India to Canada Pharmaceutical Export
Bilateral Trade Intelligence · $1.1B Total Trade · 413 Exporters · 781 Buyers · DGFT Verified · Updated March 2026
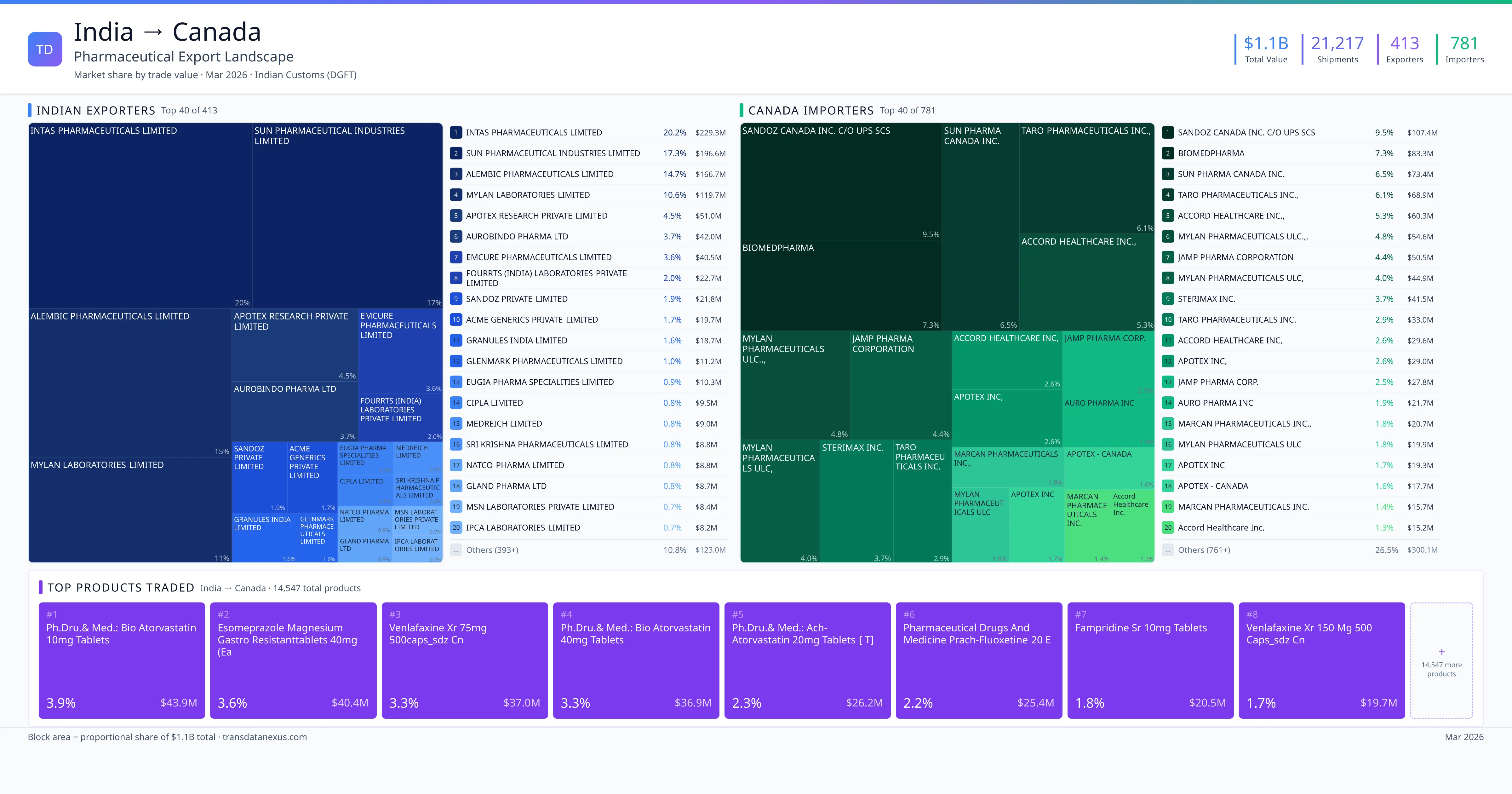
India exported $1.1B worth of pharmaceutical formulations to Canada across 21,217 verified shipments, sourced from 413 Indian exporters supplying 781 Canada buyers. The top exporters are INTAS PHARMACEUTICALS LIMITED ($229.3M) and SUN PHARMACEUTICAL INDUSTRIES LIMITED ($196.6M). The leading products are Esomeprazole ($40.4M) and Rosuvastatin ($11.7M). Average shipment value: $53.5K.
Top Pharmaceutical Formulations — India to Canada
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Ph.dru.& Med.: Bio Atorvastatin 10mg Tablets | $43.9M | 3.9% |
| 2 | Esomeprazole Magnesium Gastro Resistanttablets 40mg (each Tablet Contains: Esom… | $40.4M | 3.6% |
| 3 | Venlafaxine Xr 75mg 500caps_sdz Cn | $37.0M | 3.3% |
| 4 | Ph.dru.& Med.: Bio Atorvastatin 40mg Tablets | $36.9M | 3.3% |
| 5 | Ph.dru.& Med.: Ach-atorvastatin 20mg Tablets [ T] | $26.2M | 2.3% |
| 6 | Pharmaceutical Drugs and Medicine Prach-fluoxetine 20 Each Hard Gelatin Capsule… | $25.4M | 2.2% |
| 7 | Fampridine Sr 10mg Tablets | $20.5M | 1.8% |
| 8 | Venlafaxine Xr 150 MG 500 Caps_sdz Cn | $19.7M | 1.7% |
| 9 | Prazosin Hydrochloride Capsules USP 2mg | $18.6M | 1.6% |
| 10 | Ph.dru.& Med.: Ach-atorvastatin 40mg Tablets | $18.2M | 1.6% |
| 11 | Prazosin Hydrochloride Capsules USP 1mg | $13.5M | 1.2% |
| 12 | Ph.dru.& Med.: Ach-atorvastatin 40mg Tablets [ T] | $12.4M | 1.1% |
| 13 | Taro-rosuvastatin Tablets 40mg (each Tabcontains: Rosuvastatin Calcium Eq. To.r… | $11.7M | 1.0% |
| 14 | Pantoprazole T 40 MG 100bt Tablets Packs 53001 | $10.7M | 0.9% |
| 15 | Harmless Medicines, Pantoprazole T 40 Mg100bt Tablets (53080 Packs) | $10.6M | 0.9% |
India exports 20+ pharmaceutical formulations to Canada with a combined trade value of $1.1B. Key products include Esomeprazole ($40.4M), Rosuvastatin ($11.7M), Pantoprazole ($10.7M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 21,217 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Canada Trade Routes
These are the top pharmaceutical products exported from India to Canada, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Canada buyers, regulatory requirements, and logistics for that specific product corridor. Products include Esomeprazole ($40.4M), Rosuvastatin ($11.7M), Pantoprazole ($10.7M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Canada
413 Indian pharmaceutical companies export finished formulations to Canada. Leading exporters include Intas Pharmaceuticals Limited, Sun Pharmaceutical Industries Limited, Alembic Pharmaceuticals Limited, Mylan Laboratories Limited. The top exporter accounts for 20.2% of total India–Canada pharma exports. Source: Indian Customs (DGFT).
Top Canada Buyers from India
781 companies in Canada import pharmaceutical formulations from India. Top buyers include Sandoz Canada Inc. C/o Ups Scs, Biomedpharma, Sun Pharma Canada Inc., Taro Pharmaceuticals Inc.,. The largest buyer accounts for 9.5% of India–Canada pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Canada Pharmaceutical Shipments
Indian Export Ports
Canada Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Canada Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The India-Canada pharmaceutical trade corridor has experienced significant growth over the past decade, evolving from modest exchanges to a robust and dynamic partnership. In the fiscal year 2024–25, India's pharmaceutical exports to Canada reached approximately $540 million, marking a substantial increase from previous years. This growth trajectory underscores the expanding demand for Indian pharmaceutical products in the Canadian market.
Key milestones in this bilateral trade relationship include the successful completion of a historic trade mission to India in November 2017, which facilitated new partnerships between Indian and Canadian businesses. (canada.ca) Additionally, the launch of the Comprehensive Economic Partnership Agreement (CEPA) negotiations in March 2026 signifies a strategic effort to further enhance economic ties, with a target to increase bilateral trade to $50 billion by 2030.
2India's Market Position
India holds a significant position in Canada's pharmaceutical import landscape. In the fiscal year 2024–25, India's pharmaceutical exports to Canada were valued at approximately $540 million, contributing to a total bilateral trade of $8.6 billion. This positions India as a key supplier, reflecting the strategic importance of this trade corridor for Canada.
3Recent Developments
Between 2024 and 2026, several developments have influenced the India-Canada pharmaceutical trade corridor. Notably, in March 2026, the two nations agreed to strengthen their economic partnership by finalizing the CEPA negotiations, aiming to increase bilateral trade to $50 billion by 2030. This agreement is expected to further enhance the trade of pharmaceutical products, among other sectors, by reducing trade barriers and improving market access.
## Highlights:
Trade Policy & Tariff Intelligence — India and Canada
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
As of January 1, 2026, Canada maintains a Most-Favored-Nation (MFN) tariff rate of zero for pharmaceutical products classified under HS codes 3003 and 3004, which encompass finished pharmaceutical formulations such as tablets, capsules, syrups, and injections. This zero-duty rate applies to imports from all World Trade Organization (WTO) member countries, including India. Therefore, Indian pharmaceutical exports to Canada benefit from this preferential tariff treatment.
In addition to the MFN rate, Canada has established preferential duty rates for certain trade partners through various trade agreements. However, as of March 2026, there are no specific preferential duty rates for Indian pharmaceutical products under any existing trade agreements. Consequently, Indian pharmaceutical exports to Canada are subject to the standard MFN tariff rates.
2Trade Agreements
Currently, there is no Free Trade Agreement (FTA) between India and Canada. However, in November 2025, the Prime Ministers of both countries announced their intention to launch negotiations toward a Comprehensive Economic Partnership Agreement (CEPA). This agreement aims to enhance bilateral trade and investment, potentially impacting sectors such as pharmaceuticals. Public consultations were conducted from December 13, 2025, to January 27, 2026, to gather input from stakeholders on the proposed CEPA. As of March 2026, discussions are ongoing, and no formal agreement has been finalized. (international.canada.ca)
3IP & Patent Landscape
Canada's intellectual property (IP) framework, particularly concerning patents, significantly influences the entry of generic drugs into the market. The country adheres to international IP standards, including those set by the World Trade Organization's Trade-Related Aspects of Intellectual Property Rights (TRIPS) agreement. This adherence provides patent holders with exclusive rights, typically lasting 20 years from the filing date. Generic manufacturers, including those from India, must navigate this exclusivity period, which can delay the introduction of generic versions of patented drugs. Additionally, Canada's regulatory environment includes provisions for compulsory licensing under specific circumstances, allowing the government to authorize the production of a patented product without the consent of the patent holder, primarily to address public health needs. However, such measures are rarely invoked and are subject to stringent conditions. Therefore, Indian generic pharmaceutical companies must carefully assess the patent landscape and regulatory environment to strategize their market entry into Canada effectively.
FAQ — India to Canada Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Canada?
India exported pharmaceuticals worth $1.1B to Canada across 21,217 verified shipments.
Who are the top Indian pharmaceutical exporters to Canada?
1. INTAS PHARMACEUTICALS LIMITED — $229.3M. 2. SUN PHARMACEUTICAL INDUSTRIES LIMITED — $196.6M. 3. ALEMBIC PHARMACEUTICALS LIMITED — $166.7M. Total: 413 suppliers.
Which companies in Canada import pharmaceuticals from India?
1. SANDOZ CANADA INC. C/O UPS SCS — $107.4M. 2. BIOMEDPHARMA — $83.3M. 3. SUN PHARMA CANADA INC. — $73.4M. 781 buyers total.
What pharmaceutical products does India export most to Canada?
1. Ph.dru.& Med.: Bio Atorvastatin 10mg Tablets ($43.9M, 3.9%); 2. Esomeprazole Magnesium Gastro Resistanttablets 40mg (each Tablet Contains: Esom… ($40.4M, 3.6%); 3. Venlafaxine Xr 75mg 500caps_sdz Cn ($37.0M, 3.3%); 4. Ph.dru.& Med.: Bio Atorvastatin 40mg Tablets ($36.9M, 3.3%); 5. Ph.dru.& Med.: Ach-atorvastatin 20mg Tablets [ T] ($26.2M, 2.3%)
Which ports handle pharmaceutical shipments from India to Canada?
Export: SAHAR AIR, NHAVA SHEVA SEA, DELHI AIR, COCHIN SEA, KHATUWAS ICD. Import: TORONTO, Toronto, Montreal, MONTREAL, MONTREAL-DORAVAL APT.
Why does Canada import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $1.1B corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Canada?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Canada pharmaceutical trade?
$53.5K per consignment across 21,217 shipments.
How many Indian pharmaceutical companies export to Canada?
413 Indian companies. Largest: INTAS PHARMACEUTICALS LIMITED with $229.3M.
How can I find verified Indian pharmaceutical suppliers for Canada?
TransData Nexus covers 413 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Canada Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 21,217 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Canada identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 21,217 verified shipments from 413 Indian exporters to 781 Canada buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
413 Exporters
781 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists