To export finished pharmaceutical formulations to Aruba, Indian pharmaceutical companies must adhere to the regulatory requirements set by Aruba's National Drug Regulatory Authority (NDRA), which operates under WHO-GMP standards. The registration process for Indian generics involves several key steps:
1. Dossier Submission: Companies are required to submit a comprehensive dossier that includes detailed information about the product, manufacturing process, quality control measures, and clinical data. The dossier should be prepared in the Common Technical Document (CTD) format, which is internationally recognized and facilitates the review process.
2. Bioequivalence Studies: For generic products, demonstrating bioequivalence to the reference listed drug is essential. This involves conducting clinical studies to confirm that the generic product performs similarly to the innovator drug in terms of absorption and therapeutic effect.
3. Approval Timeline and Fees: The approval timeline can vary based on the complexity of the product and the completeness of the submitted dossier. Typically, the review process may take several months. While specific fee structures are not detailed in the available sources, applicants should anticipate costs associated with dossier preparation, submission, and potential facility inspections.
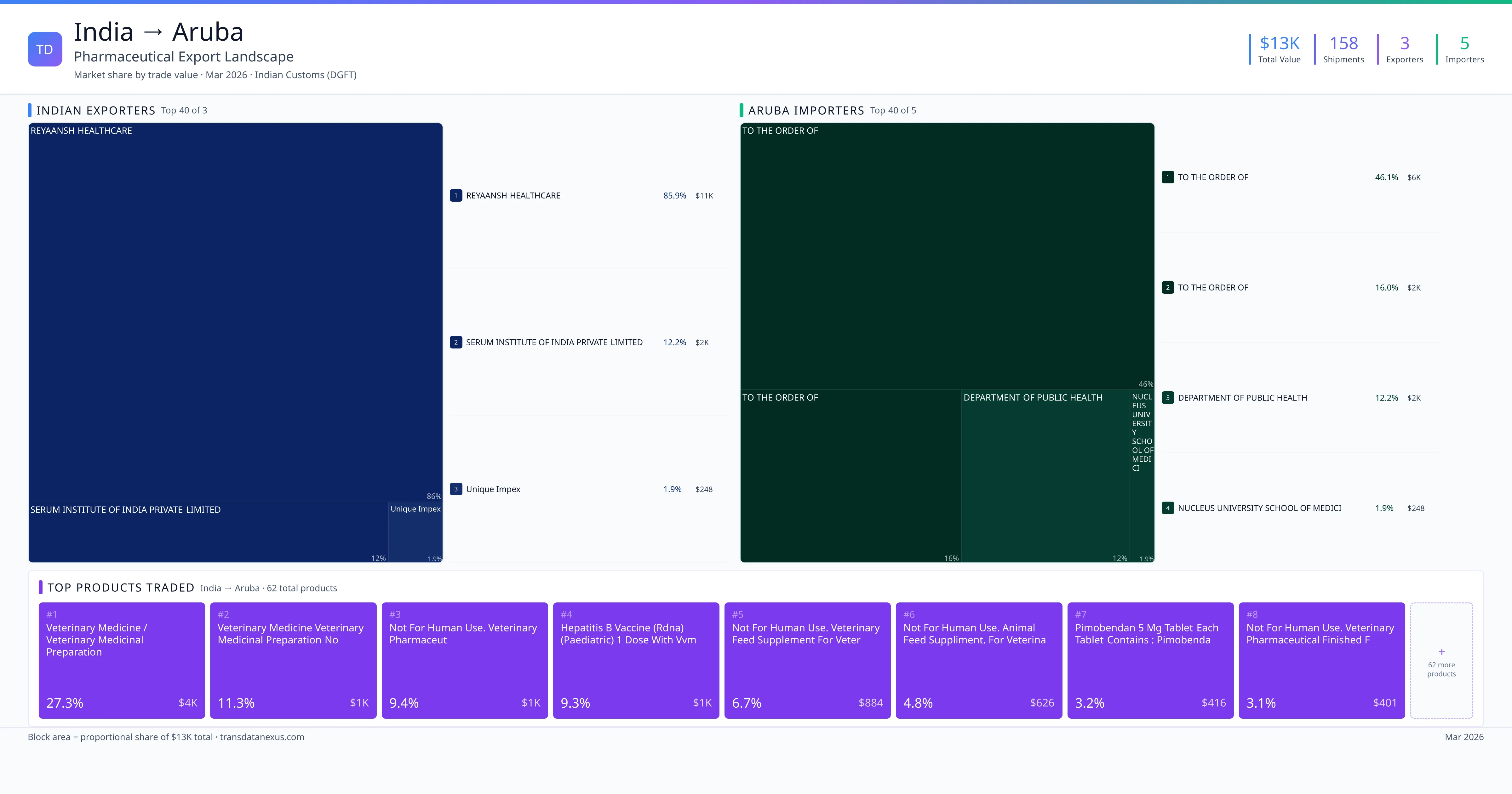
Given the limited trade volume between India and Aruba—totaling $13,000 USD over 158 shipments from 2022 to 2026—it's crucial for exporters to ensure compliance with these regulatory requirements to facilitate market entry.