India to Albania Pharmaceutical Export
Bilateral Trade Intelligence · $1.9M Total Trade · 44 Exporters · 51 Buyers · DGFT Verified · Updated March 2026
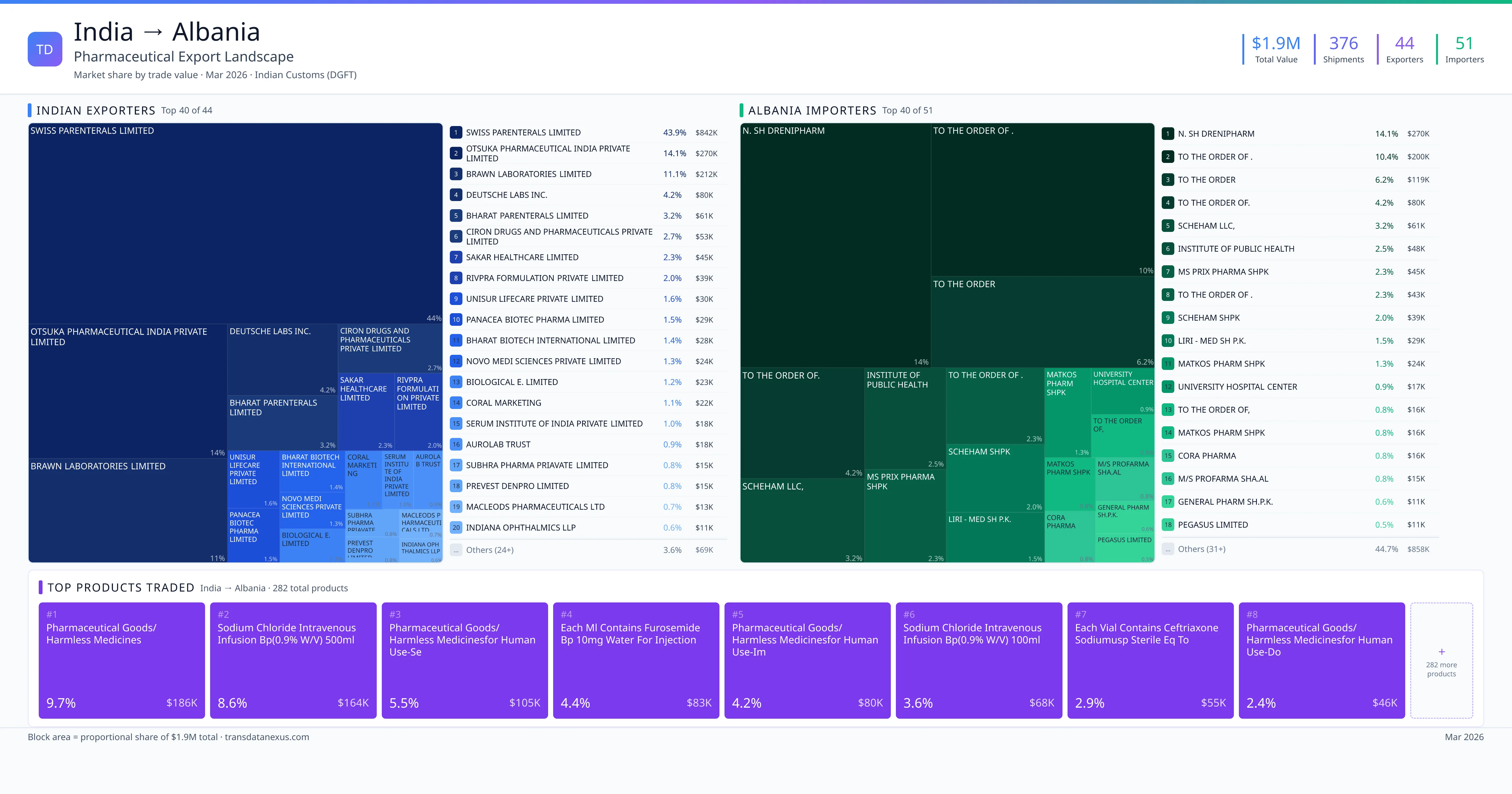
India exported $1.9M worth of pharmaceutical formulations to Albania across 376 verified shipments, sourced from 44 Indian exporters supplying 51 Albania buyers. The top exporters are SWISS PARENTERALS LIMITED ($842.3K) and OTSUKA PHARMACEUTICAL INDIA PRIVATE LIMITED ($269.9K). The leading products are Sodium ($164K) and Ceftriaxone ($45K). Average shipment value: $5.1K.
Top Pharmaceutical Formulations — India to Albania
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Pharmaceutical Goods/harmless Medicines | $185.5K | 9.7% |
| 2 | Sodium Chloride Intravenous Infusion Bp(0.9% W/v) 500ml Nipple Head Plastic Bot… | $164.3K | 8.6% |
| 3 | Each ML Contains Furosemide BP 10mg Water for Injection BP Q S | $83.4K | 4.4% |
| 4 | Pharmaceutical Goods/harmless Medicinesfor Human Use-imipenem and Cilastatin fo… | $80.4K | 4.2% |
| 5 | Sodium Chloride Intravenous Infusion Bp(0.9% W/v) 100ml Nipple Head Plastic Bot… | $68.2K | 3.6% |
| 6 | Pharmaceutical Goods/harmless Medicinesfor Human Use-sevoflurane USP 250ml (1,0… | $66.1K | 3.4% |
| 7 | Each Vial Contains Ceftriaxone Sodiumusp Sterile Eq to Ceftriaxone 1 Gm | $55.4K | 2.9% |
| 8 | Pharmaceutical Goods/harmless Medicinesfor Human Use-donalgin 2 ML Metamizole S… | $46.3K | 2.4% |
| 9 | Ceftriaxone for Injection USP (sefixi 1000-kosovo) (as Per Invoice) | $44.5K | 2.3% |
| 10 | Pharmaceutical Goods/harmless Medicinesfor Human Use-hyoscine Butylbromide Inje… | $41.4K | 2.2% |
| 11 | Pharmaceutical Goods/harmless Medicinesfor Human Use-sevoflurane USP 250ml ( 60… | $39.1K | 2.0% |
| 12 | Pantoprazole for Injection 40mg Pantrazbpl Unit Pack 10 Vials in a Pack | $34.2K | 1.8% |
| 13 | Sodium Chloride Intravenous Infusion Bp(0.9% W/v) 250 ML Nipple Head Plastic Bo… | $33.1K | 1.7% |
| 14 | Tacpan 1.0mg Capsules (each Hard Gelatincapsules Contains: Tacrolimus Equivalen… | $29.4K | 1.5% |
| 15 | Pharmaceuticals Goods/harmless Medicinesfor Human Use-vancomycin Injection 500m… | $29.0K | 1.5% |
India exports 20+ pharmaceutical formulations to Albania with a combined trade value of $1.9M. Key products include Sodium ($164K), Ceftriaxone ($45K), Pantoprazole ($34K), Methylprednisolone ($24K). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 376 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Albania Trade Routes
These are the top pharmaceutical products exported from India to Albania, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Albania buyers, regulatory requirements, and logistics for that specific product corridor. Products include Sodium ($164K), Ceftriaxone ($45K), Pantoprazole ($34K), Methylprednisolone ($24K) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Albania
44 Indian pharmaceutical companies export finished formulations to Albania. Leading exporters include Swiss Parenterals Limited, Otsuka Pharmaceutical India Private Limited, Brawn Laboratories Limited, Deutsche Labs Inc.. The top exporter accounts for 43.9% of total India–Albania pharma exports. Source: Indian Customs (DGFT).
Top Albania Buyers from India
51 companies in Albania import pharmaceutical formulations from India. Top buyers include N. Sh Drenipharm, To The Order Of ., To The Order, To The Order Of.. The largest buyer accounts for 14.1% of India–Albania pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Albania Pharmaceutical Shipments
Indian Export Ports
Albania Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Albania Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and Albania has experienced a steady growth trajectory over the past two decades. In the early 2000s, the trade volume was modest, with India exporting primarily generic medicines to Albania. By 2022, this bilateral trade had expanded significantly, with India exporting finished pharmaceutical formulations valued at $1.9 million USD across 376 shipments. This growth reflects India's increasing capacity to meet Albania's pharmaceutical needs and Albania's growing demand for affordable, high-quality medicines.
Key milestones in this trade relationship include the establishment of direct shipping routes and the participation of Indian pharmaceutical companies in Albanian trade fairs, which have facilitated business partnerships. The average shipment value of $5,000 USD indicates a diversified product range, catering to various therapeutic areas. The presence of 44 Indian exporters and 51 Albanian buyers underscores the robust and expanding nature of this trade corridor.
2India's Market Position
India's pharmaceutical exports to Albania, totaling $1.9 million USD, represent a small fraction of Albania's total pharmaceutical imports, which amounted to $302.39 million USD in 2024. However, India's share is notable when compared to other exporting countries. For instance, Albania imported $43.8 million USD worth of pharmaceutical products from Italy and $60.77 million USD from Germany in 2024. This positions India as a growing player in Albania's pharmaceutical market, with potential for increased market penetration.
The strategic importance of this corridor lies in India's competitive advantage in producing high-quality generic medicines at cost-effective prices, aligning with Albania's healthcare needs and budget constraints. The existing trade volume and the presence of multiple Indian exporters and Albanian buyers suggest a foundation for deeper economic cooperation and market expansion.
3Recent Developments
Between 2024 and 2026, several developments have influenced the India-Albania pharmaceutical trade corridor. In March 2024, India signed the India–EFTA Trade and Economic Partnership Agreement, which is set to enter into force on 1 October 2025. While Albania is not a member of the European Free Trade Association (EFTA), this agreement may indirectly affect trade dynamics in the region, potentially opening avenues for Indian pharmaceutical exports to neighboring markets.
Additionally, in April 2021, India extended humanitarian assistance to Albania by providing 50,000 doses of the COVID-19 vaccine (Covishield), strengthening bilateral relations and fostering goodwill. Such diplomatic gestures may enhance trust and pave the way for increased trade collaborations in the pharmaceutical sector.
These developments indicate a positive trajectory for the India-Albania pharmaceutical trade corridor, with potential for growth and deeper economic integration in the coming years.
Albania Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
To market pharmaceutical products in Albania, Indian companies must obtain marketing authorization from the National Agency of Medicines and Medical Devices (AKBPM). This process involves submitting a comprehensive dossier that includes detailed information on the product's composition, manufacturing process, and clinical data. The dossier must adhere to the Common Technical Document (CTD) format, which is internationally recognized and facilitates the evaluation process. The CTD structure comprises modules covering administrative information, quality data, non-clinical and clinical study reports, and labeling information. While the ACTD (Asian Common Technical Document) and eCTD (electronic Common Technical Document) formats are also used in some regions, the CTD format is the standard for submissions to AKBPM. The approval timeline can vary, but it typically ranges from several months to over a year, depending on the complexity of the product and the completeness of the submitted dossier. Fees for registration are determined by AKBPM and are subject to change; therefore, it is advisable to consult the latest fee schedule provided by the agency.
2GMP & Facility Requirements
Indian manufacturing facilities exporting to Albania must comply with Good Manufacturing Practice (GMP) standards as stipulated by the Albanian Ministry of Health and Social Protection. These standards are designed to ensure that pharmaceutical products are consistently produced and controlled to quality standards appropriate to their intended use. The AKBPM conducts inspections to verify compliance with these GMP requirements. Manufacturers are required to submit a Plant Master File (PMF) and may undergo on-site inspections as part of the evaluation process. The AKBPM assesses the manufacturing processes, quality control measures, and overall facility conditions to ensure they meet the necessary standards. Manufacturers should be prepared for periodic GMP reviews and inspections, which are conducted to maintain the validity of their GMP approval letters. It is essential for manufacturers to stay updated on any changes to GMP regulations and to maintain continuous compliance to ensure uninterrupted market access in Albania.
3Import Documentation
For the importation of pharmaceutical products into Albania, Indian companies must provide specific documentation to comply with Albanian regulations. This includes a Certificate of Pharmaceutical Product (CPP), which attests to the product's compliance with the standards of the country of origin. Additionally, a Certificate of Analysis (CoA) is required to confirm the product's quality and safety. The AKBPM may also request evidence of GMP compliance from the manufacturing facility. Customs procedures for pharmaceutical imports into Albania involve submitting these documents to the General Directorate of Customs, along with a detailed invoice and packing list. The customs authorities will verify the documentation and may conduct inspections to ensure compliance with all applicable laws and regulations. It is crucial for importers to ensure that all documentation is accurate, complete, and up-to-date to facilitate a smooth import process.
Product Categories & Therapeutic Trends — India to Albania
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Albania are predominantly in the form of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. The top ten products exported encompass a range of therapeutic areas, with a notable emphasis on intravenous solutions and antibiotics. For instance, sodium chloride intravenous infusions (500ML and 100ML) constitute 12.2% of the total export value, while antibiotics like imipenem and cilastatin for injection account for 4.2%. This distribution indicates a significant demand in Albania for intravenous therapies and antibiotics, aligning with the country's healthcare needs.
The prominence of intravenous solutions suggests a substantial requirement for rehydration and electrolyte balance treatments, which are critical in managing various medical conditions. The inclusion of antibiotics like imipenem and cilastatin highlights the necessity for effective antimicrobial agents to combat infections. These therapeutic categories are essential in addressing acute and chronic health issues prevalent in Albania, thereby driving the demand for such pharmaceutical products.
2Emerging Opportunities
Albania's pharmaceutical market is witnessing a growing interest in biosimilars, driven by the need for cost-effective alternatives to expensive biologic therapies. The country's healthcare system is increasingly open to incorporating biosimilars, presenting opportunities for Indian exporters to introduce these products. Additionally, the rising prevalence of non-communicable diseases (NCDs) in Albania is creating a demand for specialized treatments, opening avenues for Indian pharmaceutical companies to supply innovative therapies.
The global trend of patent expirations, known as the "patent cliff," is also influencing the Albanian market. As patents for several high-cost medications expire, there is a growing opportunity for Indian generic drug manufacturers to enter the Albanian market, offering affordable alternatives to branded drugs. This shift not only benefits the Albanian healthcare system by reducing costs but also provides Indian exporters with a competitive edge in supplying generics.
3Demand Drivers
Albania is experiencing a demographic shift with an aging population, leading to an increased prevalence of age-related health conditions. This demographic change is contributing to a higher demand for pharmaceutical products, particularly those addressing chronic diseases and geriatric care. The country's healthcare spending has been on an upward trajectory, reflecting a commitment to improving health services and infrastructure. In 2018, per capita healthcare spending in Albania was US$275, marking a 20% increase from 2017.
The rising disease burden, especially concerning non-communicable diseases (NCDs) such as cardiovascular diseases, is further driving the demand for pharmaceutical imports. The treatment of cardiovascular diseases in Albania is projected to generate revenues of US$64.63 million in 2025, with an expected annual growth rate of 5.59% from 2025 to 2030. This trend underscores the expanding market for pharmaceutical products that address chronic health conditions, presenting significant opportunities for Indian exporters to meet Albania's evolving healthcare needs.
Trade Policy & Tariff Intelligence — India and Albania
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Albania's Most-Favored-Nation (MFN) tariff rates for pharmaceutical products under Harmonized System (HS) codes 3003 and 3004 are set at 0% for imports from all countries, including India. This zero-duty rate applies to medicaments consisting of two or more constituents mixed together for therapeutic or prophylactic uses, not in measured doses or put up for retail sale (HS 3003), as well as to medicaments consisting of mixed or unmixed products for therapeutic or prophylactic uses, put up in measured doses or in forms or packings for retail sale (HS 3004). (dogana.gov.al)
Albania has established preferential duty rates under various trade agreements. Notably, the Free Trade Agreement (FTA) between Albania and the European Free Trade Association (EFTA) member states—Liechtenstein, Switzerland, Norway, and Iceland—entered into force on 1 November 2010. This agreement abolished customs duties on industrial products, including pharmaceuticals, originating from the EFTA states. However, as India is not a member of EFTA, these preferential rates do not apply to Indian pharmaceutical exports to Albania. (efta.int)
Regarding anti-dumping or countervailing duty measures, there is no publicly available information indicating that Albania has imposed such measures on Indian pharmaceutical products. Albania's trade policies are generally aligned with its commitments under the World Trade Organization (WTO), which discourages the use of anti-dumping and countervailing duties except in specific circumstances.
2Trade Agreements
Albania is a member of the Central European Free Trade Agreement (CEFTA), which includes countries such as Albania, Bosnia and Herzegovina, Kosovo, Moldova, Montenegro, North Macedonia, and Serbia. However, CEFTA does not include India, and therefore, Indian pharmaceutical exports to Albania do not benefit from any preferential treatment under this agreement. (dogana.gov.al)
As of March 2026, there are no publicly available reports indicating that Albania is engaged in negotiations for a Free Trade Agreement (FTA) with India. The absence of such an agreement means that Indian pharmaceutical exports to Albania are subject to Albania's MFN tariff rates, without any preferential treatment.
3IP & Patent Landscape
Albania's intellectual property (IP) framework, including patent laws, is harmonized with European Union standards, as Albania is a candidate country for EU membership. This alignment provides robust protection for pharmaceutical patents, including those held by Indian companies. The patent term in Albania is 20 years from the filing date, with the possibility of a supplementary protection certificate extending protection by up to five years for certain pharmaceutical products. Additionally, Albania's IP laws include provisions for data exclusivity, granting a period during which generic manufacturers cannot rely on the originator's clinical trial data to obtain marketing authorization. These measures can affect the entry of Indian generic drugs into the Albanian market, potentially delaying market access until the expiration of the data exclusivity period.
Market Opportunity Assessment — Albania for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Albania's pharmaceutical market is experiencing steady growth, with projections indicating a compound annual growth rate (CAGR) of 6.2% from 2026 to 2032. This growth is driven by factors such as the rising prevalence of chronic diseases, increased healthcare expenditures, and alignment with European Union regulatory standards. The market is characterized by a significant reliance on imports, as domestic manufacturing capabilities are limited. Generic drugs dominate the market, accounting for the majority of sales, although branded medications lead in terms of value.
In 2022, India's pharmaceutical exports to Albania were valued at $1.9 million, comprising 376 shipments from 44 Indian exporters to 51 Albanian buyers. The average shipment value was approximately $5,000. The top three products exported were:
- 1Pharmaceutical goods/harmless medicines: $186,000 (9.7%)
- 2Sodium chloride intravenous infusion BP (0.9% w/v) 500ml: $164,000 (8.6%)
- 3Furosemide BP 10mg water for injection: $83,000 (4.4%)
These figures suggest a growing demand for essential medicines and injectables in Albania, presenting opportunities for Indian pharmaceutical exporters to expand their presence in the Albanian market.
2Healthcare System
Albania has integrated Universal Health Coverage (UHC) into its National Health Strategy 2016–2021, aiming to achieve the Sustainable Development Goals. The state provides the majority of health services, including promotion, prevention, diagnosis, and treatment. Healthcare spending accounts for 2.6% of the GDP, partially financed by compulsory health insurance. (acpd.org.al) The Ministry of Health and Social Protection oversees the healthcare system, ensuring access to medical, pharmaceutical, and hospital services.
3Opportunity for Indian Exporters
The Albanian pharmaceutical market presents significant opportunities for Indian exporters, particularly in the segments of essential medicines and injectables. The projected market growth and the dominance of generic drugs indicate a favorable environment for cost-effective pharmaceutical products. Indian exporters can leverage their strengths in producing high-quality generics to meet the increasing demand in Albania. The existing trade data highlights a solid foundation for expanding product offerings and increasing market share in the Albanian market.
FAQ — India to Albania Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Albania?
India exported pharmaceuticals worth $1.9M to Albania across 376 verified shipments.
Who are the top Indian pharmaceutical exporters to Albania?
1. SWISS PARENTERALS LIMITED — $842.3K. 2. OTSUKA PHARMACEUTICAL INDIA PRIVATE LIMITED — $269.9K. 3. BRAWN LABORATORIES LIMITED — $211.9K. Total: 44 suppliers.
Which companies in Albania import pharmaceuticals from India?
1. N. SH DRENIPHARM — $269.9K. 2. TO THE ORDER OF . — $199.7K. 3. TO THE ORDER — $118.7K. 51 buyers total.
What pharmaceutical products does India export most to Albania?
1. Pharmaceutical Goods/harmless Medicines ($185.5K, 9.7%); 2. Sodium Chloride Intravenous Infusion Bp(0.9% W/v) 500ml Nipple Head Plastic Bot… ($164.3K, 8.6%); 3. Each ML Contains Furosemide BP 10mg Water for Injection BP Q S ($83.4K, 4.4%); 4. Pharmaceutical Goods/harmless Medicinesfor Human Use-imipenem and Cilastatin fo… ($80.4K, 4.2%); 5. Sodium Chloride Intravenous Infusion Bp(0.9% W/v) 100ml Nipple Head Plastic Bot… ($68.2K, 3.6%)
Which ports handle pharmaceutical shipments from India to Albania?
Export: AHEMDABAD ICD, THAR DRY PORT ICD/AHMEDABAD GUJARAT ICD (INSAU6), KHATUWAS ICD (INCML6), Tughlakabad, AHEMDABAD ICD (INSBI6). Import: DURRES, TIRANA, PRISTINA.
Why does Albania import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $1.9M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Albania?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Albania pharmaceutical trade?
$5.1K per consignment across 376 shipments.
How many Indian pharmaceutical companies export to Albania?
44 Indian companies. Largest: SWISS PARENTERALS LIMITED with $842.3K.
How can I find verified Indian pharmaceutical suppliers for Albania?
TransData Nexus covers 44 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Albania Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 376 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Albania identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 376 verified shipments from 44 Indian exporters to 51 Albania buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
44 Exporters
51 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists