India to Angola Pharmaceutical Export
Bilateral Trade Intelligence · $134.3M Total Trade · 228 Exporters · 276 Buyers · DGFT Verified · Updated March 2026
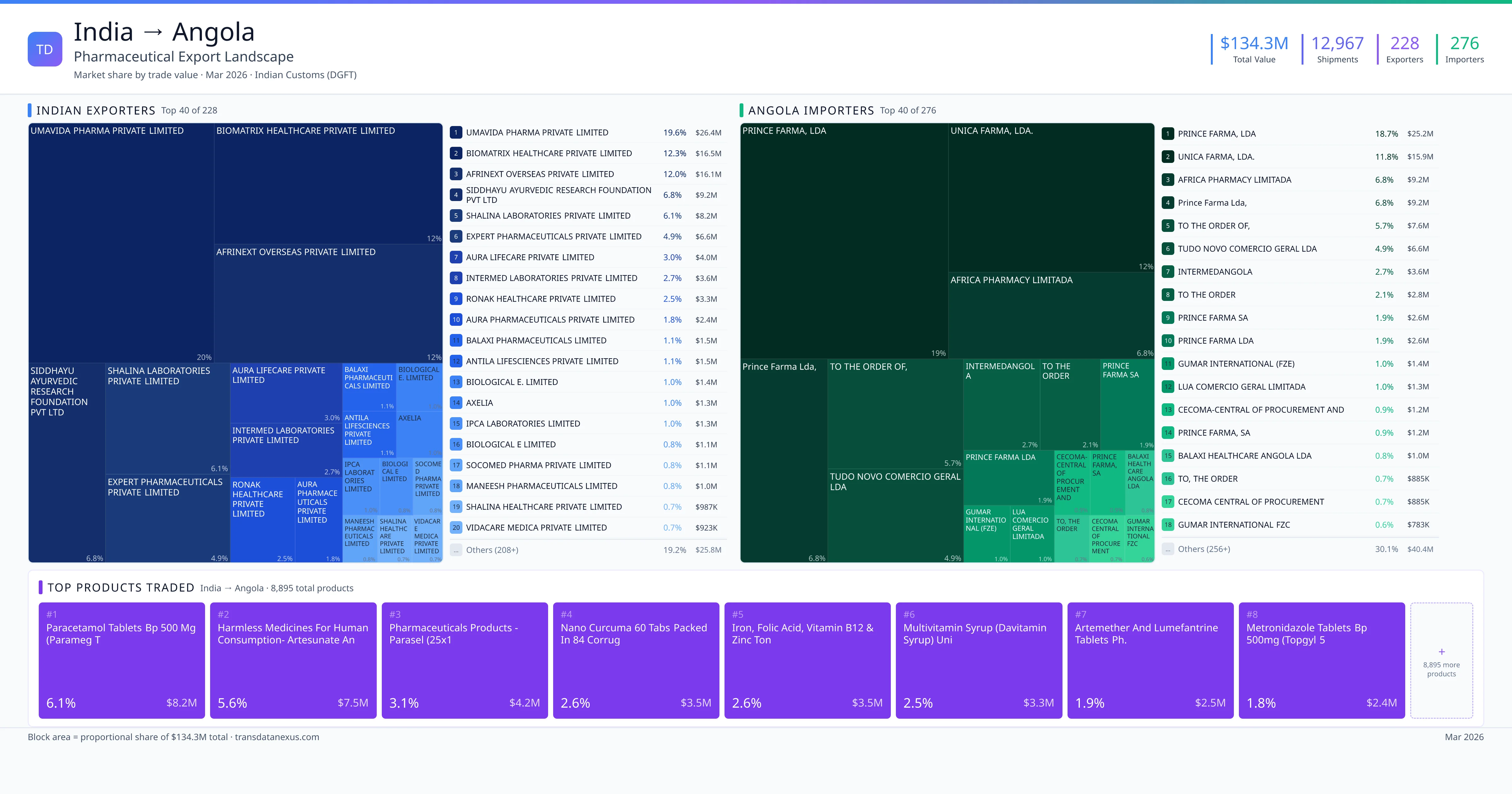
India exported $134.3M worth of pharmaceutical formulations to Angola across 12,967 verified shipments, sourced from 228 Indian exporters supplying 276 Angola buyers. The top exporters are UMAVIDA PHARMA PRIVATE LIMITED ($26.4M) and BIOMATRIX HEALTHCARE PRIVATE LIMITED ($16.5M). The leading products are Paracetamol ($8.2M) and Iron ($3.5M). Average shipment value: $10.4K.
Top Pharmaceutical Formulations — India to Angola
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Paracetamol Tablets BP 500 MG (parameg T | $8.2M | 6.1% |
| 2 | Harmless Medicines for Human Consumption- Artesunate and Amodiaquine Tablets 25… | $7.5M | 5.6% |
| 3 | Pharmaceuticals Products - Parasel (25x1 | $4.2M | 3.1% |
| 4 | Nano Curcuma 60 Tablets Packed in 84 Corrug | $3.5M | 2.6% |
| 5 | Iron, Folic Acid, Vitamin B12 & Zinc Ton | $3.5M | 2.6% |
| 6 | Multivitamin Syrup (davitamin Syrup) Uni | $3.3M | 2.5% |
| 7 | Artemether and Lumefantrine Tablets Ph. | $2.5M | 1.9% |
| 8 | Metronidazole Tablets BP 500mg (topgyl 5 | $2.4M | 1.8% |
| 9 | Pharmaceuticals Products- Parasel(100x10 | $2.4M | 1.8% |
| 10 | Paracetamol Oral Suspension BP 250 Mg/5 | $2.2M | 1.7% |
| 11 | Terbutaline Sulphate, Bromhexine Hydroch | $2.2M | 1.6% |
| 12 | Paracetamol Oral Suspension BP 125 Mg/5 | $2.0M | 1.5% |
| 13 | Ibuprofen Oral Suspension BP 100mg/5ml | $1.8M | 1.4% |
| 14 | Paracetamol Tablets BP 500mg (acemol) | $1.7M | 1.3% |
| 15 | Clindamycin Hydrochloride Capsules USP 3 | $1.5M | 1.1% |
India exports 20+ pharmaceutical formulations to Angola with a combined trade value of $134.3M. Key products include Paracetamol ($8.2M), Iron ($3.5M), Artemether ($2.5M), Metronidazole ($2.4M), Terbutaline ($2.2M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 12,967 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Angola Trade Routes
These are the top pharmaceutical products exported from India to Angola, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Angola buyers, regulatory requirements, and logistics for that specific product corridor. Products include Paracetamol ($8.2M), Iron ($3.5M), Artemether ($2.5M), Metronidazole ($2.4M), Terbutaline ($2.2M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Angola
228 Indian pharmaceutical companies export finished formulations to Angola. Leading exporters include Umavida Pharma Private Limited, Biomatrix Healthcare Private Limited, Afrinext Overseas Private Limited, Siddhayu Ayurvedic Research Foundation Pvt Ltd. The top exporter accounts for 19.6% of total India–Angola pharma exports. Source: Indian Customs (DGFT).
Top Angola Buyers from India
276 companies in Angola import pharmaceutical formulations from India. Top buyers include Prince Farma, Lda, Unica Farma, Lda., Africa Pharmacy Limitada, Prince Farma Lda,. The largest buyer accounts for 18.7% of India–Angola pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Angola Pharmaceutical Shipments
Indian Export Ports
Angola Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Angola Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and Angola has experienced significant growth over the past decade. In 2024, India exported pharmaceutical products worth approximately $73.95 million to Angola, marking a substantial increase from previous years. This surge is attributed to India's reputation for producing high-quality, affordable generic medicines, which have become essential in Angola's healthcare system.
Key milestones in this bilateral trade include the signing of five cooperation agreements in November 2025, covering sectors such as fisheries, transport, foreign relations, environment, and energy. These agreements aim to deepen multi-sectoral cooperation and have positively impacted the pharmaceutical trade by fostering a more conducive environment for business and regulatory collaboration.
2India's Market Position
India holds a dominant position in Angola's pharmaceutical imports, supplying a significant portion of the country's medicinal needs. In 2024, Angola's total imports of pharmaceutical products were valued at $248.44 million, with India accounting for approximately $73.95 million of this total. This underscores India's strategic importance in Angola's healthcare sector, providing essential medicines that are both affordable and effective.
3Recent Developments
Between 2024 and 2026, several developments have influenced the India-Angola pharmaceutical trade corridor. Notably, in November 2025, Angola and India signed five cooperation agreements covering strategic sectors, including health and local pharmaceutical production. These agreements aim to enhance bilateral relations and have positively impacted the pharmaceutical trade by fostering a more conducive environment for business and regulatory collaboration.
Additionally, in February 2026, India and the European Union concluded negotiations for a Free Trade Agreement (FTA), which is expected to reshape global pharmaceutical trade dynamics. While this agreement primarily focuses on EU-India relations, it may indirectly affect the India-Angola trade corridor by influencing global trade patterns and regulatory standards.
Angola Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
To export finished pharmaceutical formulations to Angola, Indian companies must navigate a structured registration process overseen by the Regulatory Agency for Medicines and Healthcare Technologies (ARMED), operating under the Ministry of Health. This process ensures that all imported medicines meet the country's safety, efficacy, and quality standards.
Drug Registration and Marketing Authorization
The registration process involves several key steps:
1. Application Submission: Companies must submit a comprehensive application to ARMED, including detailed product information, manufacturing details, and evidence of compliance with international standards.
2. Laboratory Testing: Submitted products undergo laboratory tests to verify their compliance with established norms, standards, procedures, and regulations.
3. Labeling Requirements: All products must have labels in Portuguese, accompanied by consumer medicine information leaflets and instructions for medical devices.
4. Approval and Registration: Upon successful evaluation, ARMED grants marketing authorization, allowing the product to be imported and distributed within Angola.
Dossier Format Requirements
While specific dossier format requirements are not explicitly detailed in the available sources, it is common for regulatory authorities to prefer standardized formats such as the Common Technical Document (CTD) or the ASEAN Common Technical Dossier (ACTD). Companies should prepare their dossiers in these formats to facilitate the review process.
Approval Timeline and Fees
The approval timeline can vary based on the completeness of the application and the complexity of the product. Specific timelines and fee structures are not detailed in the available sources. Companies are advised to consult directly with ARMED or local regulatory experts to obtain the most accurate and current information.
2GMP & Facility Requirements
ARMED mandates that all pharmaceutical products imported into Angola comply with Good Manufacturing Practices (GMP) to ensure product quality and safety. Manufacturing facilities must adhere to GMP standards, which encompass aspects such as documentation, sanitation, equipment validation, and complaint handling.
While specific details about the inspection process and mutual recognition agreements are not provided in the available sources, it is standard practice for regulatory authorities to conduct inspections or accept certifications from recognized international bodies to verify GMP compliance. Companies should ensure that their manufacturing facilities are certified by reputable organizations and be prepared for potential inspections by ARMED.
3Import Documentation
To import pharmaceutical products into Angola, companies must ensure that their local distributors hold the necessary import registration status for pharmaceuticals. The required documentation typically includes:
- Import License: Only registered companies can apply for an import license, which is mandatory for importing pharmaceuticals.
- Certificates of Pharmaceutical Products (CPP): These certificates attest to the product's compliance with international standards.
- Certificate of Analysis (CoA): This document provides detailed information about the product's composition and quality.
- GMP Certificate: This certificate confirms that the manufacturing facility adheres to GMP standards.
Additionally, customs procedures require:
- Original Air Way Bill (AWB) or Bill of Lading (BL): These documents serve as proof of shipment.
- Original Commercial Invoice: This invoice should list Harmonized Tariff Schedule (HTS) codes for each item.
- Packing List: This list details the contents of each shipment.
Ensuring that all documentation is accurate and complete is crucial to facilitate smooth customs clearance and avoid potential delays.
Product Categories & Therapeutic Trends — India to Angola
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Angola are predominantly concentrated in analgesics and antimalarials, reflecting Angola's pressing healthcare needs. Paracetamol tablets, a common analgesic, lead the export list with a value of $8.2 million, accounting for 6.1% of total exports. This aligns with Angola's high incidence of infectious diseases, including malaria, which remains a significant public health concern. The export of artesunate and amodiaquine tablets, valued at $7.5 million (5.6% of total exports), underscores the critical demand for antimalarial treatments. In 2024, Angola reported over 7 million malaria cases and approximately 11,000 deaths, primarily affecting children and pregnant women. (afro.who.int)
The prominence of these therapeutic categories is driven by Angola's substantial disease burden and the limited capacity of its domestic pharmaceutical industry. The country's healthcare infrastructure faces challenges such as outdated facilities and a shortage of skilled professionals, leading to a heavy reliance on imported medicines to meet the population's needs.
2Emerging Opportunities
Angola's pharmaceutical market is experiencing growth, with revenues projected to reach $321.9 million in 2025, reflecting a compound annual growth rate (CAGR) of 3.77% from 2025 to 2030. This expansion presents opportunities for Indian exporters to introduce biosimilars and generic drugs, particularly as Angola seeks to reduce its dependency on imported branded medications. The government's initiatives to regulate the pharmaceutical sector and promote local production through tax incentives and infrastructure investments further create a conducive environment for such products.
3Demand Drivers
Angola's disease burden, characterized by high rates of malaria, tuberculosis, and non-communicable diseases, significantly drives the demand for pharmaceutical imports. The country's healthcare spending per capita was $101.26 in 2022, marking a 51.02% increase from 2021, indicating a growing commitment to improving health services. However, challenges such as limited infrastructure and a shortage of skilled professionals persist, necessitating continued reliance on imported pharmaceuticals to meet the population's needs.
Trade Policy & Tariff Intelligence — India and Angola
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Angola's tariff structure for pharmaceutical formulations, classified under HS codes 3003 and 3004, is not publicly detailed. The absence of specific tariff rates in available sources suggests that these products may be subject to standard import duties applicable to pharmaceutical goods. However, without explicit data, it's challenging to determine the exact Most-Favored-Nation (MFN) tariff rates for these products.
Regarding preferential duty rates, there is no publicly available information indicating any preferential treatment for pharmaceutical imports from India into Angola. This implies that Indian pharmaceutical products are likely subject to standard import duties without any reductions or exemptions under existing trade agreements.
2Trade Agreements
As of March 2026, there is no publicly available information indicating the existence of a Free Trade Agreement (FTA) between India and Angola. The absence of such an agreement suggests that bilateral trade, including pharmaceutical exports from India to Angola, operates under standard World Trade Organization (WTO) rules without preferential treatment.
In May 2025, Angola and India signed several cooperation agreements, including a Memorandum of Understanding in the field of Traditional Medicine. While this agreement may enhance collaboration in the pharmaceutical sector, it does not appear to establish preferential trade terms or tariff reductions for pharmaceutical products.
3IP & Patent Landscape
Angola's intellectual property (IP) framework, including patent provisions, is not extensively detailed in publicly available sources. However, the National Drug Regulatory Authority (WHO-GMP) in Angola oversees the regulation of pharmaceutical products, which may include considerations related to IP and patent rights. The impact of Angola's IP provisions on the importation of generic drugs from India is not clearly defined in the available information. Therefore, without explicit details, it's challenging to assess how Angola's IP and patent provisions affect the entry of Indian generic pharmaceutical products into the Angolan market.
Supply Chain & Logistics — India to Angola Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to Angola primarily utilize sea freight due to its cost-effectiveness for large volumes. The journey typically spans approximately 7,500 kilometers, with transit times ranging from 25 to 35 days, depending on factors such as shipping line schedules, port congestion, and weather conditions.
Air freight offers a faster alternative, with the quickest flights taking about 16 hours and 12 minutes, departing from Chhatrapati Shivaji International Airport (BOM) in Mumbai and arriving at Quatro de Fevereiro International Airport (LAD) in Luanda. However, such flights are infrequent, operating every 1-2 weeks, and are generally more expensive than sea freight.
The Red Sea disruption, which occurred in early 2026, had a significant impact on global shipping routes, including those from India to Angola. This event led to increased transit times and freight costs due to the need for rerouted vessels and additional fuel consumption. While the situation has since stabilized, residual effects may continue to influence shipping schedules and costs in the short term.
2Port Infrastructure
In India, key export ports handling pharmaceutical shipments include Nhava Sheva Sea Port (INNSA1) in Maharashtra, which accounts for 28.2% of the total shipments, and Ahmedabad ICD, contributing 4.3%. These ports are well-equipped to handle large volumes of cargo and are connected to major shipping routes facilitating efficient export operations.
In Angola, the primary import port is Luanda, handling 57.7% of the shipments. Luanda's port infrastructure is capable of accommodating various types of cargo, including pharmaceuticals, and is supported by customs facilities that ensure compliance with national regulations.
3Cold Chain & Compliance
Maintaining the integrity of temperature-sensitive pharmaceutical products during transit from India to Angola is paramount. These products require strict adherence to Good Distribution Practices (GDP) and Good Manufacturing Practices (GMP) to ensure safety, efficacy, and quality.
Temperature-controlled logistics involve using insulated packaging, such as thermal boxes, and refrigerated transport to maintain the required temperature ranges. Continuous temperature monitoring and data logging are essential to document compliance and ensure product integrity throughout the supply chain.
In Angola, the National Drug Regulatory Authority, adhering to WHO-GMP standards, oversees the registration and licensing of pharmaceutical products. This includes ensuring that imported medicines meet established norms, standards, and procedures, and that they are labeled in Portuguese, as per Presidential Decree No. 315/20 of 17 December 2020.
By adhering to these stringent cold chain management practices and regulatory requirements, pharmaceutical shipments from India to Angola can maintain product quality and comply with both Indian and Angolan regulations.
Market Opportunity Assessment — Angola for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Angola's pharmaceutical market is projected to reach a revenue of approximately $321.90 million in 2025, with an expected compound annual growth rate (CAGR) of 3.77% from 2025 to 2030, culminating in a market volume of $387.38 million by 2030. This growth is driven by factors such as population expansion, urbanization, and increasing prevalence of chronic and infectious diseases. The market is heavily import-dependent, with a significant reliance on foreign pharmaceutical products due to limited local manufacturing capabilities. Generic drugs hold a substantial share of the market, offering cost-effective alternatives to branded medications and contributing to the overall market expansion.
2Healthcare System
Angola's healthcare system faces challenges, including underdeveloped infrastructure and limited access to quality care for a significant portion of the population. The government has been making efforts to improve healthcare access and quality, but the system remains underfunded and underdeveloped, creating significant gaps in service delivery. Health spending as a percentage of GDP was 2.93% in 2022, indicating a need for increased investment in the sector. The government has been making efforts to improve healthcare access and quality, but the system remains underfunded and underdeveloped, creating significant gaps in service delivery.
3Opportunity for Indian Exporters
Indian pharmaceutical exporters have a significant opportunity to expand their presence in Angola's market, particularly in the generic drug segment. The top 10 products exported from India to Angola include Paracetamol Tablets BP 500 mg, Artesunate and Amodiaquine Tablets, and Multivitamin Syrup, indicating a strong demand for essential and over-the-counter medications. The growth forecast for Indian pharmaceutical imports into Angola from 2025 to 2028 is positive, aligning with the overall market expansion. Given the import-dependent nature of Angola's pharmaceutical market and the increasing demand for affordable healthcare solutions, Indian exporters are well-positioned to meet the country's needs and capitalize on this growth trajectory.
Competitive Landscape — India vs Other Pharmaceutical Suppliers to Angola
Competing origins, India's edge, challenges and threats
1Competing Origins
Angola's pharmaceutical market is supplied by a diverse array of international and domestic sources. In 2024, Angola imported pharmaceutical products valued at approximately $248.44 million. India emerged as the dominant supplier, accounting for a significant portion of these imports. Other notable contributors include China, the European Union (EU), and domestic manufacturers.
China's pharmaceutical exports to Angola are substantial, with a notable presence in the market. For instance, in 2023, Angola imported hygienic or pharmaceutical articles of vulcanized rubber from China valued at $109.36K, representing 50,889 kg. The EU, particularly Portugal, also plays a significant role. In 2024, Angola imported pharmaceutical products from Portugal amounting to $66.15 million. Domestic manufacturers, such as Elnor Pharma, contribute to the local supply chain by distributing both brand and generic medicines.
While precise market share percentages are not readily available, India's substantial export value and the number of shipments suggest a leading position in Angola's pharmaceutical imports. The combined contributions from China, the EU, and domestic manufacturers indicate a competitive and diversified supply landscape.
2India's Competitive Edge
India's pharmaceutical industry offers several competitive advantages in the Angolan market. The total export value from India to Angola between 2022 and 2026 was $134.3 million, with an average shipment value of $10,000. This indicates a broad portfolio catering to various therapeutic needs.
A key strength lies in India's cost competitiveness, enabling the provision of affordable medicines without compromising quality. Additionally, India's adherence to WHO-GMP standards ensures that products meet international quality benchmarks. The widespread use of English facilitates effective communication and collaboration with Angolan partners. These factors collectively enhance India's appeal as a reliable pharmaceutical supplier to Angola.
3Challenges & Threats
Despite its strengths, India's pharmaceutical exports to Angola face several challenges. Regulatory tightening by Angola's National Drug Regulatory Authority, which adheres to WHO-GMP standards, may impose stricter compliance requirements on foreign suppliers. Competition from China, with its cost-effective manufacturing capabilities, poses a threat to India's market share. Additionally, perceptions regarding the quality of Indian pharmaceuticals may impact their acceptance among Angolan consumers and healthcare providers. Addressing these challenges is crucial for maintaining and enhancing India's position in the Angolan pharmaceutical market.
FAQ — India to Angola Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Angola?
India exported pharmaceuticals worth $134.3M to Angola across 12,967 verified shipments.
Who are the top Indian pharmaceutical exporters to Angola?
1. UMAVIDA PHARMA PRIVATE LIMITED — $26.4M. 2. BIOMATRIX HEALTHCARE PRIVATE LIMITED — $16.5M. 3. AFRINEXT OVERSEAS PRIVATE LIMITED — $16.1M. Total: 228 suppliers.
Which companies in Angola import pharmaceuticals from India?
1. PRINCE FARMA, LDA — $25.2M. 2. UNICA FARMA, LDA. — $15.9M. 3. AFRICA PHARMACY LIMITADA — $9.2M. 276 buyers total.
What pharmaceutical products does India export most to Angola?
1. Paracetamol Tablets BP 500 MG (parameg T ($8.2M, 6.1%); 2. Harmless Medicines for Human Consumption- Artesunate and Amodiaquine Tablets 25… ($7.5M, 5.6%); 3. Pharmaceuticals Products - Parasel (25x1 ($4.2M, 3.1%); 4. Nano Curcuma 60 Tablets Packed in 84 Corrug ($3.5M, 2.6%); 5. Iron, Folic Acid, Vitamin B12 & Zinc Ton ($3.5M, 2.6%)
Which ports handle pharmaceutical shipments from India to Angola?
Export: AHEMDABAD ICD, NHAVA SHEVA SEA (INNSA1), CONCOR ICD/MIHAN, COCHIN SEA, JNPT/ NHAVA SHEVA SEA. Import: Luanda, LUANDA, LUANDA -, Kinshasa, Lobito.
Why does Angola import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $134.3M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Angola?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Angola pharmaceutical trade?
$10.4K per consignment across 12,967 shipments.
How many Indian pharmaceutical companies export to Angola?
228 Indian companies. Largest: UMAVIDA PHARMA PRIVATE LIMITED with $26.4M.
How can I find verified Indian pharmaceutical suppliers for Angola?
TransData Nexus covers 228 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Angola Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 12,967 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Angola identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 12,967 verified shipments from 228 Indian exporters to 276 Angola buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
228 Exporters
276 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists