India to Belgium Pharmaceutical Export
Bilateral Trade Intelligence · $765.8M Total Trade · 136 Exporters · 241 Buyers · DGFT Verified · Updated March 2026
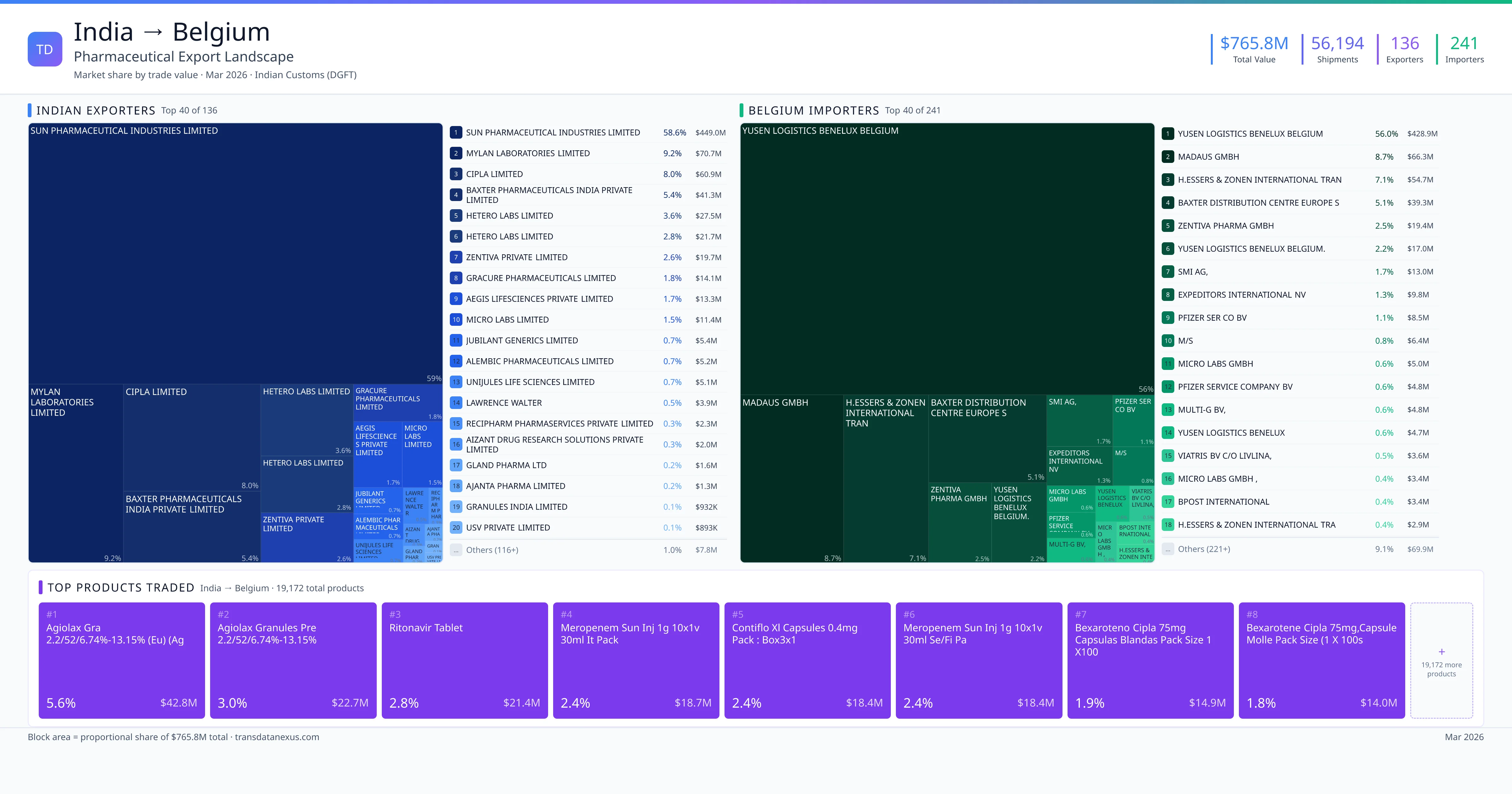
India exported $765.8M worth of pharmaceutical formulations to Belgium across 56,194 verified shipments, sourced from 136 Indian exporters supplying 241 Belgium buyers. The top exporters are SUN PHARMACEUTICAL INDUSTRIES LIMITED ($449.0M) and MYLAN LABORATORIES LIMITED ($70.7M). The leading products are Ritonavir ($21.4M) and Meropenem ($18.7M). Average shipment value: $13.6K.
Top Pharmaceutical Formulations — India to Belgium
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Agiolax Gra 2.2/52/6.74%-13.15% (eu) (ag | $42.8M | 5.6% |
| 2 | Agiolax Granules Pre 2.2/52/6.74%-13.15% | $22.7M | 3.0% |
| 3 | Ritonavir Tablet | $21.4M | 2.8% |
| 4 | Meropenem Sun Injection 1g 10x1v 30ml It Pack | $18.7M | 2.4% |
| 5 | Contiflo Xl Capsules 0.4mg Pack : Box3x1 | $18.4M | 2.4% |
| 6 | Meropenem Sun Injection 1g 10x1v 30ml Se/fi Pa | $18.4M | 2.4% |
| 7 | Bexaroteno Cipla 75mg Capsulas Blandas Pack Size 1 X100s | $14.9M | 1.9% |
| 8 | Bexarotene Cipla 75mg,capsule Molle Pack Size (1 X 100s) | $14.0M | 1.8% |
| 9 | Sertraline Tablets 100mg Pack :box 2x14s | $12.4M | 1.6% |
| 10 | Aciclovir Tablets BP 200mg ( Aciclovir T | $12.1M | 1.6% |
| 11 | Ritonavir Tablets | $11.6M | 1.5% |
| 12 | Sertraline Sun Tablets 100mg Pack : Box 2x14 | $10.6M | 1.4% |
| 13 | Cefaclor Basics Ts Sus 250mg/5ml When Co | $10.4M | 1.4% |
| 14 | Candesartan Cilexetil Tablets 16mg (tandesar Tablets 16mg Each Tablets Cont:can… | $9.2M | 1.2% |
| 15 | Klabax Tablets 500mg Pack : 14s Strip Ba | $8.6M | 1.1% |
India exports 20+ pharmaceutical formulations to Belgium with a combined trade value of $765.8M. Key products include Ritonavir ($21.4M), Meropenem ($18.7M), Sertraline ($12.4M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 56,194 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Belgium Trade Routes
These are the top pharmaceutical products exported from India to Belgium, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Belgium buyers, regulatory requirements, and logistics for that specific product corridor. Products include Ritonavir ($21.4M), Meropenem ($18.7M), Sertraline ($12.4M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Belgium
136 Indian pharmaceutical companies export finished formulations to Belgium. Leading exporters include Sun Pharmaceutical Industries Limited, Mylan Laboratories Limited, Cipla Limited, Baxter Pharmaceuticals India Private Limited. The top exporter accounts for 58.6% of total India–Belgium pharma exports. Source: Indian Customs (DGFT).
Top Belgium Buyers from India
241 companies in Belgium import pharmaceutical formulations from India. Top buyers include Yusen Logistics Benelux Belgium, Madaus Gmbh, H.essers & Zonen International Tran, Baxter Distribution Centre Europe S. The largest buyer accounts for 56.0% of India–Belgium pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Belgium Pharmaceutical Shipments
Indian Export Ports
Belgium Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Belgium Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The India-Belgium pharmaceutical trade corridor has experienced significant growth over the past two decades. In the fiscal year 2022–23, India's pharmaceutical exports to Belgium were valued at approximately $714.92 million, marking a substantial increase from previous years. This upward trajectory reflects the strengthening of bilateral trade relations and India's growing presence in the European pharmaceutical market.
A pivotal milestone in this relationship was the conclusion of the EU-India Free Trade Agreement (FTA) on January 27, 2026. This agreement aims to eliminate or reduce tariffs on over 96% of EU goods exports to India, including pharmaceutical products. The FTA is expected to enhance market access for Indian pharmaceutical companies, potentially doubling EU goods exports to India by 2032. (policy.trade.ec.europa.eu)
2India's Market Position
India's pharmaceutical exports to Belgium have been a significant component of its overall pharmaceutical trade. In 2024, India exported $320.11 million worth of pharmaceutical products to Belgium, contributing to the EU's total pharmaceutical imports. This positions India as a key supplier in the European market, underscoring the strategic importance of the India-Belgium pharmaceutical trade corridor.
3Recent Developments
The EU-India Free Trade Agreement, concluded on January 27, 2026, is a landmark development in the bilateral pharmaceutical trade. By reducing tariffs and simplifying regulatory procedures, the FTA is poised to facilitate smoother market access for Indian pharmaceutical products in Belgium and the broader EU market. This agreement is anticipated to bolster India's pharmaceutical exports, enhance competitiveness, and foster deeper economic integration between India and Belgium. (policy.trade.ec.europa.eu)
Belgium Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
Indian pharmaceutical companies seeking to export finished formulations to Belgium must obtain marketing authorization through the European Medicines Agency (EMA). The process involves submitting a Marketing Authorization Application (MAA) that demonstrates the product's quality, safety, and efficacy. For generic medicines, the application must include evidence of bioequivalence to the reference product, typically through appropriate bioavailability studies. The Common Technical Document (CTD) format is standard for these submissions, comprising modules on quality, non-clinical, and clinical data. The approval timeline varies but generally spans several months, depending on the complexity of the application and the EMA's workload. Fees associated with the application process are outlined in the EMA's fee schedule and are subject to change; applicants should consult the latest guidelines for current information. (ema.europa.eu)
2GMP & Facility Requirements
Belgium, as an EU member state, adheres to Good Manufacturing Practice (GMP) standards set by the European Medicines Agency (EMA). Indian manufacturing facilities exporting to Belgium must comply with these GMP requirements, which include maintaining high-quality production processes and facilities. The EMA conducts inspections to verify compliance, and these inspections may involve fees as specified in the EMA's fee schedule. For inspections outside the European Economic Area (EEA), applicants are also required to cover the travel and accommodation expenses of the inspectors and any experts involved. (ema.europa.eu)
3Import Documentation
To import pharmaceutical products into Belgium, Indian exporters must provide specific documentation, including a Certificate of Pharmaceutical Product (CPP), Certificate of Analysis (CoA), and proof of GMP compliance. These documents facilitate customs clearance and ensure that the products meet Belgian regulatory standards. The import process involves customs procedures that verify the authenticity and compliance of the imported goods with Belgian regulations. It's essential for exporters to ensure that all documentation is accurate and up-to-date to prevent delays or issues during the import process.
Product Categories & Therapeutic Trends — India to Belgium
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Belgium are predominantly concentrated in the gastrointestinal (GI) and infectious disease therapeutic areas. The leading products include AGIOLAX GRA 2.2/52/6.74%-13.15% (AG), AGIOLAX GRANULES PRE 2.2/52/6.74%-13.15%, and RITONAVIR TABLET, collectively accounting for a significant portion of the export value. This dominance aligns with Belgium's healthcare needs, where gastrointestinal disorders and infectious diseases are prevalent. The high demand for these treatments underscores the importance of effective management and therapeutic interventions in these areas.
The substantial export of AGIOLAX products, which are used to treat constipation, indicates a significant demand for gastrointestinal treatments in Belgium. Similarly, the inclusion of RITONAVIR TABLET, an antiretroviral medication, reflects the ongoing need for effective therapies against infectious diseases, particularly HIV/AIDS. These trends highlight the critical role of Indian pharmaceutical exports in addressing Belgium's therapeutic requirements in these domains.
2Emerging Opportunities
Belgium's pharmaceutical market is experiencing patent expirations, creating opportunities for Indian generics to enter the market. The uptake of generics in Belgium has been relatively low compared to other European countries, with generics constituting 38% of all pharmaceutical units dispensed through community pharmacies as of 2023, up from 26% in 2012. This presents a significant opportunity for Indian exporters to provide cost-effective alternatives to branded medications. Additionally, the market for biosimilars in Belgium remains underdeveloped, with market shares for most biosimilars still below 20% in 2019. This indicates a potential for growth in the biosimilar segment, where Indian manufacturers can leverage their expertise to meet the demand for affordable biologic treatments.
3Demand Drivers
Belgium's aging population is a significant driver of pharmaceutical import demand. The fiscal cost of aging, primarily related to pension and health outlays, is mounting, with pension spending expected to increase rapidly unless measures related to pension generosity and retirement eligibility are implemented. This demographic shift leads to a higher prevalence of chronic diseases, increasing the demand for pharmaceutical products. Furthermore, the burden of disease attributable to high body mass index (BMI) in Belgium is substantial, affecting various health outcomes such as type 2 diabetes, osteoarthritis, and cardiovascular diseases. This underscores the need for effective treatments and management strategies, driving the demand for pharmaceutical imports from countries like India.
Supply Chain & Logistics — India to Belgium Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to Belgium predominantly utilize sea freight due to its cost-effectiveness for large volumes. Major Indian ports such as Nhava Sheva (Jawaharlal Nehru Port), Mundra, and Chennai serve as primary departure points. These ports are well-equipped to handle pharmaceutical exports, offering facilities that comply with international shipping standards. The journey to Belgium typically involves transiting through the Suez Canal, connecting the Indian Ocean to the Mediterranean Sea, and then proceeding to Belgian ports. Sea freight transit times range from 12 to 18 days, depending on the specific ports of departure and arrival, as well as maritime conditions.
Air freight, while faster, is less commonly used for pharmaceutical shipments from India to Belgium due to higher costs. When utilized, air freight can deliver goods in approximately 1 to 3 days. However, the preference for sea freight is evident, with a significant majority of pharmaceutical products being shipped by sea rather than air.
Recent disruptions in the Red Sea, such as the blockage of the Suez Canal in March 2021, have highlighted the vulnerability of these critical shipping routes. Such events can lead to significant delays and necessitate rerouting, which may increase transit times and costs. While the Suez Canal blockage was a notable incident, the Red Sea remains a vital artery for global trade, and any disruptions can have cascading effects on supply chains.
2Port Infrastructure
In India, key export ports handling pharmaceutical shipments include Nhava Sheva (Jawaharlal Nehru Port), Mundra, and Chennai. Nhava Sheva, located near Mumbai, is the largest container port in India and offers extensive facilities for pharmaceutical exports. Mundra Port, situated in Gujarat, is known for its modern infrastructure and efficient cargo handling capabilities. Chennai Port serves as a major gateway for exports from the southern region of India, equipped with specialized terminals for various cargo types.
In Belgium, the primary import ports for pharmaceutical products are Antwerp and Brussels. Antwerp is one of the largest ports in Europe, offering advanced logistics facilities and a strategic location for European distribution. Brussels, while not a seaport, serves as a significant air cargo hub, facilitating the import of goods through Brussels Airport, which is well-connected to global air freight networks.
3Cold Chain & Compliance
Pharmaceutical shipments, especially those containing temperature-sensitive formulations, require stringent cold chain logistics to maintain product efficacy and comply with Good Distribution Practice (GDP) standards. This involves using insulated packaging, temperature-controlled containers, and continuous monitoring of temperature conditions throughout transit. Compliance with GDP ensures that pharmaceutical products are stored, transported, and handled under controlled conditions, safeguarding their quality and safety. Additionally, adherence to packaging standards is crucial to prevent contamination and ensure the integrity of the products upon arrival.
Market Opportunity Assessment — Belgium for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Belgium's pharmaceutical market is substantial, with total local production valued at €202.6 billion in 2022. The market has experienced growth, with projections estimating a total market size of €177.23 billion by 2023. The market is characterized by a high penetration of generic drugs, accounting for 79% of the pharmaceutical market by volume, surpassing the OECD average of 56%. This indicates a mature and competitive market environment.
2Healthcare System
Belgium's healthcare system is predominantly funded through compulsory health insurance, covering approximately 99% of the population. In 2021, the total health expenditure was 11.0% of GDP, with public expenditure accounting for 77.6%. (eurohealthobservatory.who.int) The system emphasizes accessibility, quality, and efficiency, with ongoing reforms aimed at improving care delivery and system resilience.
3Opportunity for Indian Exporters
Indian pharmaceutical exporters have a significant opportunity in Belgium's market, particularly in the generic drug segment, which comprises 79% of the market by volume. The top products exported from India to Belgium include AGIOLAX GRA 2.2/52/6.74%-13.15% (EU), AGIOLAX GRANULES PRE 2.2/52/6.74%-13.15%, and RITONAVIR TABLET, indicating a strong presence in gastrointestinal and antiviral medications. The growth forecast for Indian pharmaceutical imports into Belgium from 2025 to 2028 is positive, with the market projected to witness mixed growth rate patterns during this period. This trend suggests a favorable environment for Indian exporters to expand their market share in Belgium.
Competitive Landscape — India vs Other Pharmaceutical Suppliers to Belgium
Competing origins, India's edge, challenges and threats
1Competing Origins
Belgium's pharmaceutical market is supplied by a diverse array of countries, including domestic manufacturers, other European Union (EU) member states, China, and emerging markets. While specific market share data for each country is not readily available, it is evident that Belgium's pharmaceutical imports are substantial, with a total import value of $3.62 billion in 2024. This figure encompasses imports from various origins, highlighting the competitive nature of the Belgian pharmaceutical market.
India's pharmaceutical exports to Belgium amounted to $765.8 million in 2024, accounting for approximately 21% of Belgium's total pharmaceutical imports. This positions India as a significant supplier, though it faces competition from other countries, including EU manufacturers and China. The presence of multiple suppliers contributes to a dynamic and competitive landscape, with each origin striving to meet Belgium's demand for high-quality and cost-effective pharmaceutical products.
2India's Competitive Edge
India's pharmaceutical industry offers several competitive advantages that enhance its appeal to Belgian buyers:
- Cost Competitiveness: Indian manufacturers provide high-quality pharmaceutical products at competitive prices, making them attractive to cost-conscious Belgian importers.
- Regulatory Compliance: Many Indian pharmaceutical companies adhere to stringent international standards, including WHO-GMP and EU-GMP guidelines, ensuring the safety and efficacy of their products.
- Product Variety: India's pharmaceutical sector boasts a broad portfolio, encompassing generic medicines, active pharmaceutical ingredients (APIs), vaccines, biosimilars, and custom research manufacturing (CRM), catering to diverse therapeutic needs.
- English-Speaking Workforce: The widespread use of English in India facilitates effective communication and collaboration with international partners, including those in Belgium.
3Challenges & Threats
Despite its strengths, India's pharmaceutical exports to Belgium face several challenges:
- Regulatory Compliance: Navigating the complex regulatory landscape of the European Medicines Agency (EMA) and Belgium's National Competent Authority requires continuous vigilance and adaptation to evolving standards.
- Competition from China: Chinese pharmaceutical manufacturers often offer products at lower prices, posing a competitive threat to Indian exporters in the Belgian market.
- Quality Perception: While many Indian pharmaceutical products meet international standards, some buyers may have concerns regarding quality, necessitating ongoing efforts to build and maintain trust.
Over time, India's position in the Belgian pharmaceutical market has been influenced by these factors, requiring strategic responses to maintain and enhance its market share.
FAQ — India to Belgium Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Belgium?
India exported pharmaceuticals worth $765.8M to Belgium across 56,194 verified shipments.
Who are the top Indian pharmaceutical exporters to Belgium?
1. SUN PHARMACEUTICAL INDUSTRIES LIMITED — $449.0M. 2. MYLAN LABORATORIES LIMITED — $70.7M. 3. CIPLA LIMITED — $60.9M. Total: 136 suppliers.
Which companies in Belgium import pharmaceuticals from India?
1. YUSEN LOGISTICS BENELUX BELGIUM — $428.9M. 2. MADAUS GMBH — $66.3M. 3. H.ESSERS & ZONEN INTERNATIONAL TRAN — $54.7M. 241 buyers total.
What pharmaceutical products does India export most to Belgium?
1. Agiolax Gra 2.2/52/6.74%-13.15% (eu) (ag ($42.8M, 5.6%); 2. Agiolax Granules Pre 2.2/52/6.74%-13.15% ($22.7M, 3.0%); 3. Ritonavir Tablet ($21.4M, 2.8%); 4. Meropenem Sun Injection 1g 10x1v 30ml It Pack ($18.7M, 2.4%); 5. Contiflo Xl Capsules 0.4mg Pack : Box3x1 ($18.4M, 2.4%)
Which ports handle pharmaceutical shipments from India to Belgium?
Export: DHANNAD ICD, GOA, NHAVA SHEVA SEA (INNSA1), MUNDRA SEA, HYDERABAD ACC (INHYD4). Import: Antwerpen, ANTWERPEN, BRUSSELS, Antwerp Churchill Te, ANTWERP CHURCHILL TE.
Why does Belgium import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $765.8M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Belgium?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Belgium pharmaceutical trade?
$13.6K per consignment across 56,194 shipments.
How many Indian pharmaceutical companies export to Belgium?
136 Indian companies. Largest: SUN PHARMACEUTICAL INDUSTRIES LIMITED with $449.0M.
How can I find verified Indian pharmaceutical suppliers for Belgium?
TransData Nexus covers 136 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Belgium Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 56,194 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Belgium identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 56,194 verified shipments from 136 Indian exporters to 241 Belgium buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
136 Exporters
241 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists