India to Afghanistan Pharmaceutical Export
Bilateral Trade Intelligence · $233.2M Total Trade · 363 Exporters · 483 Buyers · DGFT Verified · Updated March 2026
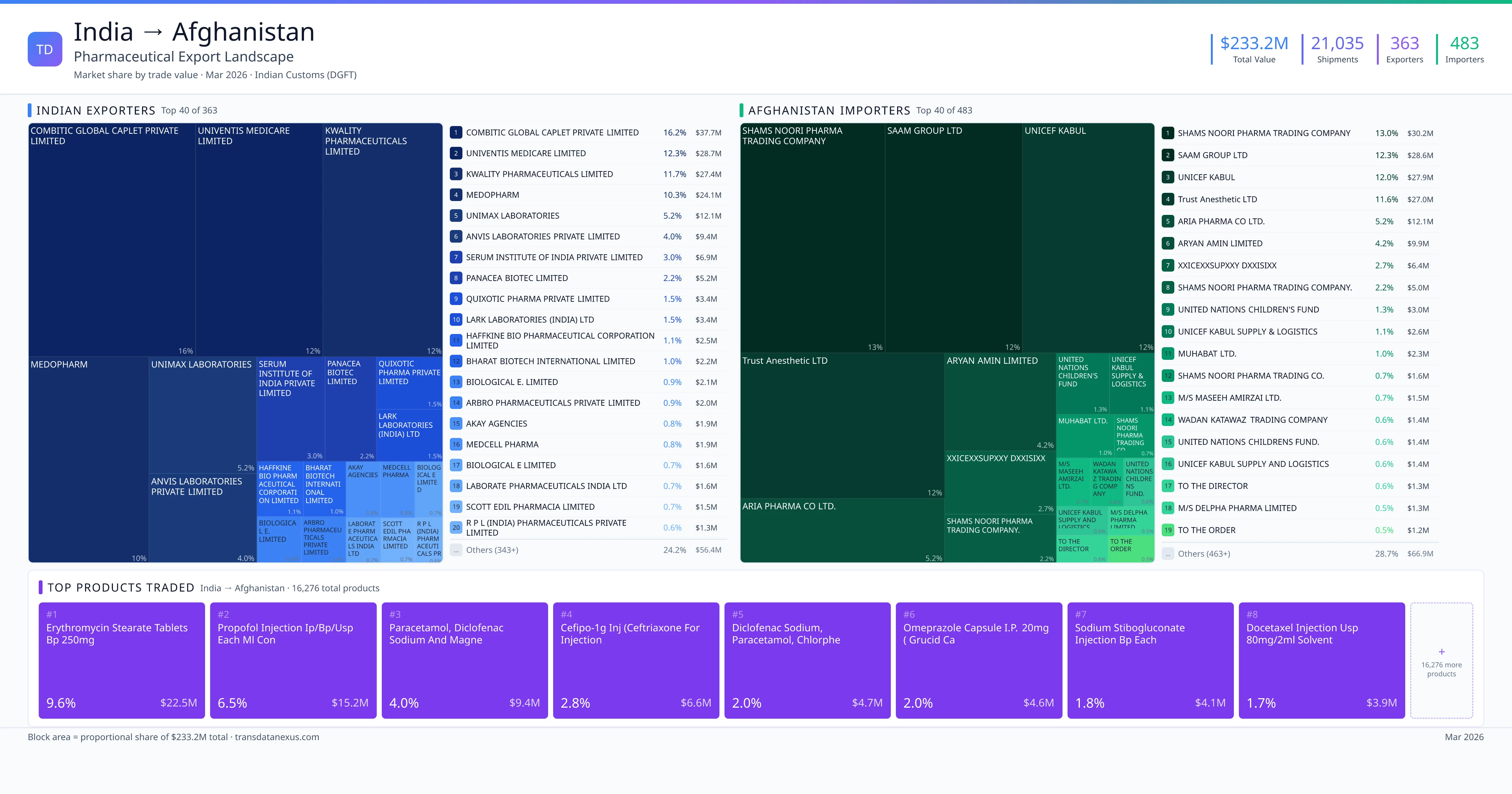
India exported $233.2M worth of pharmaceutical formulations to Afghanistan across 21,035 verified shipments, sourced from 363 Indian exporters supplying 483 Afghanistan buyers. The top exporters are COMBITIC GLOBAL CAPLET PRIVATE LIMITED ($37.7M) and UNIVENTIS MEDICARE LIMITED ($28.7M). The leading products are Erythromycin ($22.5M) and Paracetamol ($9.4M). Average shipment value: $11.1K.
Top Pharmaceutical Formulations — India to Afghanistan
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Erythromycin Stearate Tablets BP 250mg | $22.5M | 9.6% |
| 2 | Propofol Injection Ip/bp/usp Each ML Con | $15.2M | 6.5% |
| 3 | Paracetamol, Diclofenac Sodium and Magne | $9.4M | 4.0% |
| 4 | Cefipo-1g Injection (ceftriaxone for Injection | $6.6M | 2.8% |
| 5 | Diclofenac Sodium, Paracetamol, Chlorphe | $4.7M | 2.0% |
| 6 | Omeprazole Capsule I.p. 20mg ( Grucid Ca | $4.6M | 2.0% |
| 7 | Sodium Stibogluconate Injection BP Each | $4.1M | 1.8% |
| 8 | Docetaxel Injection USP 80mg/2ml Solvent | $3.9M | 1.7% |
| 9 | Vaccine for Polio-poliomyelitis | $3.4M | 1.4% |
| 10 | Cefone Injection 1000mg (ceftriaxone for | $3.3M | 1.4% |
| 11 | Spectinomycin for Injectable Suspension | $3.1M | 1.3% |
| 12 | Alphacold Capsules (as Per Permission) | $3.1M | 1.3% |
| 13 | Bactofix Ds Dry Syrup (cefixime for Oral | $3.0M | 1.3% |
| 14 | Oral Polio Vaccine (bi-valent),item Code | $2.5M | 1.1% |
| 15 | Bivalent (type 1 & Type3) Oral Poliomyelitis Vaccine (bopv) 20 Dose Vial Live O… | $2.4M | 1.0% |
India exports 20+ pharmaceutical formulations to Afghanistan with a combined trade value of $233.2M. Key products include Erythromycin ($22.5M), Paracetamol ($9.4M), Diclofenac ($4.7M), Omeprazole ($4.6M), Sodium ($4.1M). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 21,035 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — India to Afghanistan Trade Routes
These are the top pharmaceutical products exported from India to Afghanistan, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Afghanistan buyers, regulatory requirements, and logistics for that specific product corridor. Products include Erythromycin ($22.5M), Paracetamol ($9.4M), Diclofenac ($4.7M), Omeprazole ($4.6M), Sodium ($4.1M) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Indian Exporters to Afghanistan
363 Indian pharmaceutical companies export finished formulations to Afghanistan. Leading exporters include Combitic Global Caplet Private Limited, Univentis Medicare Limited, Kwality Pharmaceuticals Limited, Medopharm. The top exporter accounts for 16.2% of total India–Afghanistan pharma exports. Source: Indian Customs (DGFT).
Top Afghanistan Buyers from India
483 companies in Afghanistan import pharmaceutical formulations from India. Top buyers include Shams Noori Pharma Trading Company, Saam Group Ltd, Unicef Kabul, Trust Anesthetic Ltd. The largest buyer accounts for 13.0% of India–Afghanistan pharma imports. Source: Indian Customs (DGFT).
Port Analysis — India to Afghanistan Pharmaceutical Shipments
Indian Export Ports
Afghanistan Import Ports
Trade Statistics
Other Destinations
Product Routes
India–Afghanistan Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade between India and Afghanistan has experienced significant growth and transformation over the past few decades. Historically, Afghanistan's pharmaceutical market was predominantly supplied by neighboring Pakistan. However, geopolitical tensions and border closures, particularly after October 2025, disrupted these traditional supply chains, compelling Afghanistan to seek alternative sources for its pharmaceutical needs.
In response to these challenges, India emerged as a pivotal supplier, leveraging its robust pharmaceutical industry to meet Afghanistan's growing demand for quality medicines. This shift was further solidified by the signing of a $100 million Memorandum of Understanding (MoU) in November 2025 between India's Zydus Lifesciences and Afghanistan's Rofi’s International Group of Companies. The agreement outlined plans for initial exports of medicines to Afghanistan, with intentions to establish a representative office and eventually commence local drug production, thereby enhancing the bilateral trade relationship. (aa.com.tr)
The growth trajectory of this trade corridor is evident in the substantial increase in export values. In 2024, India's pharmaceutical exports to Afghanistan were valued at $100.27 million, marking a significant rise from previous years. This upward trend underscores the strengthening economic ties and the strategic importance of the pharmaceutical sector in bilateral relations.
2India's Market Position
India has solidified its position as a dominant supplier in Afghanistan's pharmaceutical market. The disruption of traditional supply routes, particularly with Pakistan, has led to a substantial shift in Afghanistan's pharmaceutical imports. While specific data on Afghanistan's total pharmaceutical imports is limited, the increased reliance on Indian exports indicates a significant market share. The strategic importance of this corridor is further highlighted by India's commitment to establishing manufacturing facilities within Afghanistan, aiming to meet local demand more effectively and foster economic collaboration.
3Recent Developments
Between 2024 and 2026, several key developments have shaped the India-Afghanistan pharmaceutical trade corridor. In November 2025, Afghan Minister of Industry and Commerce, Alhaj Nooruddin Azizi, visited India to discuss expanding pharmaceutical trade and investment opportunities. This visit led to the establishment of an air freight corridor between Kabul and Delhi, facilitating faster and more efficient trade. Additionally, in December 2025, Afghan Health Minister Noor Jalal Jalali emphasized the critical role of India's pharmaceutical sector in addressing Afghanistan's healthcare needs, highlighting the long-standing trust Afghan consumers place in Indian products. These developments reflect a concerted effort to strengthen bilateral ties and ensure a steady supply of quality medicines to Afghanistan.
Afghanistan Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1Registration Process
To export finished pharmaceutical formulations to Afghanistan, Indian pharmaceutical companies must navigate a structured registration process overseen by the Afghanistan Food and Drug Authority (AFDA). This process ensures that imported medicines meet the country's standards for quality, safety, and efficacy.
Application Submission: The registration process begins with the submission of a completed application form for each product, accompanied by a cover letter on the applicant company's official letterhead. If the applicant is an Afghan importing company, a letter of authorization from the manufacturing company is required. This letter should be on the manufacturer's original letterhead, dated, and signed by the president or deputy of the company. It must be sent via the official email address of the manufacturing company.
Dossier Requirements: The application must include a comprehensive dossier containing:
- Certificates: A Certificate of Pharmaceutical Product (CoPP) endorsed by the Ministry of Health, Ministry of Commerce, and Ministry of Foreign Affairs in the country of origin, and authenticated by the Afghanistan Embassy.
- Manufacturing and Product Information: Copies of the manufacturing license, product manufacturing license, and product registration certificate from at least one other country.
- Quality Control Documents: Certificates of Analysis (CoA) for at least two batches of the Active Pharmaceutical Ingredient (API) and three batches of the finished product, along with stability data.
Dossier Format: The dossier should adhere to the Common Technical Document (CTD) format, which is internationally recognized and facilitates the evaluation process. This format includes sections on administrative data, quality, non-clinical, and clinical information.
Approval Timeline and Fees: The approval timeline can vary based on the completeness of the application and the workload of the AFDA. While specific timelines and fees are not explicitly detailed in the available sources, applicants should anticipate a thorough evaluation process. It is advisable to consult directly with the AFDA or a local regulatory affairs consultant for the most accurate and current information.
2GMP & Facility Requirements
Afghanistan requires that pharmaceutical products imported into the country comply with Good Manufacturing Practice (GMP) standards to ensure product quality and safety. Manufacturing facilities in India must hold a valid GMP certificate issued by the national regulatory authority, such as the Central Drugs Standard Control Organization (CDSCO). This certificate should be included in the registration dossier.
The inspection process involves the AFDA evaluating the manufacturing facility's compliance with GMP standards. While specific details on the inspection process are limited, adherence to WHO-GMP guidelines is essential. Mutual recognition agreements between Afghanistan and India may streamline this process, but applicants should verify the current status of such agreements with the AFDA.
3Import Documentation
To import pharmaceutical products into Afghanistan, companies must obtain an import license from the General Directorate of Pharmacy Affairs (GDPA) under the Ministry of Public Health. This license is mandatory for all entities wishing to import medicines and medical equipment into the country.
Required documentation includes:
- Import License: Issued by the GDPA, authorizing the importation of pharmaceutical products.
- Certificates: A Certificate of Pharmaceutical Product (CoPP) and a Good Manufacturing Practice (GMP) certificate from the country of origin.
- Product Information: Detailed product labeling, including trade names, active ingredients, dosage forms, therapeutic classifications, and storage conditions.
Customs procedures for pharmaceutical imports into Afghanistan require compliance with the GDPA's regulations. Importers must ensure that all documentation is accurate and complete to facilitate smooth customs clearance. Given the evolving regulatory landscape, it is recommended to consult directly with the GDPA or a local regulatory affairs consultant for the most current information.
Product Categories & Therapeutic Trends — India to Afghanistan
Dominant categories, emerging opportunities, and demand drivers
1Dominant Categories
India's pharmaceutical exports to Afghanistan are predominantly concentrated in the therapeutic areas of antibiotics, analgesics, anti-inflammatory agents, and gastrointestinal medications. This distribution aligns with Afghanistan's pressing healthcare needs, characterized by a high prevalence of infectious diseases, pain management requirements, and gastrointestinal disorders.
The leading export, Erythromycin Stearate Tablets BP 250mg, valued at $22.5 million (9.6% of total exports), underscores the demand for antibiotics to combat bacterial infections. Similarly, Propofol Injection IP/BP/USP Each ML Con ($15.2 million, 6.5%) and Paracetamol, Diclofenac Sodium, and Magnesium ($9.4 million, 4.0%) highlight the necessity for analgesics and anti-inflammatory agents in managing pain and inflammation. The inclusion of Omeprazole Capsule I.P. 20mg (Grucid CA) ($4.6 million, 2.0%) reflects the need for gastrointestinal treatments, addressing issues such as acid reflux and ulcers.
These therapeutic categories dominate due to Afghanistan's high burden of infectious diseases, musculoskeletal disorders, and gastrointestinal ailments. The country's fragile healthcare infrastructure and limited access to specialized care further amplify the reliance on these essential medications.
2Emerging Opportunities
Afghanistan's healthcare system faces significant challenges, including a high burden of non-communicable diseases (NCDs) such as cardiovascular diseases, diabetes, and mental health conditions. These conditions are expected to account for over 60% of deaths by 2030, predominantly affecting women. (emro.who.int) This shift presents emerging opportunities for Indian pharmaceutical exporters to introduce generics and biosimilars targeting NCDs, offering cost-effective alternatives to branded therapies.
Additionally, the global trend of patent expirations, known as the "patent cliff," is creating openings for generic versions of previously patented drugs. Indian manufacturers, with their established expertise in generics, are well-positioned to meet this demand in Afghanistan, providing affordable access to essential medications.
3Demand Drivers
Afghanistan's disease burden is a significant driver of pharmaceutical import demand. The country faces a high prevalence of infectious diseases, including tuberculosis, malaria, and HIV, alongside a rising incidence of NCDs such as cardiovascular diseases and diabetes. The Global Burden of Disease 2016 study indicates that ischemic heart disease and road injuries are leading causes of disability-adjusted life years (DALYs) among males, while lower respiratory infections and ischemic heart disease are leading causes among females.
The healthcare system's underfunding exacerbates these challenges, with critical shortages in infrastructure, medical supplies, and healthcare professionals. In 2022, healthcare spending per capita was $80.65, a 1.07% decline from 2021, reflecting limited financial resources allocated to health services. This financial constraint, coupled with a reliance on out-of-pocket expenditures, underscores the need for affordable pharmaceutical imports to meet the population's health needs.
In summary, India's pharmaceutical exports to Afghanistan are driven by the country's substantial disease burden and the critical need for accessible, cost-effective medications. The dominance of antibiotics, analgesics, anti-inflammatory agents, and gastrointestinal medications aligns with Afghanistan's healthcare priorities, while emerging opportunities in generics and biosimilars present avenues for further collaboration.
Trade Policy & Tariff Intelligence — India and Afghanistan
Tariff structure, trade agreements, IP and patent landscape
1Tariff & Duty Structure
Afghanistan's tariff structure for pharmaceutical products, particularly those classified under HS codes 3003 and 3004, is not publicly detailed. The Harmonized System (HS) codes 3003 and 3004 pertain to medicaments consisting of two or more constituents mixed together for therapeutic or prophylactic uses, not in measured doses or put up for retail sale, and those put up in measured doses or in forms or packings for retail sale, respectively. Specific Most-Favored-Nation (MFN) tariff rates for these products in Afghanistan are not readily available. Additionally, preferential duty rates under any trade agreements, as well as exemptions or reductions in duties for pharmaceutical formulations entering Afghanistan, are not specified in the available sources.
2Trade Agreements
As of March 2026, there is no publicly available information indicating the existence of a Free Trade Agreement (FTA) between India and Afghanistan. The available data does not specify any bilateral trade agreements between the two countries that would impact pharmaceutical trade. Furthermore, there are no details on current or upcoming negotiations aimed at establishing such agreements.
3IP & Patent Landscape
The available information does not provide specific details regarding intellectual property (IP) or patent provisions in Afghanistan that affect the importation of generic drugs from India. There is no mention of data exclusivity periods, compulsory licensing provisions, or other IP-related measures that could influence the entry of Indian generic pharmaceuticals into the Afghan market.
Supply Chain & Logistics — India to Afghanistan Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from India to Afghanistan primarily utilize both sea and air transport, each offering distinct advantages. Sea freight is often employed for bulk shipments due to its cost-effectiveness, though it entails longer transit times. Air freight, conversely, is preferred for high-value or time-sensitive goods, providing faster delivery but at a higher cost.
The journey from Indian ports to Afghanistan typically involves transshipment through neighboring countries, such as Pakistan or Iran, before reaching Afghan borders. For instance, shipments from Ludhiana ICD, a key export port in India, may transit through Karachi, a significant import port in Afghanistan, before entering Afghanistan. The exact transit time varies based on the chosen route and mode of transport. Air freight can deliver goods within 1–3 days, while sea freight may take 20–40 days, including transshipment and border crossings. (sascompany.ir)
Recent disruptions in the Red Sea have impacted global shipping routes, potentially affecting transit times and costs for shipments from India to Afghanistan. While specific data on the extent of these disruptions is limited, it's advisable to consult with logistics providers for the most current information and to plan accordingly.
2Port Infrastructure
In India, the primary export ports for pharmaceutical shipments include Ludhiana ICD, GRFL Sahnewal Ludhiana ICD, and Barhi ICD. Ludhiana ICD, for example, serves as a significant hub for exports, handling a substantial volume of pharmaceutical goods. In Afghanistan, key import ports include Karachi, Kandahar, and Kabul. Kandahar, in particular, handles a significant portion of imports, including pharmaceuticals. These ports are equipped to handle pharmaceutical shipments, though infrastructure and capacity may vary. It's essential to coordinate with logistics partners to ensure efficient handling and compliance with all regulatory requirements.
3Cold Chain & Compliance
Maintaining the integrity of temperature-sensitive pharmaceutical products is paramount. Cold chain logistics involves strict temperature control throughout the supply chain to ensure product efficacy and safety. This includes using insulated packaging, real-time temperature monitoring, and adhering to Good Distribution Practice (GDP) guidelines. Compliance with international standards, such as those set by the World Health Organization (WHO), is essential to meet regulatory requirements and maintain product quality. Additionally, customs clearance processes in Afghanistan require precise documentation, including certificates of analysis and temperature control declarations, to verify compliance and ensure timely delivery.
Market Opportunity Assessment — Afghanistan for Indian Pharma
Market size, healthcare system, growth outlook
1Market Size & Growth
Afghanistan's pharmaceutical market is predominantly import-dependent, with over 95% of its medicines sourced from other countries. The market has experienced growth, with a compound annual growth rate (CAGR) of 6.3% projected during the 2026-2032 period. This growth is driven by increased demand for essential medicines, humanitarian aid, and the expansion of private pharmacy networks in urban areas.
The generic drug penetration rate in Afghanistan is significant, as generics constitute a substantial portion of the imported pharmaceutical market. The global generics market is projected to grow at a CAGR of 7.2%, with the Asia-Pacific region leading in generics market share. While specific figures for Afghanistan are limited, the reliance on imported generics suggests a high penetration rate.
2Healthcare System
Afghanistan has implemented the Basic Package of Health Services (BPHS) to promote free universal coverage at the primary healthcare level across the country. However, challenges such as persistent insecurity, conservative social norms, and weak public financing have hindered the full realization of Universal Health Coverage (UHC). The country has also developed an Integrated Package of Essential Health Services (IPEHS) to expand coverage and scope, but its implementation has been limited due to resource constraints and political challenges.
Regarding health insurance, there is no comprehensive national health insurance scheme in Afghanistan. The majority of the population relies on out-of-pocket payments for healthcare services, leading to financial barriers in accessing necessary treatments.
3Opportunity for Indian Exporters
The substantial import dependency of Afghanistan's pharmaceutical market presents a significant opportunity for Indian exporters. In 2022, India exported finished pharmaceutical formulations valued at $233.2 million to Afghanistan, with 363 Indian exporters supplying 483 Afghan buyers. The top 10 products exported include antibiotics like Erythromycin Stearate Tablets BP 250mg and Ceftriaxone for Injection, indicating a strong demand for essential medicines.
The projected CAGR of 6.3% for the Afghan pharmaceutical market during 2026-2032 suggests continued growth, driven by increasing demand for affordable healthcare solutions and the expansion of private pharmacy networks. Indian exporters can leverage this growth by focusing on high-demand generic medicines, particularly in therapeutic areas such as infectious diseases, cardiovascular conditions, and respiratory disorders. Additionally, aligning with Afghanistan's healthcare programs, such as the BPHS, can enhance market penetration and contribute to the country's progress toward Universal Health Coverage.
Competitive Landscape — India vs Other Pharmaceutical Suppliers to Afghanistan
Competing origins, India's edge, challenges and threats
1Competing Origins
Afghanistan's pharmaceutical market is supplied by a diverse array of international and domestic sources. India stands out as the predominant exporter, accounting for approximately 86% of Afghanistan's pharmaceutical imports, with 680,036 shipments. China also plays a significant role, meeting about 25% of Afghanistan's medicinal needs, offering products priced approximately 15% lower than those from India or Iran. (english.news.cn) European Union manufacturers, while historically influential, have seen a decline in their market share due to increased competition from Asian suppliers and shifting global trade dynamics. Domestic production in Afghanistan remains limited, with local companies producing a variety of medical products such as tablets, capsules, and powders, but they still meet only a fraction of the national demand.
2India's Competitive Edge
India's pharmaceutical industry offers several competitive advantages in the Afghan market. The country's manufacturers are known for cost competitiveness, providing affordable medicines that are crucial for Afghanistan's price-sensitive market. Many Indian pharmaceutical companies adhere to WHO-GMP standards, ensuring high-quality products that meet international regulatory requirements. Additionally, India's English-speaking workforce facilitates smoother communication and collaboration with Afghan importers and regulatory bodies. The broad portfolio of Indian pharmaceutical products, encompassing a wide range of therapeutic categories, allows Afghan buyers to source diverse medications from a single supplier, streamlining procurement processes.
3Challenges & Threats
Despite its strengths, India's pharmaceutical exports to Afghanistan face several challenges. Regulatory tightening in Afghanistan, including stringent quality control measures and compliance with WHO-GMP standards, can pose obstacles for Indian exporters. Competition from Chinese manufacturers, who offer lower-priced alternatives, intensifies market pressures, potentially eroding India's market share. Additionally, concerns about the quality of imported medicines, including issues related to counterfeit and substandard products, can affect the perception of Indian pharmaceuticals among Afghan consumers and healthcare providers.
FAQ — India to Afghanistan Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Afghanistan?
India exported pharmaceuticals worth $233.2M to Afghanistan across 21,035 verified shipments.
Who are the top Indian pharmaceutical exporters to Afghanistan?
1. COMBITIC GLOBAL CAPLET PRIVATE LIMITED — $37.7M. 2. UNIVENTIS MEDICARE LIMITED — $28.7M. 3. KWALITY PHARMACEUTICALS LIMITED — $27.4M. Total: 363 suppliers.
Which companies in Afghanistan import pharmaceuticals from India?
1. SHAMS NOORI PHARMA TRADING COMPANY — $30.2M. 2. SAAM GROUP LTD — $28.6M. 3. UNICEF KABUL — $27.9M. 483 buyers total.
What pharmaceutical products does India export most to Afghanistan?
1. Erythromycin Stearate Tablets BP 250mg ($22.5M, 9.6%); 2. Propofol Injection Ip/bp/usp Each ML Con ($15.2M, 6.5%); 3. Paracetamol, Diclofenac Sodium and Magne ($9.4M, 4.0%); 4. Cefipo-1g Injection (ceftriaxone for Injection ($6.6M, 2.8%); 5. Diclofenac Sodium, Paracetamol, Chlorphe ($4.7M, 2.0%)
Which ports handle pharmaceutical shipments from India to Afghanistan?
Export: LUDHIANA ICD, GRFL SAHNEWAL LUDHIANA ICD, BARHI ICD, CHENNAI AIR, PALWAL ICD. Import: Karachi, Kandahar, KABUL, KANDAHAR, Kabul.
Why does Afghanistan import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $233.2M corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Afghanistan?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Afghanistan pharmaceutical trade?
$11.1K per consignment across 21,035 shipments.
How many Indian pharmaceutical companies export to Afghanistan?
363 Indian companies. Largest: COMBITIC GLOBAL CAPLET PRIVATE LIMITED with $37.7M.
How can I find verified Indian pharmaceutical suppliers for Afghanistan?
TransData Nexus covers 363 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Afghanistan Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 21,035 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Afghanistan identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 21,035 verified shipments from 363 Indian exporters to 483 Afghanistan buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
363 Exporters
483 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists