India to Chile Pharmaceutical Export
Bilateral Trade Intelligence · $230.4M Total Trade · 261 Exporters · 512 Buyers · DGFT Verified · Updated March 2026
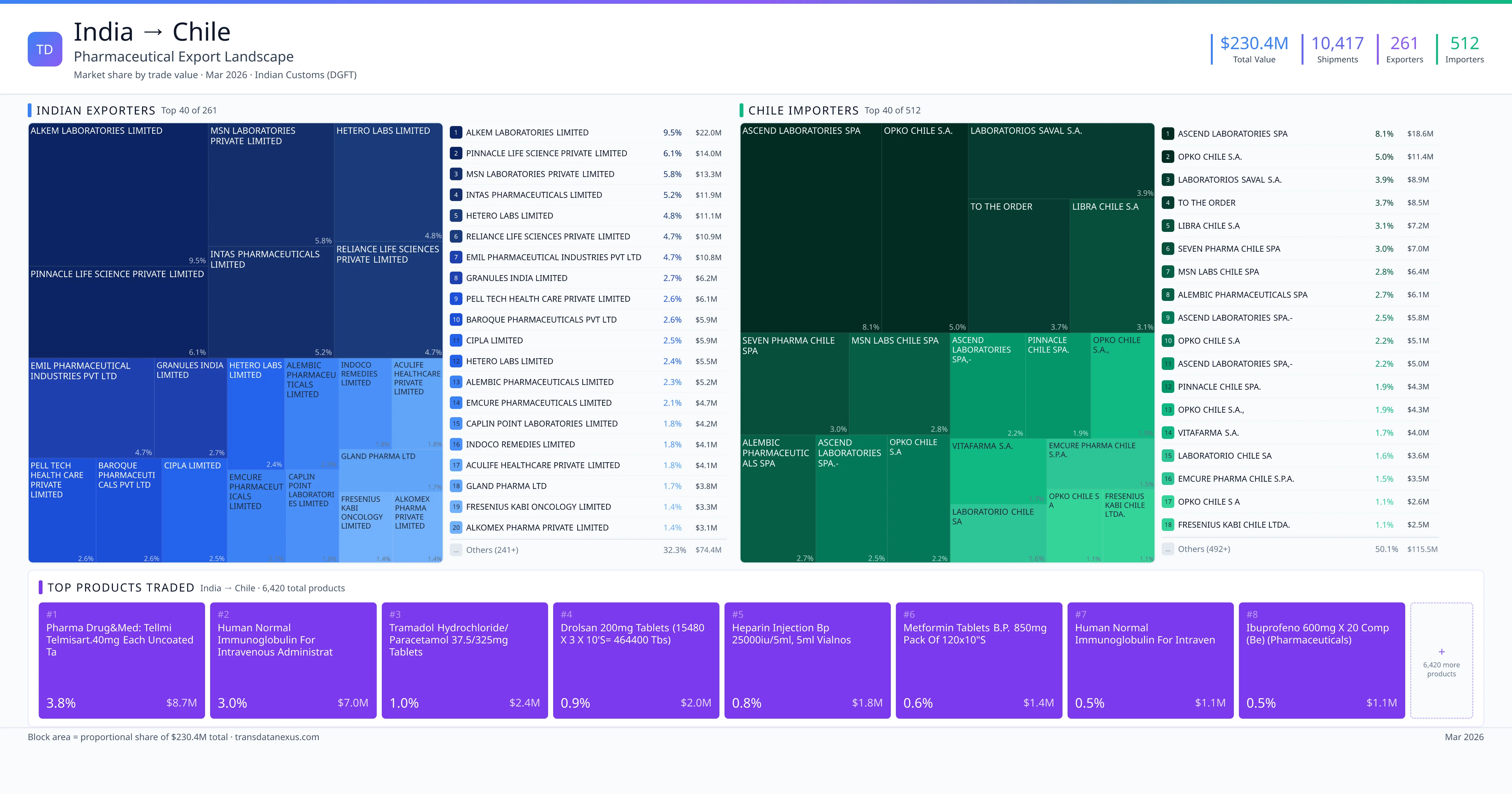
India exported $230.4M worth of pharmaceutical formulations to Chile across 10,417 verified shipments, sourced from 261 Indian exporters supplying 512 Chile buyers. The top exporters are ALKEM LABORATORIES LIMITED ($22.0M) and PINNACLE LIFE SCIENCE PRIVATE LIMITED ($14.0M). The leading products are Immunoglobulin ($3.3M) and Tramadol ($2.4M). Average shipment value: $22.1K.
Top Pharmaceutical Formulations — India to Chile
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Pharma Drug&med: Tellmi Telmisart.40mg Each Uncoated Tablet Contains: Telmisart… | $8.7M | 3.8% |
| 2 | Human Normal Immunoglobulin for Intravenous Administration B.p.5% Solution 100ml | $3.3M | 1.5% |
| 3 | Tramadol Hydrochloride/ Paracetamol 37.5/325mg Tablets Pack of 3x10 (each Coate… | $2.4M | 1.0% |
| 4 | Drolsan 200mg Tablets (15480 X 3 X 10's= 464400 Tbs) | $2.0M | 0.9% |
| 5 | Heparin Injection BP 25000iu/5ml, 5ml Vialnos | $1.8M | 0.8% |
| 6 | Human Normal Immunoglobulin for Intravenous Administration B.p.5% Solution 100m… | $1.6M | 0.7% |
| 7 | Metformin Tablets B.p. 850mg Pack of 120x10"s | $1.4M | 0.6% |
| 8 | Human Normal Immunoglobulin for Intravenous Administration B P 5 Solution 100ml… | $1.3M | 0.5% |
| 9 | Human Normal Immunoglobulin for Intraven | $1.1M | 0.5% |
| 10 | Ibuprofeno 600mg X 20 Comp (be) (pharmaceuticals) | $1.1M | 0.5% |
| 11 | Compresso Pap 90 Cpc | $1.0M | 0.4% |
| 12 | Urofenil Tablets | $1.0M | 0.4% |
| 13 | Heparin Injection Bp, 25000iu/5ml, 5ml Vial | $930.0K | 0.4% |
| 14 | Flusacort Salmeterol+fluticasone Propionate Inhaler | $926.9K | 0.4% |
| 15 | Venlafaxina 75mg 50x10s Capsules Ap Chl | $904.1K | 0.4% |
India exports 20+ pharmaceutical formulations to Chile with a combined trade value of $230.4M. Key products include Immunoglobulin ($3.3M), Tramadol ($2.4M), Heparin ($1.8M), Metformin ($1.4M), Salmeterol ($927K). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 10,417 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Chile Trade Routes
These are the top pharmaceutical products exported from India to Chile, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Chile buyers, regulatory requirements, and logistics for that specific product corridor. Products include Immunoglobulin ($3.3M), Tramadol ($2.4M), Heparin ($1.8M), Metformin ($1.4M), Salmeterol ($927K) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Chile
261 Indian pharmaceutical companies export finished formulations to Chile. Leading exporters include Alkem Laboratories Limited, Pinnacle Life Science Private Limited, Msn Laboratories Private Limited, Intas Pharmaceuticals Limited. The top exporter accounts for 9.5% of total India–Chile pharma exports. Source: Indian Customs (DGFT).
Top Chile Buyers from India
512 companies in Chile import pharmaceutical formulations from India. Top buyers include Ascend Laboratories Spa, Opko Chile S.a., Laboratorios Saval S.a., To The Order. The largest buyer accounts for 8.1% of India–Chile pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Chile Pharmaceutical Shipments
Indian Export Ports
Chile Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Chile Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and Chile has experienced significant growth over the past decade. In 2024, India's pharmaceutical exports to Chile reached approximately $200.57 million, marking a substantial increase from previous years. This upward trajectory reflects the strengthening economic ties and the growing demand for affordable, high-quality medicines in Chile. The expansion is also attributed to India's competitive manufacturing capabilities and adherence to international quality standards, which have bolstered its position in the global pharmaceutical market.
Key milestones in this bilateral trade include the signing of the Partial Scope Trade Agreement in 2007, which was deepened in 2017, providing tariff preferences ranging from 20% to 100% for pharmaceutical products. This agreement has facilitated smoother trade flows and enhanced market access for Indian pharmaceutical companies.
2India's Market Position
India holds a significant share in Chile's pharmaceutical import market, ranking third or fourth among Chile's pharmaceutical import sources as of 2023. This position underscores India's strategic importance in supplying essential medicines to Chile. The country's robust pharmaceutical industry, characterized by a vast array of generic medicines and adherence to international quality standards, makes it a preferred partner for Chilean importers seeking cost-effective and reliable pharmaceutical products.
3Recent Developments
In February 2024, the Pharmaceuticals Export Promotion Council of India (Pharmexcil) hosted the iPHEX-LATAM event in Guatemala, Colombia, and Chile. This event featured a delegation of 100 business representatives from 78 Indian pharmaceutical companies, aiming to forge connections between top Indian and Latin American pharmaceutical firms. Such initiatives are expected to further enhance bilateral trade relations and open new avenues for collaboration in the pharmaceutical sector.
Chile Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to register products in Chile must comply with the regulations set by the Public Health Institute (Instituto de Salud Pública or ISP), the regulatory authority responsible for controlling pharmaceutical products in Chile. The registration process involves submitting detailed information about the pharmaceutical products, including their composition, intended use, packaging, labeling, and manufacturer information. Additionally, the products must meet the World Health Organization's Good Manufacturing Practices (WHO-GMP) standards to ensure quality and safety. (britcham.cl)
The registration dossier should be prepared in the Common Technical Document (CTD) format, which is internationally recognized and facilitates the evaluation process. The timeline for registration can vary, but it typically involves a thorough review by the ISP, which may take several months. Therefore, Indian exporters should plan accordingly and ensure that all documentation is complete and accurate to expedite the approval process.
2GMP & Facility Requirements
Chile requires that pharmaceutical products imported into the country comply with Good Manufacturing Practices (GMP) standards. The ISP is responsible for implementing and enforcing GMP compliance, conducting inspections, and ensuring that pharmaceutical manufacturers adhere to the prescribed guidelines. Indian manufacturing sites must demonstrate adherence to these standards, which may involve providing evidence of GMP certification from recognized authorities.
The inspection process includes evaluating the manufacturing facilities, quality control measures, and documentation practices to ensure that the products meet the required quality and safety standards. Mutual recognition agreements between regulatory authorities can streamline this process, but Indian exporters should be prepared for potential inspections and ensure that their facilities are compliant with both Indian and international GMP standards.
3Import Documentation
Importing pharmaceutical products into Chile requires obtaining authorization from the ISP. The authorization process involves submitting detailed information about the pharmaceutical products, including their composition, intended use, packaging, labeling, and manufacturer information. Additionally, importers must provide certificates such as the Certificate of Pharmaceutical Product (CPP), Certificate of Analysis (CoA), and evidence of GMP compliance. (britcham.cl)
Customs procedures for pharmaceutical imports into Chile include submitting the necessary documentation to the ISP for approval, followed by clearance through Chilean customs. It is essential for importers to adhere to all regulations and requirements set forth by the Chilean health authorities to avoid any potential issues or delays in the importation process.
Product Categories & Therapeutic Trends — India to Chile
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
The dominant therapeutic areas in India's pharmaceutical exports to Chile include drugs for the nervous system, anti-infectives for systemic use, and medications for the gastrointestinal tract and metabolism. In 2022, these categories accounted for the majority of pharmaceutical sales in Chile, with drugs for the nervous system at $486.4 million, anti-infectives for systemic use at $447.7 million, and medications for the gastrointestinal tract and metabolism at $571.3 million.
This trend aligns with Chile's healthcare needs, as these therapeutic areas address prevalent health concerns in the country. The demand for such medications presents a significant opportunity for Indian pharmaceutical exporters to cater to the Chilean market by providing cost-effective and high-quality products in these categories.
2Emerging Opportunities
The global pharmaceutical industry is witnessing patent expirations, leading to increased demand for generic medicines. Additionally, the rising prevalence of chronic diseases and the need for affordable healthcare solutions are driving the demand for biosimilars and other therapeutic areas. Indian pharmaceutical exporters can leverage these trends by focusing on developing and supplying generic versions of off-patent drugs and biosimilars to the Chilean market.
By aligning their product portfolios with these emerging opportunities, Indian exporters can strengthen their presence in Chile and contribute to meeting the country's evolving healthcare needs.
3Demand Drivers
Chile's disease burden, aging population, and increasing healthcare spending are significant drivers of import demand for pharmaceutical products. The country's healthcare system is under pressure to provide effective treatments for a growing number of chronic diseases and age-related health issues. This situation creates a substantial market for pharmaceutical imports, particularly for cost-effective and high-quality medicines.
The Chilean government's efforts to improve healthcare infrastructure and expand insurance coverage further contribute to the rising demand for pharmaceutical products. Indian exporters can capitalize on these trends by offering a diverse range of products that meet the needs of the Chilean population.
Trade Policy & Tariff Intelligence — India and Chile
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
The Partial Scope Trade Agreement between India and Chile, signed in 2007 and deepened in 2017, provides tariff preferences ranging from 20% to 100% for pharmaceutical products. This agreement has facilitated smoother trade flows and enhanced market access for Indian pharmaceutical companies.
Additionally, the Most Favored Nation (MFN) tariff rates and preferential rates under the agreement further reduce the duty burden on pharmaceutical imports from India. These favorable tariff structures make Indian pharmaceutical products more competitive in the Chilean market.
2Trade Agreements
The Partial Scope Trade Agreement between India and Chile, signed in 2007 and deepened in 2017, provides tariff preferences ranging from 20% to 100% for pharmaceutical products. This agreement has facilitated smoother trade flows and enhanced market access for Indian pharmaceutical companies.
Current or upcoming negotiations may further enhance bilateral trade relations and open new avenues for collaboration in the pharmaceutical sector. Staying informed about these developments is crucial for Indian exporters to leverage potential benefits.
3IP & Patent Landscape
Chile's patent provisions, data exclusivity, and compulsory licensing policies can impact the entry of Indian generics into the market. Understanding these aspects is essential for Indian pharmaceutical companies to navigate the Chilean market effectively. While Chile has a robust intellectual property framework, there are provisions for compulsory licensing under specific circumstances, such as public health emergencies. Indian exporters should monitor these regulations to ensure compliance and identify opportunities for market entry.
Supply Chain & Logistics — India to Chile Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Major sea routes from Indian ports to Chile include the Pacific Ocean route, with transit times varying depending on the specific ports of departure and arrival. Air routes are also utilized, with transit times typically ranging from 20 to 30 hours, including layovers. It's important to note that geopolitical events, such as disruptions in the Red Sea, can affect shipping routes and transit times. Exporters should stay updated on global events that may impact logistics and plan shipments accordingly.
2Port Infrastructure
Key export ports in India include Nhava Sheva Sea (INNSA1), Delhi Air, and JNPT, which collectively handle a significant portion of pharmaceutical exports. In Chile, major import ports are San Antonio, Santiago de Chile, and Valparaiso. These ports are equipped to handle pharmaceutical shipments, ensuring efficient customs clearance and distribution within the country.
3Cold Chain & Compliance
Temperature-controlled logistics are essential for maintaining
FAQ — India to Chile Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Chile?
India exported pharmaceuticals worth $230.4M to Chile across 10,417 verified shipments.
Who are the top Indian pharmaceutical exporters to Chile?
1. ALKEM LABORATORIES LIMITED — $22.0M. 2. PINNACLE LIFE SCIENCE PRIVATE LIMITED — $14.0M. 3. MSN LABORATORIES PRIVATE LIMITED — $13.3M. Total: 261 suppliers.
Which companies in Chile import pharmaceuticals from India?
1. ASCEND LABORATORIES SPA — $18.6M. 2. OPKO CHILE S.A. — $11.4M. 3. LABORATORIOS SAVAL S.A. — $8.9M. 512 buyers total.
What pharmaceutical products does India export most to Chile?
1. Pharma Drug&med: Tellmi Telmisart.40mg Each Uncoated Tablet Contains: Telmisart… ($8.7M, 3.8%); 2. Human Normal Immunoglobulin for Intravenous Administration B.p.5% Solution 100ml ($3.3M, 1.5%); 3. Tramadol Hydrochloride/ Paracetamol 37.5/325mg Tablets Pack of 3x10 (each Coate… ($2.4M, 1.0%); 4. Drolsan 200mg Tablets (15480 X 3 X 10's= 464400 Tbs) ($2.0M, 0.9%); 5. Heparin Injection BP 25000iu/5ml, 5ml Vialnos ($1.8M, 0.8%)
Which ports handle pharmaceutical shipments from India to Chile?
Export: NHAVA SHEVA SEA (INNSA1), DELHI AIR, JNPT, HYDERABAD ICD, JNPT/ NHAVA SHEVA SEA. Import: San Antonio, SANTIAGO DE CHILE -, Valparaiso, SAN ANTONIO, VALPARAISO.
Why does Chile import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $230.4M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Chile?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Chile pharmaceutical trade?
$22.1K per consignment across 10,417 shipments.
How many Indian pharmaceutical companies export to Chile?
261 Indian companies. Largest: ALKEM LABORATORIES LIMITED with $22.0M.
How can I find verified Indian pharmaceutical suppliers for Chile?
TransData Nexus covers 261 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Chile Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 10,417 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Chile identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 10,417 verified shipments from 261 Indian exporters to 512 Chile buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
261 Exporters
512 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists