K | 0.7% |
| 15 | Non Harmfull Pharmaceutical Products for Human Consumption Ibuprofene 400tablet… | $88.0K | 0.6% |
India exports 20+ pharmaceutical formulations to Congo Dr with a combined trade value of $14.1M. Key products include Ethambutol ($565K), Paracetamol ($154K), Artesunate ($115K), Dolutegravir ($108K), Tramadol ($103K). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 1,952 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Congo Dr Trade Routes
These are the top pharmaceutical products exported from India to Congo Dr, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Congo Dr buyers, regulatory requirements, and logistics for that specific product corridor. Products include Ethambutol ($565K), Paracetamol ($154K), Artesunate ($115K), Dolutegravir ($108K), Tramadol ($103K) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Congo Dr
95 Indian pharmaceutical companies export finished formulations to Congo Dr. Leading exporters include Shalina Laboratories Private Limited, Biological E Limited, Biomatrix Healthcare Private Limited, Aura Pharmaceuticals Private Limited. The top exporter accounts for 14.7% of total India–Congo Dr pharma exports. Source: Indian Customs (DGFT).
Top Congo Dr Buyers from India
80 companies in Congo Dr import pharmaceutical formulations from India. Top buyers include Shalina Healthcare Sarl, Unique Pharma, Pev Lmte Ministere De La Sante, Ste Prince Pharma Sarl. The largest buyer accounts for 14.7% of India–Congo Dr pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Congo Dr Pharmaceutical Shipments
Indian Export Ports
Congo Dr Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Congo Dr Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and the Democratic Republic of Congo (Congo DR) has experienced significant growth over the past decade. In 2024, India exported pharmaceutical products worth $149.98 million to Congo DR, marking a substantial increase from previous years. This surge reflects India's expanding role as a key supplier of essential medicines to the African continent.
The trade relationship is characterized by a diverse range of products, including vaccines, antibiotics, and other essential medicines. The consistent growth trajectory underscores the strengthening bilateral ties and the increasing reliance of Congo DR on Indian pharmaceutical exports to meet its healthcare needs.
2India's Market Position
India holds a significant share in Congo DR's pharmaceutical import market, positioning itself as a primary supplier of essential medicines. In 2024, pharmaceutical products accounted for $149.98 million of Congo DR's total imports from India, indicating a strong market presence.
This dominance is strategic, as India's pharmaceutical industry is renowned for its cost-effectiveness and adherence to international quality standards, making it a preferred partner for Congo DR's healthcare sector.
3Recent Developments
Between 2024 and 2026, the India-Congo DR pharmaceutical trade witnessed several key developments. Notably, in June 2020, Congo DR's government suspended all import duties on medical products and pharmaceutical equipment for six months, aiming to enhance the availability of essential medicines.
Additionally, the Congolese Pharmaceutical Regulatory Authority (ACOREP) has been actively streamlining import procedures to facilitate smoother trade flows. These regulatory adjustments have positively impacted the bilateral trade, fostering a more conducive environment for pharmaceutical exports from India to Congo DR.
Congo Dr Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to register products in Congo DR must navigate a structured process overseen by the Congolese Pharmaceutical Regulatory Authority (ACOREP). The registration requires submission of a comprehensive dossier, typically in the Common Technical Document (CTD) format, which includes detailed information on the product's quality, safety, and efficacy.
While Good Manufacturing Practice (GMP) certification is officially mandated, enforcement can be inconsistent. A Certificate of Pharmaceutical Product (CPP) is a critical document in the registration process. Quality control measures include initial screening using methods like the GPHF Minilab, with more comprehensive analysis performed at WHO-prequalified laboratories abroad.
The entire process, from clinical trial application to market entry, is managed by ACOREP, which aims to provide a decision on clinical trial applications within 30 days of submission, following ethics committee approval. Failure to meet regulations, including the mandatory use of French on all documentation and labeling, can lead to significant sanctions, mirroring global standards that include product recalls and fines.
2GMP & Facility Requirements
Congo DR requires that Indian manufacturing sites adhere to Good Manufacturing Practice (GMP) standards to ensure the quality and safety of pharmaceutical products. The inspection process involves a thorough evaluation of manufacturing facilities, including assessments of production processes, quality control measures, and compliance with international standards.
While mutual recognition agreements may exist, the enforcement of GMP standards can vary. Therefore, Indian manufacturers must ensure strict compliance with Congo DR's regulatory requirements to facilitate smooth market entry and maintain product integrity.
3Import Documentation
Importing pharmaceutical products into Congo DR necessitates obtaining an import license from an authorized commercial bank. Shipments valued over a certain threshold require a pre-shipment inspection by Bureau Veritas. A Certificate of Pharmaceutical Product (CPP) is essential, and while GMP certification is officially required, enforcement is often inconsistent.
Quality control involves initial screening using methods like the GPHF Minilab, with more comprehensive analysis performed at WHO-prequalified laboratories abroad. The entire process, from clinical trial application to market entry, is managed by ACOREP, which aims to provide a decision on clinical trial applications within 30 days of submission, following ethics committee approval.
Product Categories & Therapeutic Trends — India to Congo Dr
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Congo DR are predominantly in the form of finished pharmaceutical formulations, including vaccines, antibiotics, and other essential medicines. These products align with Congo DR's healthcare needs, addressing prevalent diseases and conditions. The dominance of vaccines, such as the DTP HEPB HIB Pentavalent Vaccine, underscores the critical role of immunization in Congo DR's public health strategy.
The focus on finished formulations is strategic, as Congo DR's limited local manufacturing capacity necessitates reliance on imported ready-to-use medicines to meet the healthcare demands of its population.
2Emerging Opportunities
The evolving healthcare landscape in Congo DR presents emerging opportunities for Indian pharmaceutical exporters. The demand for biosimilars is on the rise, driven by the need for affordable treatment options for chronic diseases. Additionally, the government's focus on expanding healthcare infrastructure and improving access to essential medicines creates a favorable environment for Indian exporters to introduce new therapeutic areas and innovative products.
Patent expirations in key therapeutic segments further open avenues for generic drug manufacturers to enter the market, offering cost-effective alternatives to branded medications.
3Demand Drivers
Congo DR's substantial disease burden, including high incidences of infectious diseases, drives the demand for pharmaceutical imports. The aging population and increasing prevalence of non-communicable diseases further contribute to the rising need for diverse medical treatments. Healthcare spending trends indicate a growing allocation of resources towards improving health services, thereby enhancing the import demand for quality pharmaceutical products from India.
Trade Policy & Tariff Intelligence — India and Congo Dr
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
In June 2020, Congo DR's government suspended all import duties on medical products and pharmaceutical equipment for six months, aiming to enhance the availability of essential medicines.
While this suspension was temporary, it highlights the government's commitment to facilitating the import of critical healthcare products. Indian pharmaceutical exporters should monitor such policy changes to optimize their market strategies and ensure compliance with Congo DR's evolving trade regulations.
2Trade Agreements
Congo DR has engaged in various trade agreements to bolster its economic ties, including those with the European Union and neighboring African countries. While specific free trade agreements (FTAs) with India are not prominently documented, the existing trade relations and Congo DR's participation in regional economic communities suggest a conducive environment for pharmaceutical trade.
Ongoing negotiations and policy reforms may further enhance bilateral trade, presenting opportunities for Indian pharmaceutical exporters to expand their footprint in Congo DR.
3IP & Patent Landscape
Congo DR's intellectual property (IP) framework aligns with international standards, providing protection for pharmaceutical patents and data exclusivity. However, the enforcement of IP rights can be inconsistent, which may impact the market dynamics for Indian generic drug manufacturers.
The potential for compulsory licensing exists, particularly in public health emergencies, which could influence the entry strategies of Indian exporters. Staying informed about Congo DR's IP policies is crucial for navigating the competitive landscape effectively.
Supply Chain & Logistics — India to Congo Dr Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Major sea routes from Indian ports to Congo DR include the Indian Ocean, passing through the Red Sea and the Suez Canal, leading to ports such as Matadi and Kinshasa. Air routes connect major Indian airports to Congo DR's international airports, facilitating faster delivery of pharmaceutical products.
Transit times vary based on the shipping method and specific routes chosen. Potential disruptions, such as geopolitical tensions in the Red Sea region, can impact shipping schedules and costs. Indian exporters should monitor these factors to ensure timely and cost-effective delivery.
2Port Infrastructure
In India, major export ports include Jawaharlal Nehru Port Trust (JNPT), which handles a significant portion of pharmaceutical exports, and Ahmedabad ICD. In Congo DR, key import ports are Matadi, Kinshasa, and the port of Matadi. These ports are equipped to handle pharmaceutical shipments, though infrastructure quality may vary.
Efficient port operations are essential for maintaining the integrity and timely delivery of pharmaceutical products. Indian exporters should collaborate with reliable logistics partners to navigate port procedures effectively.
3Cold Chain & Compliance
Maintaining the cold chain is critical for pharmaceutical products, especially vaccines and biologics. Indian exporters must ensure that their logistics partners adhere to Good Distribution Practice (GDP) standards, including temperature-controlled storage and transportation.
Compliance with Congo DR's import regulations, including proper documentation and labeling in French, is mandatory. Failure to adhere to these standards can result in delays, fines, or product recalls.
FAQ — India to Congo Dr Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Congo Dr?
India exported pharmaceuticals worth $14.1M to Congo Dr across 1,952 verified shipments.
Who are the top Indian pharmaceutical exporters to Congo Dr?
1. SHALINA LABORATORIES PRIVATE LIMITED — $2.1M. 2. BIOLOGICAL E LIMITED — $1.6M. 3. BIOMATRIX HEALTHCARE PRIVATE LIMITED — $1.3M. Total: 95 suppliers.
Which companies in Congo Dr import pharmaceuticals from India?
1. SHALINA HEALTHCARE SARL — $2.1M. 2. UNIQUE PHARMA — $1.9M. 3. PEV LMTE MINISTERE DE LA SANTE — $1.5M. 80 buyers total.
What pharmaceutical products does India export most to Congo Dr?
1. Dtp Hepb Hib Pentavalent Vaccine Fullyliquid Combination Vaccine 10 Dose Vialpa… ($1.5M, 10.5%); 2. Dtp-hepb-hib (pentavalent Vaccine)fullyliquid Combination Vaccine 10 Dose (5ml)… ($752.4K, 5.3%); 3. Ethambutol Hydrochloride Dispersible Tablets 100mg Pack28160 ($564.6K, 4.0%); 4. Diphtheria & Tetanus Vaccine (adult)10 Dose with Vvm Vaccine for Human Medvls ($210.0K, 1.5%); 5. Material No 44103144 Mb Adult 360kitv1 Int Mfg Licence No Kd 390 ($168.8K, 1.2%)
Which ports handle pharmaceutical shipments from India to Congo Dr?
Export: JNPT, Hyderabad Air Cargo, AHEMDABAD ICD, HYDERABAD AIR, SAHAR AIR. Import: MATADI, Matadi, KINSHASA, DAR ES SALAAM, Kinshasa.
Why does Congo Dr import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $14.1M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Congo Dr?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Congo Dr pharmaceutical trade?
$7.2K per consignment across 1,952 shipments.
How many Indian pharmaceutical companies export to Congo Dr?
95 Indian companies. Largest: SHALINA LABORATORIES PRIVATE LIMITED with $2.1M.
How can I find verified Indian pharmaceutical suppliers for Congo Dr?
TransData Nexus covers 95 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Congo Dr Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 1,952 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Congo Dr identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 1,952 verified shipments from 95 Indian exporters to 80 Congo Dr buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
95 Exporters
80 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists