India to Anguilla Pharmaceutical Export
Bilateral Trade Intelligence · $124.9K Total Trade · 8 Exporters · 10 Buyers · DGFT Verified · Updated March 2026
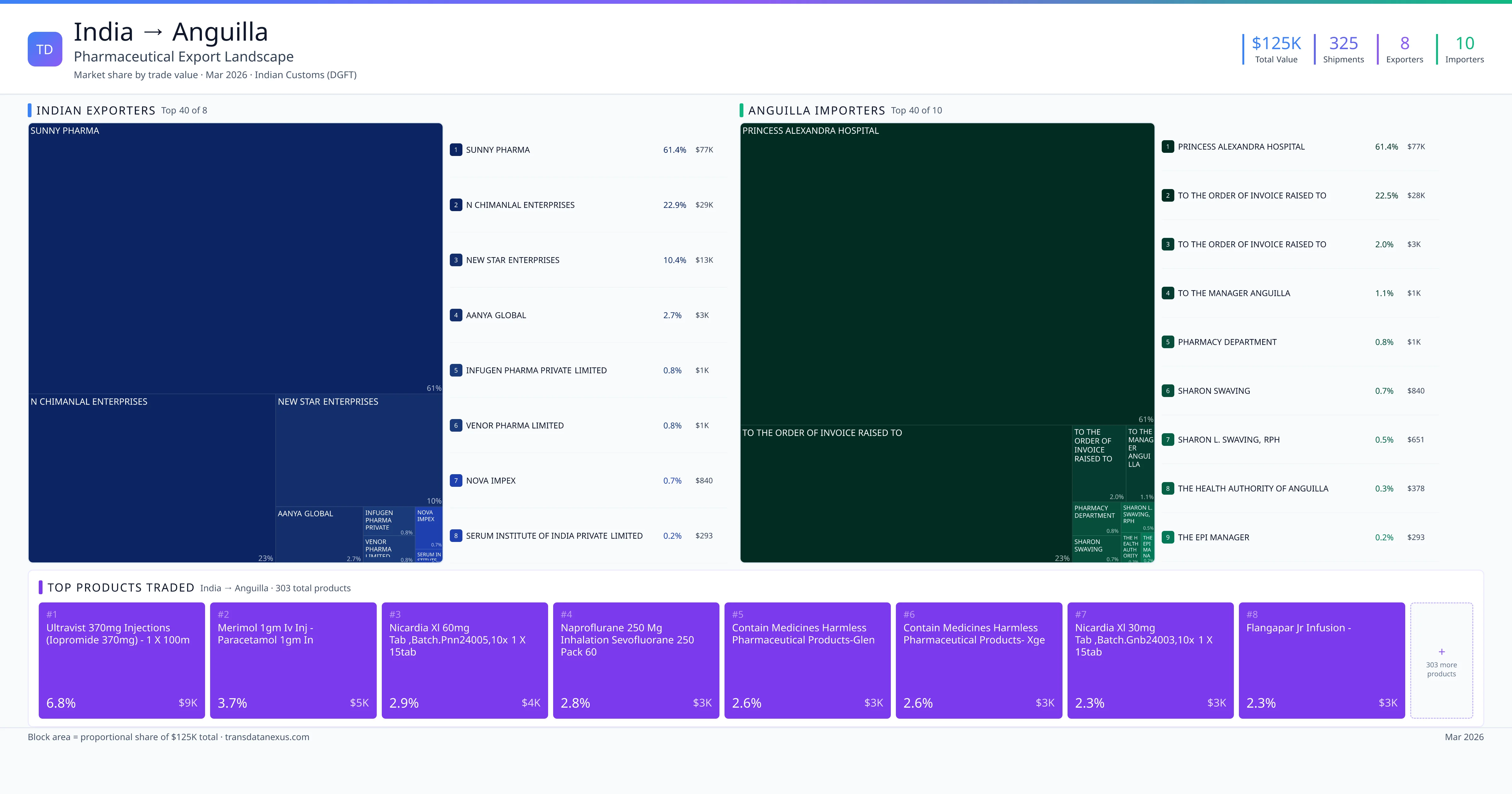
India exported $124.9K worth of pharmaceutical formulations to Anguilla across 325 verified shipments, sourced from 8 Indian exporters supplying 10 Anguilla buyers. The top exporters are SUNNY PHARMA ($76.7K) and N CHIMANLAL ENTERPRISES ($28.7K). The leading products are Ultravist 370mg Injections (iopromide 370mg) - 1 X 100ml, Merimol 1gm IV Injection - Paracetamol 1gm in, Nicardia Xl 60mg Tablets ,batch.pnn24005,10x 1 X 15tab. Average shipment value: $384.
Top Pharmaceutical Formulations — India to Anguilla
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Ultravist 370mg Injections (iopromide 370mg) - 1 X 100ml | $8.5K | 6.8% |
| 2 | Merimol 1gm IV Injection - Paracetamol 1gm in | $4.6K | 3.7% |
| 3 | Nicardia Xl 60mg Tablets ,batch.pnn24005,10x 1 X 15tab | $3.7K | 2.9% |
| 4 | Naproflurane 250 MG Inhalation Sevofluorane 250 Pack 60 X 250 ML | $3.5K | 2.8% |
| 5 | Contain Medicines Harmless Pharmaceutical Products-glenza 40 MG Tablets(enazalu… | $3.3K | 2.6% |
| 6 | Contain Medicines Harmless Pharmaceutical Products- Xgeva 120 MG Injection(denu… | $3.3K | 2.6% |
| 7 | Nicardia Xl 30mg Tablets ,batch.gnb24003,10x 1 X 15tab | $2.9K | 2.3% |
| 8 | Flangapar Jr Infusion | $2.9K | 2.3% |
| 9 | Albucel 20 Percent Solution ,batch.97010536,1 X 100ml | $2.3K | 1.9% |
| 10 | Flagapar - Jr Infusion- Paracetamol 500mg Injection - Par500a- 1040 Btl | $2.1K | 1.7% |
| 11 | Omez 40mg Injection- (omeprazole 40mg)- Vial - 48 Box | $2.1K | 1.6% |
| 12 | Hexidine Mouthwash Batch.l24011,1 X 160ml | $2.1K | 1.6% |
| 13 | Contain Medicines Harmless Pharmaceutical Products-hertraz 440 MG Injection(tra… | $2.0K | 1.6% |
| 14 | Flagapar Jr Infusion - - 1x50ml 1040 Btlnos | $1.9K | 1.5% |
| 15 | Lamez Od 200mg Tablets Batch.k2301934,10 X 1x 10tab | $1.8K | 1.4% |
India exports 20+ pharmaceutical formulations to Anguilla with a combined trade value of $124.9K.. These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 325 verified Indian Customs (DGFT) shipment records.
Top Indian Exporters to Anguilla
8 Indian pharmaceutical companies export finished formulations to Anguilla. Leading exporters include Sunny Pharma, N Chimanlal Enterprises, New Star Enterprises, Aanya Global. The top exporter accounts for 61.4% of total India–Anguilla pharma exports. Source: Indian Customs (DGFT).
Top Anguilla Buyers from India
10 companies in Anguilla import pharmaceutical formulations from India. Top buyers include Princess Alexandra Hospital, To The Order Of Invoice Raised To, To The Order Of Invoice Raised To, To The Manager Anguilla. The largest buyer accounts for 61.4% of India–Anguilla pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Anguilla Pharmaceutical Shipments
Indian Export Ports
Anguilla Import Ports
Trade Statistics
Other Destinations
India–Anguilla Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and Anguilla has been modest but consistent over recent years. Between 2022 and 2026, India exported a total of $125,000 worth of pharmaceutical products to Anguilla, distributed across 325 shipments. This indicates a steady engagement, albeit on a smaller scale compared to India's broader pharmaceutical export activities. The average shipment value stands at $384, reflecting the niche nature of this trade corridor.
The primary recipient of these exports is the Princess Alexandra Hospital in Anguilla, which has received goods totaling $77,000 across 160 shipments. This suggests that the hospital is a significant consumer of Indian pharmaceutical products, likely due to the quality and reliability associated with Indian-made medicines. The top products exported include ULTRAVIST 370MG INJECTIONS (IOPROMIDE 370MG), MERIMOL 1GM IV INJ (PARACETAMOL 1GM IN), and NICARDIA XL 60MG TAB, among others. These products are essential for various medical treatments, indicating a demand for specialized pharmaceutical formulations in Anguilla.
2India's Market Position
While specific data on India's share of Anguilla's total pharmaceutical imports is not readily available, the consistent export activity suggests a stable presence in the market. Given India's reputation as a leading global supplier of affordable and high-quality medicines, it is reasonable to infer that Indian pharmaceutical products hold a significant position in Anguilla's import portfolio. This trade corridor, though modest in volume, underscores the strategic importance of India's pharmaceutical exports to smaller Caribbean nations, highlighting the potential for growth and deeper market penetration.
3Recent Developments
Between 2024 and 2026, there have been no publicly documented policy changes, diplomatic meetings, or regulatory agreements specifically affecting the pharmaceutical trade between India and Anguilla. However, India's broader pharmaceutical export landscape has seen significant developments. In December 2025, Commerce Secretary Shri Rajesh Agrawal announced that India's pharmaceutical exports had reached $30.47 billion in the fiscal year 2024–25, marking a 9.4% increase from the previous year. This growth is attributed to a strong manufacturing base and expanding global outreach. Additionally, in February 2026, an interim trade agreement between India and the United States was reported, offering tariff relief on medicines and other key exports. While this agreement primarily focuses on the U.S. market, it reflects India's ongoing efforts to enhance its pharmaceutical trade relations globally, which could indirectly benefit smaller markets like Anguilla.
Product Categories & Therapeutic Trends — India to Anguilla
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Anguilla are predominantly in the therapeutic areas of cardiovascular diseases, pain management, and oncology. This aligns with Anguilla's healthcare needs, where noncommunicable diseases (NCDs), including cardiovascular diseases, diabetes, cancers, and chronic respiratory diseases, are the leading causes of death. According to the Pan American Health Organization (PAHO), more than 50% of deaths in Anguilla are due to NCDs, with approximately 25-30% of Anguillans suffering from hypertension.
The top exported products, such as Iopromide 370mg injections (Ultravist), Paracetamol 1g IV injections (Merimol), and Enzalutamide 40mg tablets (Glenza), are indicative of this focus. Iopromide is used in diagnostic imaging, Paracetamol is a common analgesic, and Enzalutamide is utilized in cancer treatment. These products address critical therapeutic needs in Anguilla, reflecting the country's emphasis on managing cardiovascular conditions, pain relief, and cancer care.
2Emerging Opportunities
The global pharmaceutical industry is witnessing patent expirations, known as "patent cliffs," leading to increased demand for generic medications. Anguilla, lacking a domestic pharmaceutical manufacturing industry, relies heavily on imports to meet its healthcare needs. This scenario presents a significant opportunity for Indian pharmaceutical exporters to supply high-quality generics, especially in therapeutic areas experiencing patent expirations.
Additionally, the rising prevalence of NCDs in Anguilla creates a growing market for biosimilars. Biosimilars offer cost-effective alternatives to expensive biologic therapies, making them an attractive option for Anguilla's healthcare system. Indian pharmaceutical companies, with their expertise in biosimilar development, are well-positioned to meet this demand.
3Demand Drivers
Anguilla's disease burden, characterized by a high prevalence of NCDs such as cardiovascular diseases, diabetes, cancers, and chronic respiratory diseases, significantly drives the demand for pharmaceutical imports. The Pan American Health Organization (PAHO) reports that more than 50% of deaths in Anguilla are due to NCDs, with approximately 25-30% of Anguillans suffering from hypertension.
The aging population further exacerbates this demand. In 2024, individuals over 65 years of age accounted for 12.5% of Anguilla's total population, an increase of 5.4 percentage points compared to the year 2000. This demographic shift leads to a higher incidence of age-related health conditions, increasing the need for pharmaceutical interventions.
Moreover, healthcare spending trends indicate a growing investment in health services. While specific data for Anguilla is limited, the Caribbean region has been experiencing an upward trajectory in health expenditures. The Inter-American Development Bank (IDB) projects that health spending in Latin America and the Caribbean will double by 2050. This trend suggests a potential increase in Anguilla's healthcare budget, further driving the demand for pharmaceutical imports.
Trade Policy & Tariff Intelligence — India and Anguilla
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Anguilla's tariff structure for pharmaceutical formulations under Harmonized System (HS) codes 3003 and 3004 is as follows:
- HS Code 3003.90.80: Medicaments containing specific active ingredients such as salbutamol, ephedrine, theophylline, and others are subject to a duty rate of 10%.
- HS Code 3003.90.90: Other medicaments not specified elsewhere under HS Code 3003.90 are also subject to a duty rate of 10%.
- HS Code 3004: Medicaments consisting of mixed or unmixed products for therapeutic or prophylactic uses, put up in measured doses or in forms or packings for retail sale, are generally subject to a duty rate of 10%. Specific subcategories include:
- HS Code 3004.10.10: Medicaments containing ampicillin (capsules or oral suspension) are subject to a duty rate of 10%.
- HS Code 3004.10.20: Medicaments containing amoxicillin (capsules or oral suspension) are subject to a duty rate of 10%.
- HS Code 3004.10.90: Other medicaments containing penicillins or derivatives thereof are subject to a duty rate of 10%.
- HS Code 3004.20.10: Medicaments containing tetracycline (capsules or skin ointment) are subject to a duty rate of 10%.
- HS Code 3004.20.20: Medicaments containing chloramphenicol (capsules, oral suspension, or optic drops) are subject to a duty rate of 10%.
- HS Code 3004.20.30: Medicaments containing griseofulvin (tablets of a strength of 125 mg or 500 mg) are subject to a duty rate of 10%.
- HS Code 3004.20.40: Medicaments containing nystatin (skin cream or skin ointment) are subject to a duty rate of 10%.
- HS Code 3004.20.90: Other medicaments containing antibiotics are subject to a duty rate of 10%.
- HS Code 3004.31.00: Medicaments containing insulin are subject to a duty rate of 10%.
- HS Code 3004.32.00: Medicaments containing corticosteroid hormones, their derivatives, or structural analogues are subject to a duty rate of 10%.
These duty rates are applicable to imports from all countries, including India. Additionally, a 1% Environmental Levy is applied to the customs value of imported goods, effectively making the total duty rate 11%. It's important to note that these rates are subject to change, and importers should consult the latest Anguilla Customs Tariff Schedule for the most current information. (customs.gov.ai)
2Trade Agreements
As of March 2026, there are no known Free Trade Agreements (FTAs) or bilateral trade agreements between India and Anguilla specifically addressing pharmaceutical products. Anguilla, as a British Overseas Territory, adheres to the United Kingdom's trade policies and agreements. The UK has various trade agreements with countries worldwide; however, these agreements do not extend to Anguilla. Consequently, pharmaceutical imports from India to Anguilla are subject to the standard Most Favored Nation (MFN) tariff rates without any preferential treatment. There are no current or upcoming negotiations for trade agreements between India and Anguilla that would impact pharmaceutical trade.
3IP & Patent Landscape
Anguilla's intellectual property (IP) framework is aligned with the United Kingdom's standards, as it is a British Overseas Territory. The UK provides patent protection for pharmaceutical products, including provisions for data exclusivity and compulsory licensing. Data exclusivity grants innovators a period during which generic manufacturers cannot rely on the innovator's clinical trial data to obtain marketing authorization, typically lasting eight years. Compulsory licensing allows the government to grant licenses to third parties to produce a patented product without the consent of the patent holder under specific conditions, such as public health emergencies. These provisions can affect the entry of Indian generic drugs into the Anguillan market, as they may delay the introduction of generics or allow for their production under certain circumstances. Importers should be aware of these IP regulations when considering the importation of generic pharmaceutical products into Anguilla.
FAQ — India to Anguilla Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Anguilla?
India exported pharmaceuticals worth $124.9K to Anguilla across 325 verified shipments.
Who are the top Indian pharmaceutical exporters to Anguilla?
1. SUNNY PHARMA — $76.7K. 2. N CHIMANLAL ENTERPRISES — $28.7K. 3. NEW STAR ENTERPRISES — $12.9K. Total: 8 suppliers.
Which companies in Anguilla import pharmaceuticals from India?
1. PRINCESS ALEXANDRA HOSPITAL — $76.7K. 2. TO THE ORDER OF INVOICE RAISED TO — $28.1K. 3. TO THE ORDER OF INVOICE RAISED TO — $2.5K. 10 buyers total.
What pharmaceutical products does India export most to Anguilla?
1. Ultravist 370mg Injections (iopromide 370mg) - 1 X 100ml ($8.5K, 6.8%); 2. Merimol 1gm IV Injection - Paracetamol 1gm in ($4.6K, 3.7%); 3. Nicardia Xl 60mg Tablets ,batch.pnn24005,10x 1 X 15tab ($3.7K, 2.9%); 4. Naproflurane 250 MG Inhalation Sevofluorane 250 Pack 60 X 250 ML ($3.5K, 2.8%); 5. Contain Medicines Harmless Pharmaceutical Products-glenza 40 MG Tablets(enazalu… ($3.3K, 2.6%)
Which ports handle pharmaceutical shipments from India to Anguilla?
Export: AHEMDABAD AIR, SAHAR AIR CARGO ACC (INBOM4), Bombay Air Cargo, SAHAR AIR, AHEMDABAD AIR ACC (INAMD4). Import: ANGUILLA,AI, ANGUILLA AI, ANGUILLAAI.
Why does Anguilla import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $124.9K corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Anguilla?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Anguilla pharmaceutical trade?
$384 per consignment across 325 shipments.
How many Indian pharmaceutical companies export to Anguilla?
8 Indian companies. Largest: SUNNY PHARMA with $76.7K.
How can I find verified Indian pharmaceutical suppliers for Anguilla?
TransData Nexus covers 8 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Anguilla Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 325 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Anguilla identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 325 verified shipments from 8 Indian exporters to 10 Anguilla buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
8 Exporters
10 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists