India to Algeria Pharmaceutical Export
Bilateral Trade Intelligence · $76.2M Total Trade · 105 Exporters · 170 Buyers · DGFT Verified · Updated March 2026
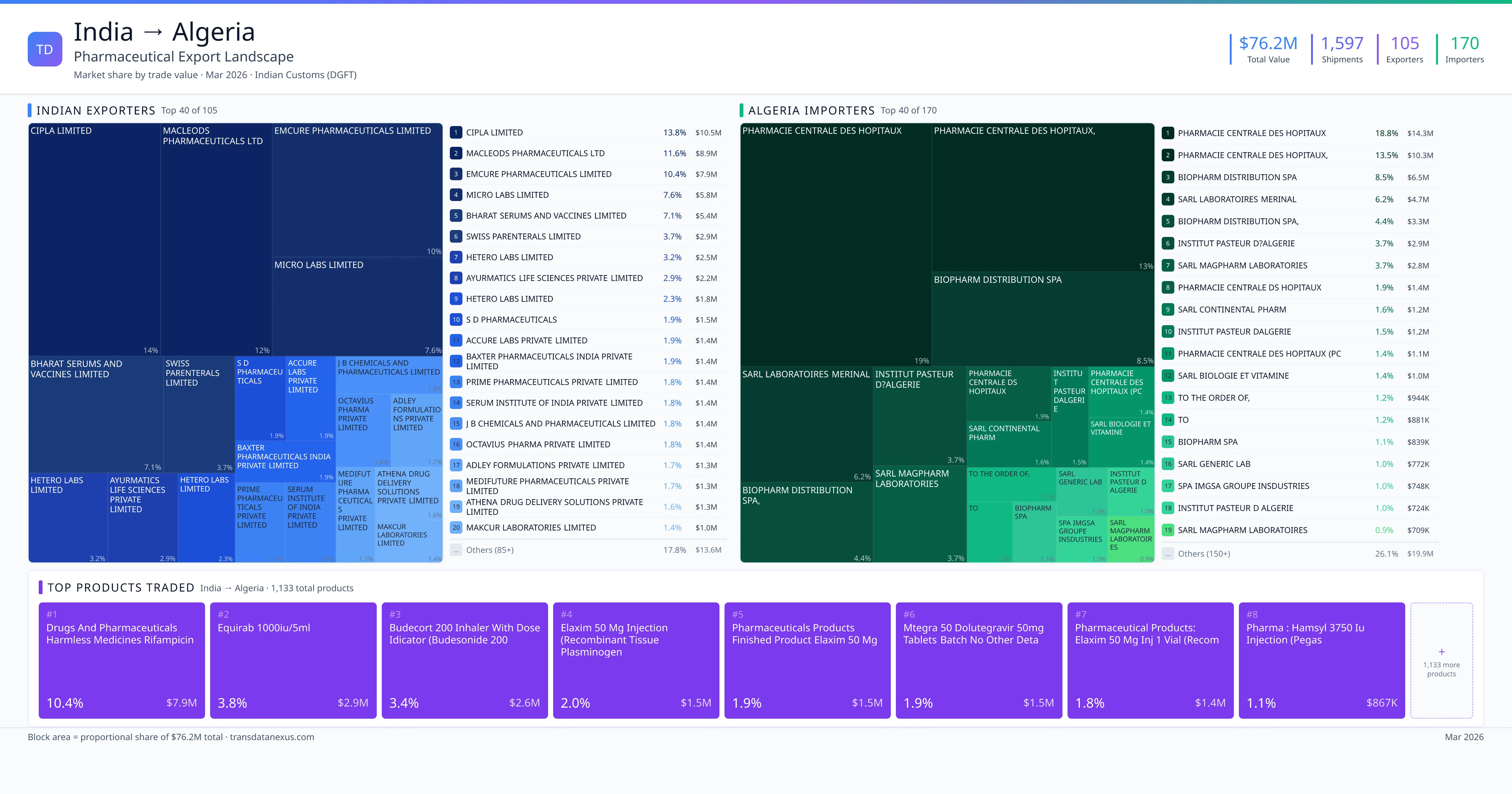
India exported $76.2M worth of pharmaceutical formulations to Algeria across 1,597 verified shipments, sourced from 105 Indian exporters supplying 170 Algeria buyers. The top exporters are CIPLA LIMITED ($10.5M) and MACLEODS PHARMACEUTICALS LTD ($8.9M). The leading products are Dolutegravir ($1.5M) and Tenofovir ($759K). Average shipment value: $47.7K.
Top Pharmaceutical Formulations — India to Algeria
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Drugs and Pharmaceuticals Harmless Medicines Rifampicin Capsules BP 150 MG | $7.9M | 10.4% |
| 2 | Equirab 1000iu/5ml | $2.9M | 3.8% |
| 3 | Budecort 200 Inhaler with Dose Idicator (budesonide 200mcg) 1 X 200 Md | $2.6M | 3.4% |
| 4 | Elaxim 50 MG Injection (recombinant Tissue Plasminogen Activator (tnk-tpa) Inje… | $1.5M | 2.0% |
| 5 | Pharmaceuticals Products Finished Product Elaxim 50 MG Injection 1 Vial Recombi… | $1.5M | 1.9% |
| 6 | Mtegra 50 Dolutegravir 50mg Tablets Batch No Other Details As Per Inv Packing L… | $1.5M | 1.9% |
| 7 | Pharmaceutical Products: Elaxim 50 MG Injection 1 Vial (recombinant Tissue Plas… | $1.4M | 1.8% |
| 8 | Pharma : Hamsyl 3750 IU Injection (pegas | $867.0K | 1.1% |
| 9 | Rhoclone 300mcg (anti Rho-d Immunoglobulin Injection (monoclonal) (liquid Injec… | $843.4K | 1.1% |
| 10 | Elaxim 50 MG Injection (recombinant Tiss | $795.6K | 1.0% |
| 11 | Foracort Hfa 200 Inhaler + (formoterol Fumarate 6mcg + Budesonide 200mcg) | $792.4K | 1.0% |
| 12 | Tenofovir Dis Emtricitab Te 300mg Em 200mg Tablet 30s Free Units 270 Tablet | $758.9K | 1.0% |
| 13 | Foracort Hfa 200 Inhaler + (formoterol F | $669.3K | 0.9% |
| 14 | Zecuf Oral Syrup Concentrate Alg Exp | $627.3K | 0.8% |
| 15 | Mesalamine Pellets | $598.4K | 0.8% |
India exports 16+ pharmaceutical formulations to Algeria with a combined trade value of $76.2M. Key products include Dolutegravir ($1.5M), Tenofovir ($759K), Mesalamine ($598K). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 1,597 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Algeria Trade Routes
These are the top pharmaceutical products exported from India to Algeria, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Algeria buyers, regulatory requirements, and logistics for that specific product corridor. Products include Dolutegravir ($1.5M), Tenofovir ($759K), Mesalamine ($598K) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Algeria
105 Indian pharmaceutical companies export finished formulations to Algeria. Leading exporters include Cipla Limited, Macleods Pharmaceuticals Ltd, Emcure Pharmaceuticals Limited, Micro Labs Limited. The top exporter accounts for 13.8% of total India–Algeria pharma exports. Source: Indian Customs (DGFT).
Top Algeria Buyers from India
170 companies in Algeria import pharmaceutical formulations from India. Top buyers include Pharmacie Centrale Des Hopitaux, Pharmacie Centrale Des Hopitaux,, Biopharm Distribution Spa, Sarl Laboratoires Merinal. The largest buyer accounts for 18.8% of India–Algeria pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Algeria Pharmaceutical Shipments
Indian Export Ports
Algeria Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Algeria Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and Algeria has experienced significant growth over the past decade. In 2017, India's pharmaceutical exports to Algeria were valued at approximately $79.23 million. By 2024, this figure had increased to $77.38 million, indicating a steady upward trajectory in bilateral trade.
A pivotal milestone in this relationship occurred in January 2026, when the India-Algeria Pharmaceutical Business Forum was held in Algiers. This forum saw the participation of nearly 100 Indian pharmaceutical companies, underscoring the growing interest and commitment to strengthening ties in the sector. The event facilitated discussions on joint investment projects and technology transfer, aiming to position Algeria as a regional hub for pharmaceutical production and export. (news.radioalgerie.dz)
The forum's success was further highlighted by the presence of over 65 Indian pharmaceutical companies, reflecting the strategic importance both nations place on this partnership. The discussions centered on leveraging Algeria's strategic geographical position and infrastructure to access broader African and Mediterranean markets. (news.radioalgerie.dz)
2India's Market Position
India's pharmaceutical exports to Algeria have consistently been a significant component of the country's total exports to Algeria. In 2024, pharmaceutical products accounted for approximately $77.38 million of the total $943.71 million in exports to Algeria. This underscores the strategic importance of the pharmaceutical sector in India's trade relations with Algeria.
The partnership is mutually beneficial, with India recognized as the "pharmacy of the world" and Algeria positioning itself as a growing pharmaceutical hub in Africa. The collaboration aims to enhance local production capabilities in Algeria, reduce dependency on imports, and expand market access for Indian pharmaceutical companies in the African continent. (news.radioalgerie.dz)
3Recent Developments
Between 2024 and 2026, several key developments have further solidified the India-Algeria pharmaceutical trade corridor. In January 2026, the India-Algeria Pharmaceutical Business Forum was held in Algiers, with nearly 100 Indian pharmaceutical companies participating. The forum focused on joint investment projects and technology transfer, aiming to establish Algeria as a regional hub for pharmaceutical production and export. (news.radioalgerie.dz)
In February 2026, a multi-product Indian business delegation, led by the Federation of Indian Export Organisations (FIEO), visited Algeria to strengthen bilateral trade ties and foster long-term business partnerships. The delegation included representatives from the pharmaceutical sector, highlighting the ongoing commitment to enhancing cooperation in this field.
These initiatives reflect a concerted effort by both nations to deepen their pharmaceutical trade relations, focusing on joint ventures, technology transfer, and expanding market access in regional and African markets.
Algeria Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
To market pharmaceutical products in Algeria, Indian companies must obtain registration from the National Agency for Pharmaceutical Products (ANPP). The registration process involves submitting a comprehensive dossier that includes product information, clinical data, and certificates such as the Certificate of Pharmaceutical Product (CPP) and Good Manufacturing Practice (GMP) certificates. The dossier should be presented in the Common Technical Document (CTD) format, which is internationally recognized and facilitates the evaluation process. The ANPP evaluates the dossier to ensure the product's safety, efficacy, and quality before granting marketing authorization.
In December 2025, the ANPP streamlined the drug registration process to enhance efficiency. Under the new system, pharmaceutical product registration files are presented to the Drug Registration Committee only once. If the committee raises objections, the registration file is suspended until the pharmaceutical company addresses and resolves all concerns. Once the concerns are resolved, the registration decision is issued without requiring the file to be re-submitted to the Registration Committee. (dzwatch.dz)
2GMP & Facility Requirements
Algeria requires pharmaceutical manufacturing facilities to comply with Good Manufacturing Practice (GMP) standards recognized by the ANPP. The ANPP conducts inspections to ensure that manufacturing sites adhere to these standards, which are essential for ensuring the quality and safety of pharmaceutical products. In September 2025, the Ministry of Pharmaceutical Industry updated the authorization framework for pharmaceutical manufacturing establishments, introducing new documentation and procedural requirements. The authorization process now includes a two-stage approval: preliminary approval for project initiation and activity launching approval for operational commencement. Noncompliance with the new regulations may lead to application rejections, affecting project timelines and operational readiness.
3Import Documentation
To import pharmaceutical products into Algeria, companies must obtain an import authorization from the Ministry of Pharmaceutical Industry. The application for authorization must be accompanied by a file containing various documents, including the Certificate of Pharmaceutical Product (CPP), Certificate of Analysis (CoA), and GMP certificates. These documents ensure that the imported products meet the required quality standards. The ANPP evaluates the documentation to ensure compliance with Algerian regulations before granting import authorization. (miph.gov.dz)
Product Categories & Therapeutic Trends — India to Algeria
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Algeria are predominantly concentrated in therapeutic areas addressing infectious diseases, respiratory conditions, and cardiovascular events. The leading product, Rifampicin Capsules BP 150 mg, accounting for $7.9 million (10.4% of total exports), is a cornerstone in the treatment of tuberculosis, a disease with a significant prevalence in Algeria. The prominence of respiratory medications, exemplified by the Budecort 200 Inhaler (Budesonide 200mcg), reflects the high incidence of asthma and chronic obstructive pulmonary disease (COPD) in the region. Additionally, cardiovascular treatments like recombinant tissue plasminogen activator (TNK-TPA) injections are critical in managing acute myocardial infarctions, aligning with Algeria's efforts to enhance acute care services.
These therapeutic categories dominate due to Algeria's substantial burden of infectious diseases, respiratory disorders, and cardiovascular events. The Algerian government's focus on improving healthcare infrastructure and access to essential medicines has led to increased demand for these treatments. The National Drug Regulatory Authority's adherence to WHO-GMP standards ensures the quality and efficacy of imported pharmaceuticals, further driving the demand for these specific therapeutic areas.
2Emerging Opportunities
Algeria's pharmaceutical market is witnessing a surge in demand for generic drugs, driven by government initiatives to reduce healthcare costs and promote domestic production. The establishment of the Ministry of Pharmaceutical Industry (MOPI) and the National Agency for Pharmaceutical Products (ANPP) has streamlined drug registration processes, significantly reducing approval times for generic medicines. This regulatory facilitation presents Indian pharmaceutical companies with opportunities to introduce cost-effective generics, particularly in areas where patent protections are expiring, known as patent cliffs. Additionally, Algeria's growing focus on biosimilars offers Indian exporters a chance to supply high-quality, affordable alternatives to expensive biologic therapies, catering to the needs of patients requiring such treatments.
3Demand Drivers
Algeria's disease burden, characterized by a high prevalence of chronic conditions such as diabetes, cardiovascular diseases, and respiratory disorders, significantly drives the demand for pharmaceutical imports. The economic burden of managing these diseases is substantial; for instance, the annual median cost of treating diabetic foot ulcers is estimated at €3,790.97 per patient, highlighting the financial strain on the healthcare system. Despite efforts to increase domestic production, Algeria continues to rely heavily on imported pharmaceuticals, with imports accounting for 70% of the market share. This dependency underscores the critical role of international suppliers, like India, in meeting the country's healthcare needs. Furthermore, Algeria's healthcare spending per capita has been on a declining trend, from $242.07 in 2019 to $179.70 in 2022, indicating budgetary constraints that may limit the affordability of imported medicines. This scenario presents both challenges and opportunities for Indian pharmaceutical exporters to offer cost-effective solutions that align with Algeria's economic realities.
Trade Policy & Tariff Intelligence — India and Algeria
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Algeria's tariff structure for pharmaceutical formulations, particularly those classified under HS codes 3003 and 3004, is influenced by its trade policies and international agreements. While specific Most-Favored-Nation (MFN) tariff rates for these products are not readily available, Algeria's MFN tariff rates for pharmaceutical products typically range from 0% to 5%, depending on the specific product and its classification. This range aligns with Algeria's commitment to facilitating access to essential medicines.
Regarding preferential duty rates, Algeria is a member of the Arab Maghreb Union (UMA), which aims to promote economic integration among member states. However, the UMA has been largely inactive in recent years, and there are no significant preferential duty rates currently in place for pharmaceutical products between India and Algeria. Additionally, Algeria has been known to implement temporary anti-dumping measures on certain pharmaceutical imports to protect domestic industries. These measures can include additional duties or restrictions on specific products deemed to be sold at unfairly low prices. It is advisable for Indian pharmaceutical exporters to monitor Algeria's trade policies regularly to stay informed about any such measures.
2Trade Agreements
As of March 2026, there is no active Free Trade Agreement (FTA) between India and Algeria. The absence of such an agreement means that pharmaceutical products exported from India to Algeria are subject to standard MFN tariff rates without any preferential treatment. While there have been discussions about enhancing bilateral trade relations, no concrete steps have been taken to establish an FTA. The lack of an FTA can impact the competitiveness of Indian pharmaceutical products in the Algerian market, as they do not benefit from reduced tariffs that could be available under preferential trade agreements.
3IP & Patent Landscape
Algeria's intellectual property (IP) framework, particularly concerning patents, is designed to protect both domestic and foreign innovations. The country adheres to international IP agreements, including the Trade-Related Aspects of Intellectual Property Rights (TRIPS) Agreement. This adherence means that patents granted in Algeria are generally recognized and enforced, providing protection for pharmaceutical inventions. However, Algeria has provisions for compulsory licensing, allowing the government to grant licenses to third parties without the consent of the patent holder under specific circumstances, such as public health emergencies. This mechanism can affect the entry of generic drugs into the market, as it may facilitate the production of generic versions of patented medicines. Indian pharmaceutical companies should be aware of these provisions when considering market entry strategies in Algeria, as they may influence the competitive landscape for generic drugs.
Market Opportunity Assessment — Algeria for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Algeria's pharmaceutical market is projected to reach a revenue of US$1.06 billion by 2025, with an anticipated compound annual growth rate (CAGR) of 4.03% from 2025 to 2030, resulting in a market volume of US$1.29 billion by 2030. The market is characterized by a significant presence of generic drugs, driven by government initiatives to reduce healthcare costs and promote domestic production.
2Healthcare System
Algeria provides universal healthcare coverage to its citizens through two main public health insurance schemes: the Caisse Nationale de la Sécurité Sociale des Travailleurs Salariés (CNAS) for salaried employees and their dependents, and the Caisse Nationale des Assurances Sociales des Non-Salariés (CASNOS) for non-salaried professionals. These programs offer free access to medical care and medications at state-run hospitals and clinics. In January 2026, the government introduced an electronic card granting free access to medicines for citizens without social coverage, further expanding healthcare access. (dzair-tube.dz)
3Opportunity for Indian Exporters
The Algerian pharmaceutical market presents significant opportunities for Indian exporters, particularly in the generic drug segment. With a projected market volume of US$1.29 billion by 2030, the demand for cost-effective medications is expected to rise, aligning with India's strengths in generic drug manufacturing. The government's focus on reducing healthcare costs and promoting domestic production creates a favorable environment for Indian pharmaceutical companies to expand their presence in Algeria.
FAQ — India to Algeria Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Algeria?
India exported pharmaceuticals worth $76.2M to Algeria across 1,597 verified shipments.
Who are the top Indian pharmaceutical exporters to Algeria?
1. CIPLA LIMITED — $10.5M. 2. MACLEODS PHARMACEUTICALS LTD — $8.9M. 3. EMCURE PHARMACEUTICALS LIMITED — $7.9M. Total: 105 suppliers.
Which companies in Algeria import pharmaceuticals from India?
1. PHARMACIE CENTRALE DES HOPITAUX — $14.3M. 2. PHARMACIE CENTRALE DES HOPITAUX, — $10.3M. 3. BIOPHARM DISTRIBUTION SPA — $6.5M. 170 buyers total.
What pharmaceutical products does India export most to Algeria?
1. Drugs and Pharmaceuticals Harmless Medicines Rifampicin Capsules BP 150 MG ($7.9M, 10.4%); 2. Equirab 1000iu/5ml ($2.9M, 3.8%); 3. Budecort 200 Inhaler with Dose Idicator (budesonide 200mcg) 1 X 200 Md ($2.6M, 3.4%); 4. Elaxim 50 MG Injection (recombinant Tissue Plasminogen Activator (tnk-tpa) Inje… ($1.5M, 2.0%); 5. Pharmaceuticals Products Finished Product Elaxim 50 MG Injection 1 Vial Recombi… ($1.5M, 1.9%)
Which ports handle pharmaceutical shipments from India to Algeria?
Export: SAHAR AIR, SAHAR AIR CARGO ACC (INBOM4), DELHI AIR, NHAVA SHEVA SEA (INNSA1), AHEMDABAD AIR. Import: ALGIERS, Alger, ALGER, Algiers, Skikda (Ex Philippev.
Why does Algeria import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $76.2M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Algeria?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Algeria pharmaceutical trade?
$47.7K per consignment across 1,597 shipments.
How many Indian pharmaceutical companies export to Algeria?
105 Indian companies. Largest: CIPLA LIMITED with $10.5M.
How can I find verified Indian pharmaceutical suppliers for Algeria?
TransData Nexus covers 105 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Algeria Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 1,597 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Algeria identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 1,597 verified shipments from 105 Indian exporters to 170 Algeria buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
105 Exporters
170 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists