India to Cambodia Pharmaceutical Export
Bilateral Trade Intelligence · $103.0M Total Trade · 380 Exporters · 579 Buyers · DGFT Verified · Updated March 2026
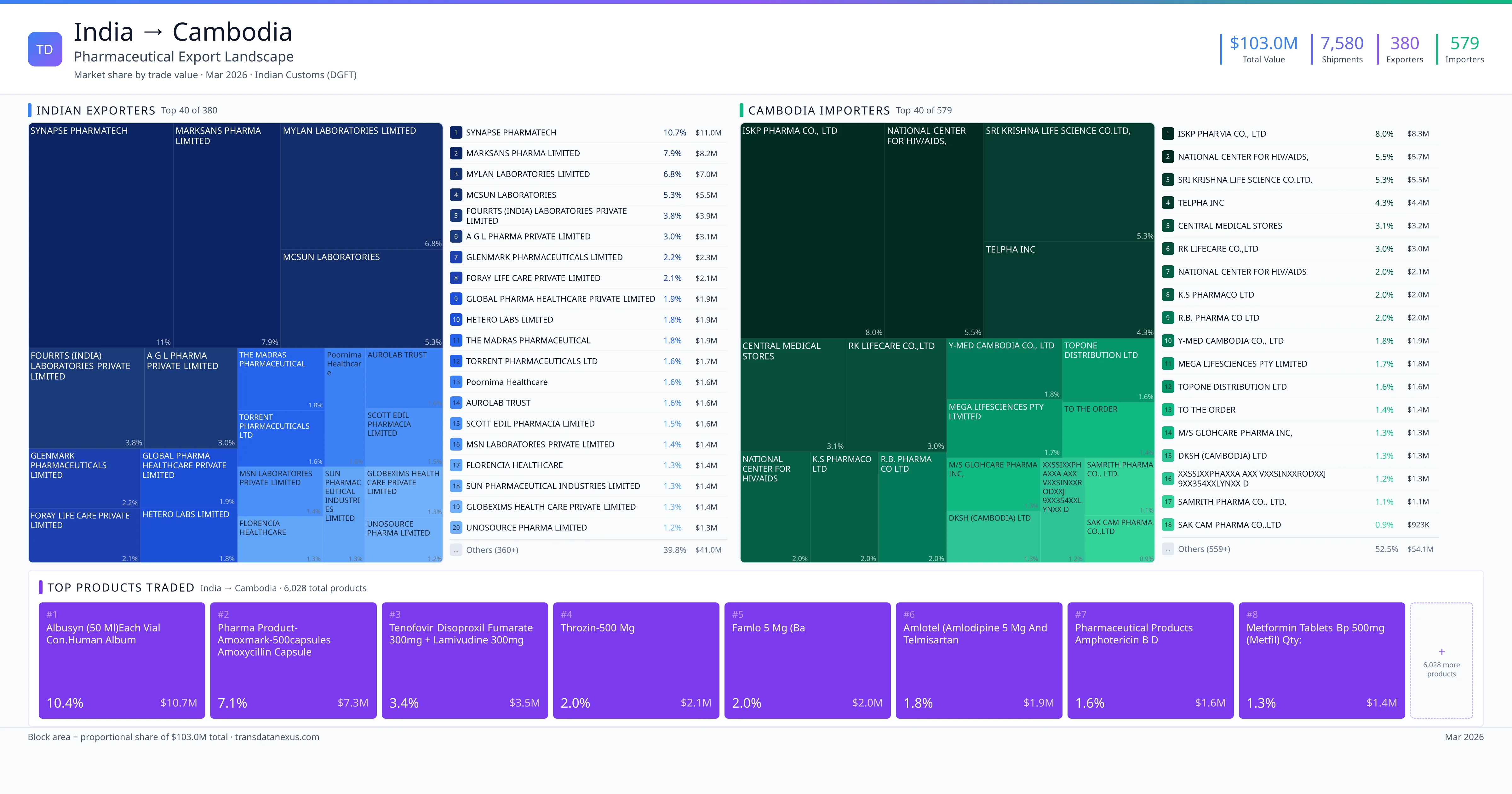
India exported $103.0M worth of pharmaceutical formulations to Cambodia across 7,580 verified shipments, sourced from 380 Indian exporters supplying 579 Cambodia buyers. The top exporters are SYNAPSE PHARMATECH ($11.0M) and MARKSANS PHARMA LIMITED ($8.2M). The leading products are Tenofovir ($3.5M) and Amlodipine ($2.0M). Average shipment value: $13.6K.
Top Pharmaceutical Formulations — India to Cambodia
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Albusyn (50 Ml)each Vial Con.human Album | $10.7M | 10.4% |
| 2 | Pharma Product-amoxmark-500capsules Amoxycillin Capsules 500 MG (each Capsule C… | $7.3M | 7.1% |
| 3 | Tenofovir Disoproxil Fumarate 300mg + Lamivudine 300mg Tablets (30s) (40000 Pac… | $3.5M | 3.4% |
| 4 | Throzin-500 MG | $2.1M | 2.0% |
| 5 | Famlo 5 MG (ba | $2.0M | 2.0% |
| 6 | Amlotel (amlodipine 5 MG and Telmisartan | $1.9M | 1.8% |
| 7 | Pharmaceutical Products Amphotericin B D | $1.6M | 1.6% |
| 8 | Metformin Tablets BP 500mg (metfil) Qty: | $1.4M | 1.3% |
| 9 | Sofosbuvir 400 MG + Daclatasvir 60 Mg, Tablets 28 (2112 Packs X 28s = 59136 ) | $1.3M | 1.2% |
| 10 | Throzin-250 MG | $1.1M | 1.0% |
| 11 | Fourts B Capsules Qty. S | $955.9K | 0.9% |
| 12 | Safix Ds B. | $947.7K | 0.9% |
| 13 | Kevixime-200 MG | $898.7K | 0.9% |
| 14 | Harmless Medicines-agesom Plus-esomprazo | $825.2K | 0.8% |
| 15 | Sofosbuvir 400 MG + Daclatasvir 60mg, Tablets 28 (13756 Packs X 28's=385168 ) | $717.8K | 0.7% |
India exports 20+ pharmaceutical formulations to Cambodia with a combined trade value of $103.0M. Key products include Tenofovir ($3.5M), Amlodipine ($2.0M), Amphotericin ($1.6M), Metformin ($1.4M), Sofosbuvir ($1.3M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 7,580 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Cambodia Trade Routes
These are the top pharmaceutical products exported from India to Cambodia, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Cambodia buyers, regulatory requirements, and logistics for that specific product corridor. Products include Tenofovir ($3.5M), Amlodipine ($2.0M), Amphotericin ($1.6M), Metformin ($1.4M), Sofosbuvir ($1.3M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Cambodia
380 Indian pharmaceutical companies export finished formulations to Cambodia. Leading exporters include Synapse Pharmatech, Marksans Pharma Limited, Mylan Laboratories Limited, Mcsun Laboratories. The top exporter accounts for 10.7% of total India–Cambodia pharma exports. Source: Indian Customs (DGFT).
Top Cambodia Buyers from India
579 companies in Cambodia import pharmaceutical formulations from India. Top buyers include Iskp Pharma Co., Ltd, National Center For Hiv/aids,, Sri Krishna Life Science Co.ltd,, Telpha Inc. The largest buyer accounts for 8.0% of India–Cambodia pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Cambodia Pharmaceutical Shipments
Indian Export Ports
Cambodia Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Cambodia Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and Cambodia has experienced significant growth over the past two decades, evolving from modest exchanges to a robust bilateral relationship. In 2024, India exported pharmaceutical products worth approximately $55.21 million to Cambodia, marking a substantial increase from previous years. This upward trajectory underscores the strengthening ties between the two nations in the healthcare sector.
Key milestones in this evolution include the establishment of the India-Cambodia Joint Working Group on Trade and Investment (JWGTI) in June 2024. The second meeting of JWGTI, held in New Delhi on June 19, 2024, focused on enhancing bilateral trade and investment, with a particular emphasis on the pharmaceutical sector. Additionally, in March 2026, a 70-member Indian pharmaceutical delegation representing 56 leading companies visited Phnom Penh to explore trade and investment opportunities, reflecting India's commitment to strengthening healthcare cooperation with Cambodia.
2India's Market Position
India holds a significant position in Cambodia's pharmaceutical import market. In 2024, Cambodia's total pharmaceutical imports were approximately $190.72 million, with India accounting for about $47.5 million, representing a substantial share of the market. This dominance is attributed to India's reputation for providing high-quality, affordable generic medicines, which are crucial for addressing Cambodia's healthcare needs. The strategic importance of this corridor is further highlighted by India's role as a major global supplier of generic medicines and vaccines, meeting over 60% of global vaccine demand. (findyournews.media)
3Recent Developments
Between 2024 and 2026, several key developments have further solidified the India-Cambodia pharmaceutical trade corridor. In June 2024, during the second meeting of the JWGTI, both nations discussed the recognition of the Indian Pharmacopoeia and potential collaboration in the pharmaceutical sector, aiming to enhance trade and investment. In November 2025, an interaction between Indian and Cambodian stakeholders in the pharmaceutical sector was organized, focusing on strengthening cooperation and partnerships, with over 180 participants from both countries. (findyournews.media) These initiatives reflect a concerted effort to deepen bilateral ties and promote mutual growth in the pharmaceutical industry.
Cambodia Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
To export finished pharmaceutical formulations to Cambodia, Indian companies must navigate a structured registration process overseen by Cambodia's Ministry of Health (MOH). This process ensures that all imported medicines meet the country's safety, efficacy, and quality standards.
Application Submission: Indian pharmaceutical companies are required to submit their product registration applications through the Cambodia Pharmaceutical Online Registration System (CamPORS). The application must include:
- Application Form: A completed form detailing the product and manufacturer information.
- Good Manufacturing Practice (GMP) Certificate: A valid GMP certificate issued by a recognized authority, such as the World Health Organization (WHO) or the Pharmaceutical Inspection Co-operation Scheme (PIC/S). This certificate must be current and accompanied by an official translation into Khmer.
- Certificate of Pharmaceutical Product (CoPP): A document confirming that the product is authorized for sale in the country of origin.
- Product Information: Detailed product information, including labeling, packaging, and a product manual.
Dossier Format: The technical dossier should align with the ASEAN Common Technical Dossier (ACTD) format, comprising:
- Part I: Administrative Information
- Part II: Quality / Pharmaceutical Data
- Part III: Non-clinical Studies
- Part IV: Clinical Efficacy & Safety
This structured approach facilitates a comprehensive evaluation of the product's quality, safety, and efficacy.
Approval Timeline and Fees: The registration process typically spans three to six months. However, this duration can extend depending on the completeness of the submitted documentation and the workload of the MOH. Applicants should anticipate potential delays and plan accordingly.
2GMP & Facility Requirements
Cambodia mandates that all pharmaceutical products imported into the country be manufactured in facilities adhering to Good Manufacturing Practice (GMP) standards. For Indian manufacturers, this entails:
- GMP Certification: Manufacturing facilities must possess a valid GMP certificate from a recognized authority, such as the WHO or PIC/S. This certification ensures that the facility's operations meet international quality standards.
- Inspection Process: The MOH may conduct inspections of foreign manufacturing facilities to verify compliance with GMP standards. These inspections assess various aspects, including facility design, equipment maintenance, staff training, and documentation practices. (fda.gov.tw)
- Mutual Recognition: Cambodia recognizes GMP certifications from countries that are members of the Pharmaceutical Inspection Co-operation Scheme (PIC/S). This mutual recognition streamlines the approval process for manufacturers from these countries.
3Import Documentation
When importing pharmaceutical products into Cambodia, Indian companies must ensure compliance with the following documentation requirements:
- Import License: An import license issued by the Cambodian MOH is mandatory for all pharmaceutical imports. This license authorizes the importation and distribution of specific pharmaceutical products within Cambodia. (cambodiantr.gov.kh)
- Certificates Required:
- Certificate of Pharmaceutical Product (CoPP): Confirms that the product is authorized for sale in the country of origin.
- Certificate of Analysis (CoA): Provides detailed information on the product's composition and quality control measures.
- GMP Certificate: Demonstrates that the manufacturing facility complies with international GMP standards.
- Customs Procedures: Imported pharmaceutical products are subject to customs clearance procedures, which include:
- Documentation Review: Customs authorities will review all submitted documents to ensure compliance with Cambodian regulations.
- Inspection: Physical inspection of the goods may be conducted to verify the product's conformity with the submitted documentation.
- Duties and Taxes: Payment of applicable import duties and taxes is required before the release of goods.
Adhering to these requirements is crucial for the successful importation and distribution of pharmaceutical products in Cambodia.
Product Categories & Therapeutic Trends — India to Cambodia
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Cambodia are predominantly concentrated in therapeutic areas addressing infectious diseases and cardiovascular conditions. The leading product, ALBUSYN (50 ML) Human Albumin, accounts for 10.4% of the total export value, underscoring the critical need for treatments related to blood volume and protein deficiencies. Amoxicillin Capsules (500 mg), representing 7.1% of exports, highlight the ongoing demand for antibiotics to combat bacterial infections. Additionally, the combination therapy of Tenofovir Disoproxil Fumarate and Lamivudine (300 mg each) tablets, comprising 3.4% of exports, indicates a significant focus on antiretroviral treatments for HIV/AIDS.
These therapeutic categories align with Cambodia's healthcare priorities, where infectious diseases, including HIV/AIDS, remain prevalent. The substantial presence of antiretroviral drugs in the export portfolio reflects the country's commitment to managing and treating HIV/AIDS effectively. The emphasis on cardiovascular medications, such as amlodipine and telmisartan combinations, addresses the rising incidence of hypertension and related cardiovascular diseases in Cambodia. This therapeutic focus is essential for mitigating the health impacts of these conditions, which are becoming increasingly common in the Cambodian population.
2Emerging Opportunities
The Cambodian pharmaceutical market is experiencing growth in several emerging product categories, particularly in over-the-counter (OTC) pharmaceuticals. The market is projected to reach a revenue of US$163.06 million in 2025, with vitamins and minerals leading the segment. This growth is driven by increased health awareness and a shift towards preventive healthcare among the Cambodian population.
Additionally, the Cambodian government's focus on improving healthcare infrastructure and expanding access to medical services presents opportunities for Indian pharmaceutical exporters to introduce a broader range of products, including generics and biosimilars. The market's reliance on imports, with over 90% of pharmaceutical products sourced from countries like India, Thailand, China, and France, indicates a substantial demand for affordable and accessible healthcare solutions.
3Demand Drivers
Cambodia's disease burden, characterized by a high prevalence of noncommunicable diseases (NCDs) such as cardiovascular diseases, diabetes, and cancer, significantly drives the demand for pharmaceutical imports. Approximately 60,000 deaths annually are attributed to NCDs, with nearly one in four Cambodians dying prematurely between the ages of 30 to 70 years due to these conditions. (who.int)
The country's healthcare spending per capita was US$109.53 in 2022, reflecting a decline from previous years. This trend underscores the need for cost-effective pharmaceutical solutions to meet the healthcare needs of the Cambodian population.
Furthermore, the Cambodian government's commitment to universal health coverage and the expansion of private healthcare facilities are expected to increase the demand for pharmaceutical products. The rising middle class and increased health awareness among Cambodians further contribute to the growing demand for both prescription and OTC medications.
Trade Policy & Tariff Intelligence — India and Cambodia
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
As of August 8, 2025, Cambodia implemented a zero-percent customs duty rate on pharmaceutical products classified under HS code 30, including finished pharmaceutical formulations such as tablets, capsules, syrups, and injections. This exemption applies to all imports of pharmaceutical products, regardless of their country of origin, effectively eliminating the Most Favored Nation (MFN) tariff rates for these goods.
In addition to the customs duty exemption, all imports are subject to a 10 percent Value Added Tax (VAT). However, certain imports, including pharmaceutical products, are exempt from this VAT, further reducing the tax burden on these goods.
Under the ASEAN Free Trade Area (AFTA), Cambodia applies the Common Effective Preferential Tariff (CEPT) scheme, which offers lower tariff rates to imports from other ASEAN member countries, provided that conditions in the Rules of Origin (ROO) are fulfilled. However, specific details regarding the CEPT rates for pharmaceutical products are not readily available. (cambodiainvestment.gov.kh)
2Trade Agreements
Cambodia is a member of the ASEAN Free Trade Area (AFTA), which includes the Common Effective Preferential Tariff (CEPT) scheme. This scheme offers lower tariff rates to imports from other ASEAN member countries, provided that conditions in the Rules of Origin (ROO) are fulfilled. However, specific details regarding the CEPT rates for pharmaceutical products are not readily available. (cambodiainvestment.gov.kh)
As of March 2026, there is no publicly available information indicating the existence of a Free Trade Agreement (FTA) between India and Cambodia. Therefore, Indian pharmaceutical exports to Cambodia are subject to the MFN tariff rates and VAT exemptions applicable to all imports, without any preferential treatment under a bilateral trade agreement.
3IP & Patent Landscape
Cambodia's intellectual property (IP) framework, including patent provisions, data exclusivity, and compulsory licensing, is aligned with the World Trade Organization's Trade-Related Aspects of Intellectual Property Rights (TRIPS) Agreement. This alignment aims to balance the protection of IP rights with the need to ensure access to affordable medicines. While specific details on data exclusivity periods and compulsory licensing provisions are not readily available, Cambodia's adherence to TRIPS suggests that these mechanisms are in place to regulate the entry of generic drugs into the market.
For Indian pharmaceutical companies exporting generic drugs to Cambodia, it is essential to navigate these IP provisions carefully to ensure compliance and to understand the potential for market entry. Engaging with local legal experts or authorities can provide more detailed insights into the current IP landscape and any recent developments affecting generic drug imports.
Supply Chain & Logistics — India to Cambodia Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to Cambodia primarily utilize sea and air transport. Sea freight is the most common method, with major Indian ports such as Nhava Sheva (Jawaharlal Nehru Port) handling a significant portion of these exports. From Nhava Sheva, shipments typically transit to Cambodian ports like Sihanoukville and Phnom Penh. The average transit time for sea freight from India to Cambodia ranges from 8 to 14 days, depending on specific ports of origin and destination.
Air freight offers faster delivery, with transit times of approximately 1 to 3 days. However, air transport is less commonly used for pharmaceutical shipments to Cambodia due to higher costs and limited capacity. The Red Sea disruption, which began in early 2026, has impacted global shipping routes, potentially affecting transit times and costs for sea freight. While specific data on the Red Sea disruption's impact on India-Cambodia pharmaceutical shipments is limited, shippers should anticipate potential delays and increased costs.
2Port Infrastructure
In India, key export ports for pharmaceutical shipments include Nhava Sheva (INNSA1), which accounts for 13.8% of exports, and Chennai Air, handling 1.5% of exports. In Cambodia, major import ports are Sihanoukville (Kompo) with 17.6% of imports, Phnom Penh - Pochent (31.3%), and Phnom Penh (17.8%). These ports are equipped to handle pharmaceutical products, with facilities for both sea and air cargo. However, capacity constraints and infrastructure limitations may affect processing times and efficiency.
3Cold Chain & Compliance
Temperature-sensitive pharmaceutical formulations require strict cold chain logistics to maintain product integrity. Shipments must be stored and transported within specified temperature ranges, typically 2°C to 8°C. Compliance with Good Distribution Practice (GDP) is essential, ensuring that products are handled, stored, and transported under appropriate conditions. Packaging must be robust, utilizing insulated materials and temperature-monitoring devices to safeguard against temperature excursions. In Cambodia, the National Trade Repository (NTR) and the Ministry of Health (MOH) oversee the registration and importation of pharmaceutical products. The Cambodia Pharmaceutical Online Registration System (CamPORS) facilitates the registration process, ensuring that imported pharmaceuticals meet safety, efficacy, and quality standards.
Market Opportunity Assessment — Cambodia for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Cambodia's pharmaceutical market is experiencing significant growth, with revenues projected to reach approximately US$190.75 million in 2025, reflecting a compound annual growth rate (CAGR) of 4.45% from 2025 to 2030, leading to an estimated market volume of US$237.14 million by 2030. This expansion is driven by factors such as increasing healthcare investments, a rising middle class, and heightened health awareness among the Cambodian population.
The market is characterized by a high dependency on imports, as Cambodia's domestic pharmaceutical production is limited. Generic drugs play a significant role in the market, offering affordable alternatives to branded medications and contributing to the overall growth. The government's efforts to improve healthcare infrastructure and regulatory standards further support the demand for both generic and branded pharmaceutical products.
2Healthcare System
Cambodia's healthcare system is evolving, with the government being the primary purchaser of drugs, medical supplies, and equipment. The Ministry of Health oversees the procurement and distribution of pharmaceuticals, ensuring that essential medications are available to the population. However, the private sector's involvement in healthcare is increasing, leading to a more competitive market and offering opportunities for pharmaceutical companies to expand their presence.
Out-of-pocket expenditures constitute a significant portion of healthcare spending in Cambodia, accounting for approximately 60% of total health expenses. This high reliance on direct payments underscores the importance of affordable pharmaceutical options for the Cambodian population.
3Opportunity for Indian Exporters
Indian pharmaceutical exporters have substantial opportunities in Cambodia, particularly in the generic drug segment. The Cambodian market's reliance on imported pharmaceuticals, combined with the government's focus on improving healthcare access, creates a favorable environment for Indian companies to supply cost-effective and high-quality generic medications.
The growth forecast for Indian pharmaceutical imports into Cambodia from 2025 to 2028 is promising, aligning with the overall market expansion. By leveraging their established presence and understanding of the Cambodian market dynamics, Indian exporters can strengthen their position and contribute to meeting the increasing demand for pharmaceuticals in Cambodia.
Competitive Landscape — India vs Other Pharmaceutical Suppliers to Cambodia
Competing origins, India's edge, challenges and threats
1Competing Origins
Cambodia's pharmaceutical market is supplied by a diverse array of international and domestic sources. In 2023, Cambodia imported pharmaceutical products valued at approximately $339.7 million. The primary suppliers include India ($51.5 million), France ($48.0 million), Thailand ($45.1 million), Pakistan ($27.3 million), and Vietnam ($23.8 million). Other notable contributors are Indonesia ($21.0 million), China ($18.4 million), South Korea ($16.1 million), and the United States ($16.0 million).
India holds a significant position in this landscape, exporting pharmaceutical products worth $47.5 million to Cambodia in 2024. This positions India as a leading supplier, with France and Thailand following closely. The presence of domestic manufacturers in Cambodia is limited, leading to a reliance on imports to meet the country's pharmaceutical needs.
2India's Competitive Edge
India's pharmaceutical industry offers several competitive advantages that bolster its position in the Cambodian market. As the world's largest provider of generic medicines, India accounts for approximately 20% of the global supply volume. This extensive production capacity enables India to offer a broad portfolio of medications, catering to diverse therapeutic areas.
Cost competitiveness is another significant advantage. India's efficient manufacturing processes and economies of scale allow it to produce high-quality pharmaceuticals at lower costs compared to many Western counterparts. Additionally, India's adherence to WHO-GMP standards ensures that its products meet international quality benchmarks, enhancing their appeal in markets like Cambodia. The widespread use of English among Indian pharmaceutical professionals facilitates smoother communication and regulatory processes, further strengthening India's position.
3Challenges & Threats
Despite its strengths, India faces several challenges in the Cambodian pharmaceutical market. Regulatory tightening by Cambodian authorities, such as the National Drug Regulatory Authority (WHO-GMP), may impose stricter compliance requirements, potentially affecting market access. Competition from neighboring countries like China, which exported $13.9 million worth of pharmaceutical products to Cambodia in 2024, presents a threat due to China's cost advantages and increasing production capabilities. Additionally, perceptions regarding the quality of Indian pharmaceuticals, despite adherence to international standards, may influence Cambodian buyers' preferences.
FAQ — India to Cambodia Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Cambodia?
India exported pharmaceuticals worth $103.0M to Cambodia across 7,580 verified shipments.
Who are the top Indian pharmaceutical exporters to Cambodia?
1. SYNAPSE PHARMATECH — $11.0M. 2. MARKSANS PHARMA LIMITED — $8.2M. 3. MYLAN LABORATORIES LIMITED — $7.0M. Total: 380 suppliers.
Which companies in Cambodia import pharmaceuticals from India?
1. ISKP PHARMA CO., LTD — $8.3M. 2. NATIONAL CENTER FOR HIV/AIDS, — $5.7M. 3. SRI KRISHNA LIFE SCIENCE CO.LTD, — $5.5M. 579 buyers total.
What pharmaceutical products does India export most to Cambodia?
1. Albusyn (50 Ml)each Vial Con.human Album ($10.7M, 10.4%); 2. Pharma Product-amoxmark-500capsules Amoxycillin Capsules 500 MG (each Capsule C… ($7.3M, 7.1%); 3. Tenofovir Disoproxil Fumarate 300mg + Lamivudine 300mg Tablets (30s) (40000 Pac… ($3.5M, 3.4%); 4. Throzin-500 MG ($2.1M, 2.0%); 5. Famlo 5 MG (ba ($2.0M, 2.0%)
Which ports handle pharmaceutical shipments from India to Cambodia?
Export: AHEMDABAD ICD, CHENNAI AIR, NHAVA SHEVA SEA (INNSA1), NHAVA SHEVA SEA, SAHAR AIR. Import: Sihanoukville (Kompo, PHNOM PENH - POCHENT, Phnom Penh, SIHANOUKVILLE (KOMPO, PHNOM PENH.
Why does Cambodia import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $103.0M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Cambodia?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Cambodia pharmaceutical trade?
$13.6K per consignment across 7,580 shipments.
How many Indian pharmaceutical companies export to Cambodia?
380 Indian companies. Largest: SYNAPSE PHARMATECH with $11.0M.
How can I find verified Indian pharmaceutical suppliers for Cambodia?
TransData Nexus covers 380 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Cambodia Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 7,580 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Cambodia identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 7,580 verified shipments from 380 Indian exporters to 579 Cambodia buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
380 Exporters
579 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists