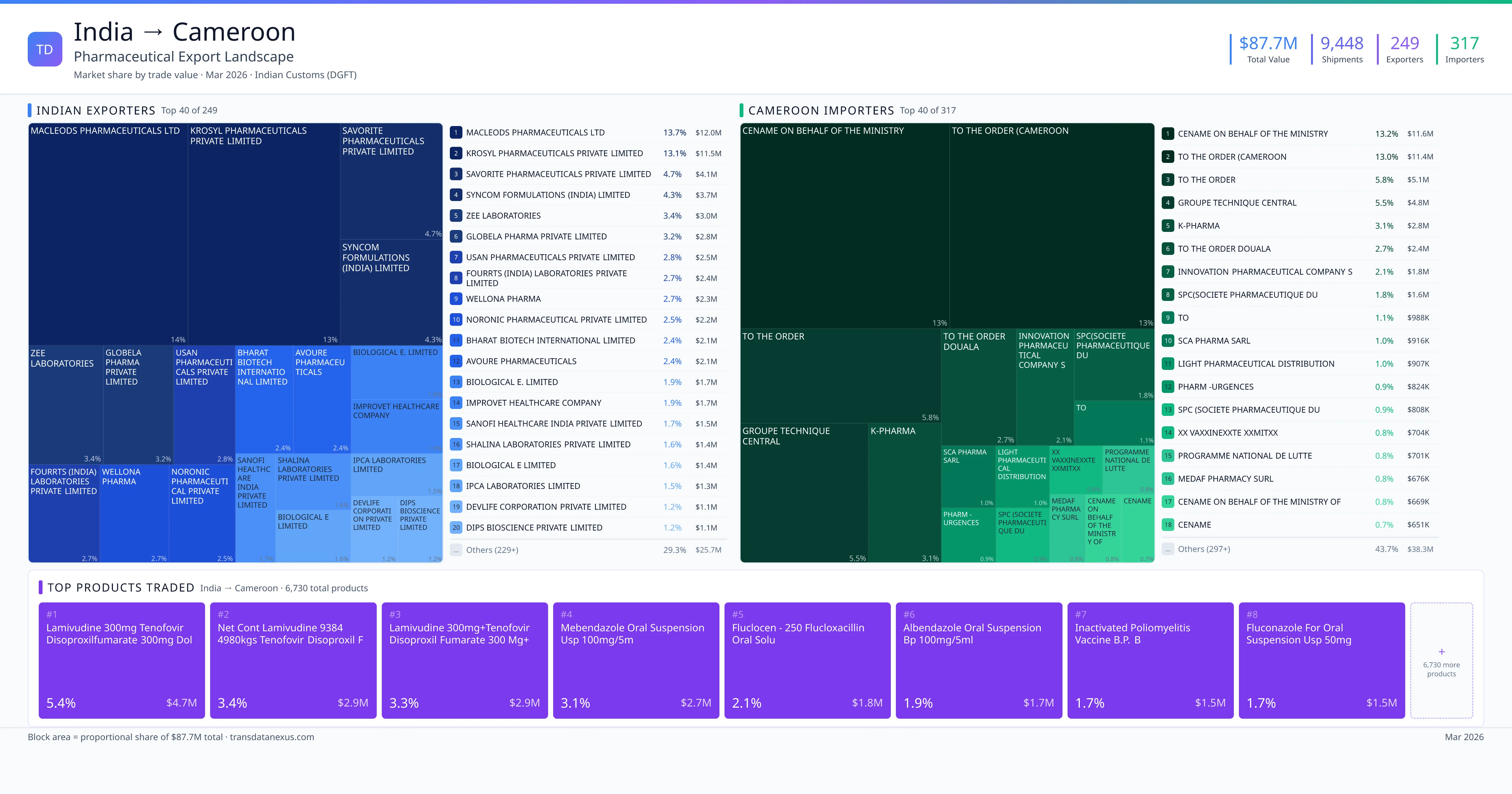
Indian pharmaceutical companies seeking to export finished formulations to Cameroon must navigate a structured registration process overseen by the Cameroon Agency for Food and Drugs Administration (CAFDA). This process ensures that all imported pharmaceutical products meet the country's standards for quality, safety, and efficacy.
Dossier Requirements and Submission
The registration dossier must be comprehensive, typically adhering to the Common Technical Document (CTD) format, which is internationally recognized and facilitates the evaluation process. The dossier should include:
- Administrative Information: Details of the applicant, manufacturing sites, and authorized representatives.
- Quality Documentation: Information on the drug substance and drug product, including manufacturing processes, quality control measures, and stability data.
- Non-Clinical and Clinical Data: Preclinical and clinical study reports demonstrating the product's safety and efficacy.
- Labeling and Packaging: Proposed labeling, packaging materials, and patient information leaflets.
The dossier must be submitted in French, Cameroon’s official language, to ensure clarity and facilitate the review process. While the CTD format is standard, applicants should verify if the eCTD (electronic Common Technical Document) format is accepted, as electronic submissions can expedite the evaluation process.
Bioequivalence and Clinical Trials
For generic formulations, demonstrating bioequivalence to the innovator product is essential. This typically involves conducting bioequivalence studies in accordance with international guidelines. If the product has not been previously marketed in Cameroon, clinical trials may be required to establish its safety and efficacy profile. These studies must comply with Good Clinical Practice (GCP) standards and be approved by the relevant ethics committees.
Approval Timeline and Fees
The approval timeline for pharmaceutical product registration in Cameroon can vary, but it generally ranges from 6 to 12 months, depending on the completeness of the submitted dossier and the complexity of the product. Applicants should anticipate potential delays and plan accordingly. Registration fees are applicable and vary based on the product type and registration category. It is advisable to consult directly with CAFDA for the most current fee structure and any additional costs associated with the registration process.