Gefitinib HS Code Analysis (2026)
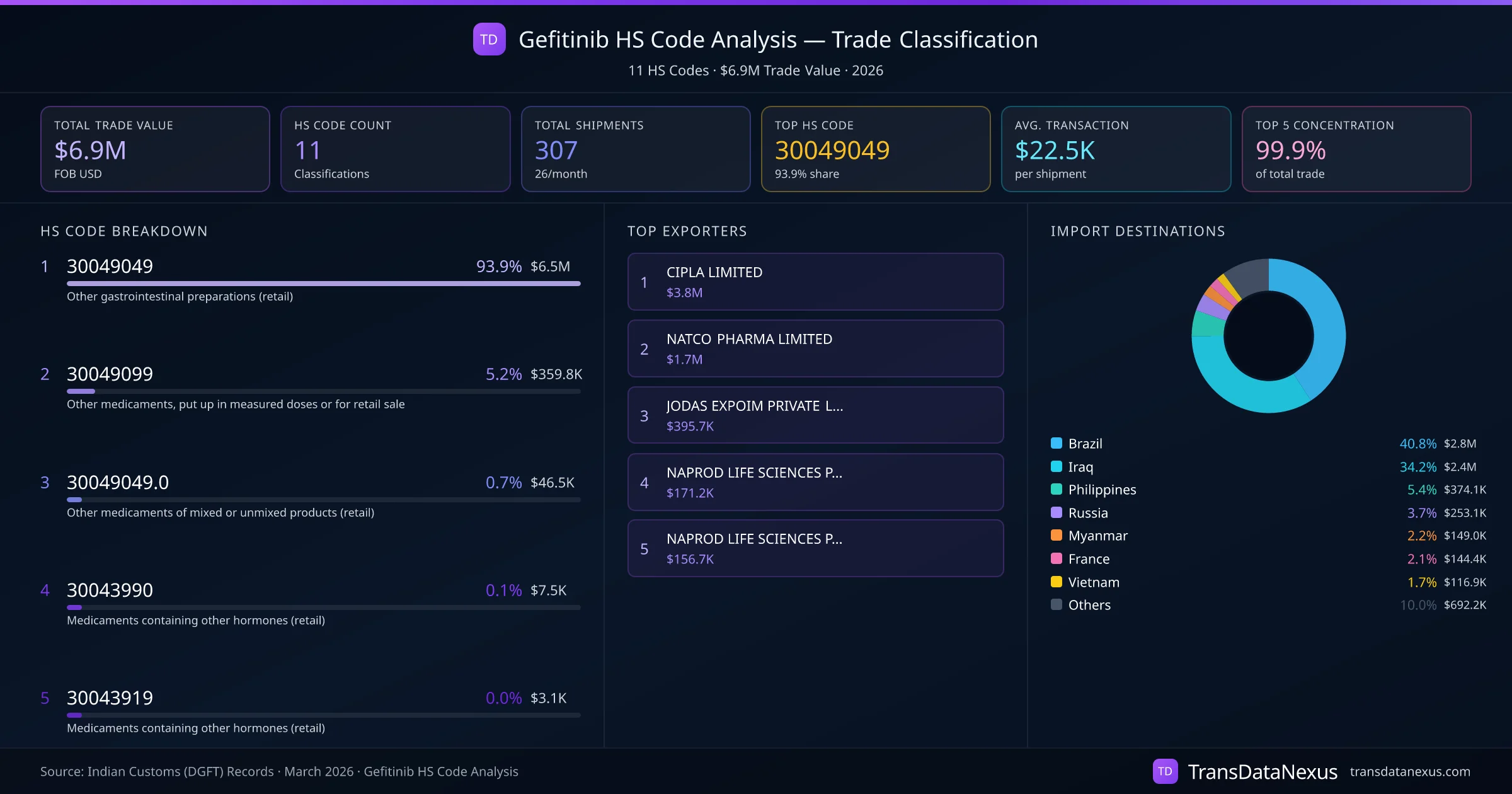
India exports Gefitinib under 11 distinct HS code classifications, representing a total trade value of $6.9M across 307 shipments. The dominant classification is HS 30049049 (Other gastrointestinal preparations (retail)), accounting for 93.9% of all Gefitinib trade at $6.5M. HS 30049099 (Other medicaments, put up in measured doses or for retail sale) follows at 5.2% ($359.8K). The top 5 HS codes together account for 99.9% of total Gefitinib trade from India. The average transaction value is $22517. Key importing markets include Brazil, Iraq, Philippines. Leading exporters include CIPLA LIMITED and NATCO PHARMA LIMITED. Data sourced from Indian Customs (DGFT) shipping bill records.
Top Gefitinib HS Code Classifications
Ranked by trade value from Indian Customs (DGFT) data
All Gefitinib HS Code Classifications
HS 30049049
Other gastrointestinal preparations (retail) · 93.9% of Gefitinib trade
$6.5M
Trade Value
93.9%
Market Share
196
Transactions
$16.20
Avg Unit Price
Common Product Descriptions:
HS 30049099
Other medicaments, put up in measured doses or for retail sale · 5.2% of Gefitinib trade
$359.8K
Trade Value
5.2%
Market Share
94
Transactions
$92.94
Avg Unit Price
Common Product Descriptions:
HS 30049049
Other medicaments of mixed or unmixed products (retail) · 0.7% of Gefitinib trade
$46.5K
Trade Value
0.7%
Market Share
4
Transactions
$13.53
Avg Unit Price
Common Product Descriptions:
HS 30043990
Medicaments containing other hormones (retail) · 0.1% of Gefitinib trade
$7.5K
Trade Value
0.1%
Market Share
1
Transactions
$24.85
Avg Unit Price
Common Product Descriptions:
HS 30043919
Medicaments containing other hormones (retail) · 0.0% of Gefitinib trade
$3.1K
Trade Value
0.0%
Market Share
2
Transactions
$24.49
Avg Unit Price
Common Product Descriptions:
HS 30041090
Other medicaments containing penicillins or streptomycins (retail) · 0.0% of Gefitinib trade
$2.6K
Trade Value
0.0%
Market Share
4
Transactions
$64.25
Avg Unit Price
Common Product Descriptions:
HS 30049099
Other medicaments of mixed or unmixed products (retail) · 0.0% of Gefitinib trade
$925
Trade Value
0.0%
Market Share
1
Transactions
$308.33
Avg Unit Price
Common Product Descriptions:
HS 30049069
Other medicaments of mixed or unmixed products (retail) · 0.0% of Gefitinib trade
$432
Trade Value
0.0%
Market Share
1
Transactions
$43.24
Avg Unit Price
Common Product Descriptions:
HS 30049052
Cardiovascular preparations — anti-anginal (retail) · 0.0% of Gefitinib trade
$377
Trade Value
0.0%
Market Share
2
Transactions
$15.39
Avg Unit Price
Common Product Descriptions:
HS 30021210
Antisera for diphtheria · 0.0% of Gefitinib trade
$87
Trade Value
0.0%
Market Share
1
Transactions
$28.90
Avg Unit Price
Common Product Descriptions:
HS 30049069
Other therapeutic/prophylactic medicaments (retail) · 0.0% of Gefitinib trade
$6
Trade Value
0.0%
Market Share
1
Transactions
$5.97
Avg Unit Price
Common Product Descriptions:
Top Suppliers
Where Gefitinib is Exported — Geographic HS Code Analysis
Import destinations for Gefitinib from India, by trade value
India exports Gefitinib to 10+ countries under 11 HS code classifications. The largest import destination is Brazil ($2.8M), followed by Iraq ($2.4M) and Philippines ($374.1K). These markets collectively account for the majority of India's Gefitinib pharmaceutical exports.
| Rank | Country | Trade Value | Market Share |
|---|---|---|---|
| #1 | Brazil | $2.8M | 40.8% |
| #2 | Iraq | $2.4M | 34.2% |
| #3 | Philippines | $374.1K | 5.4% |
| #4 | Russia | $253.1K | 3.7% |
| #5 | Myanmar | $149.0K | 2.2% |
| #6 | France | $144.4K | 2.1% |
| #7 | Vietnam | $116.9K | 1.7% |
| #8 | Australia | $81.3K | 1.2% |
| #9 | Laos | $56.3K | 0.8% |
| #10 | United States | $54.4K | 0.8% |
Pharmaceutical buyers in these markets import Gefitinib primarily under HS code 30049049 (Other gastrointestinal preparations (retail)). Import duties, regulatory requirements, and product registration vary by destination country. Contact TransData Nexus for country-specific import intelligence.
Understanding Gefitinib HS Codes
The Harmonized System (HS) is an internationally standardized system of names and numbers to classify traded products. Developed by the World Customs Organization (WCO), it is used by over 200 countries for customs tariffs, trade statistics, and regulatory compliance. Each HS code is a 6-digit number (extended to 8 digits in India's Customs Tariff Schedule) that precisely identifies a product category.
How Gefitinib HS Codes Work
Gefitinib falls under Chapter 30 (Pharmaceutical Products) of the Harmonized System. The 11 codes used for Gefitinib exports span multiple headings:
- Heading 3003 — Medicaments not put up in measured doses or for retail sale (bulk APIs, intermediates)
- Heading 3004 — Medicaments put up in measured doses or for retail sale (finished formulations)
- Heading 3005 — Wadding, gauze, bandages for medical use
- Heading 3006 — Pharmaceutical goods (sutures, reagents, dental cements)
The most significant distinction for Gefitinib is between HS 3003 (bulk/unmeasured) and HS 3004 (retail-ready). HS 3004 codes account for the majority of Gefitinib trade (including the dominant code 30049049 at 93.9%). The 8-digit Indian Customs Tariff code further specifies the therapeutic class — for example, HS 30049049 refers to "Other gastrointestinal preparations (retail)".
Regulatory & Compliance — Gefitinib Export Classification
Correct HS code classification is critical for Gefitinib exports from India. Misclassification can result in customs delays, incorrect duty assessment, and regulatory penalties. Key compliance considerations for pharmaceutical stakeholders:
Import Duty Classification
Gefitinib import duties vary significantly by HS code and destination country. HS 3004 (finished formulations) typically attracts different duty rates than HS 3003 (bulk APIs). Many developing countries offer preferential rates for essential medicines including Gefitinib.
DGFT & Indian Customs Requirements
Indian exporters must declare the correct 8-digit ITC-HS code on shipping bills filed with the Directorate General of Foreign Trade (DGFT). Gefitinib exports require Drug Controller General of India (DCGI) approval and must comply with Drug & Cosmetics Act regulations.
WHO Prequalification
For international procurement (UNICEF, Global Fund), Gefitinib products must meet WHO Prequalification standards. WHO PQ status affects which HS codes are eligible for preferential procurement pricing and duty exemptions in recipient countries.
Country-Specific Registration
Each destination country requires product registration before import. Key markets like Brazil and Iraq have specific pharmaceutical regulatory bodies that must approve the product before customs clearance.
Related Advanced Oncology — Products Sharing Similar HS Codes
Other products in the Advanced Oncology therapeutic category that may share HS code classifications with Gefitinib
Products in the Advanced Oncology category (Targeted therapy and advanced cancer treatments) often share HS codes under Chapter 30, particularly heading 3004 (medicaments put up for retail sale). Comparing HS code distributions across related products helps identify classification patterns and trade route similarities.
Tariff Schedule — Top Markets
TransData Nexus Gefitinib HS code trade compliance intelligence
1Tariff Schedule — Top Markets
The Harmonized System (HS) code 30049049 pertains to pharmaceutical products, specifically medications in measured doses. Tariff rates for this code vary across countries and are influenced by Most Favored Nation (MFN) rates, Free Trade Agreements (FTAs) with India, and exemptions for essential medicines.
Brazil: As a member of the World Trade Organization (WTO), Brazil applies MFN tariff rates to imports. While specific MFN rates for HS code 30049049 are not readily available, Brazil's tariff schedule generally imposes duties on pharmaceutical products. India and Brazil are both members of the BRICS group, but no bilateral FTA exists between them, so preferential rates are not applicable. However, Brazil's health regulations may provide exemptions or reduced tariffs for essential medicines, including certain cancer treatments like Gefitinib.
Iraq: Iraq's tariff regime includes MFN rates for WTO members. Specific MFN rates for HS code 30049049 are not specified in the available data. Iraq does not have an FTA with India, so no preferential rates apply. Nonetheless, Iraq's Ministry of Health may offer exemptions or reduced tariffs for essential medicines to ensure affordability and accessibility.
Philippines: As a member of the Association of Southeast Asian Nations (ASEAN), the Philippines benefits from the ASEAN-India Free Trade Area (AIFTA). Under this agreement, tariffs on pharmaceutical products, including those under HS code 30049049, are often reduced or eliminated. Additionally, the Philippines' Food and Drug Administration may classify certain medications as essential, potentially qualifying them for further exemptions or reduced tariffs.
Russia: Russia, a WTO member, applies MFN tariff rates to imports. Specific MFN rates for HS code 30049049 are not detailed in the available sources. There is no FTA between Russia and India, so preferential rates are not applicable. However, Russia's Ministry of Health may provide exemptions or reduced tariffs for essential medicines, including cancer treatments like Gefitinib.
Myanmar: Myanmar, as an ASEAN member, participates in the ASEAN-India Free Trade Area (AIFTA). Under this agreement, tariffs on pharmaceutical products, including those under HS code 30049049, are often reduced or eliminated. Myanmar's Ministry of Health and Sports may also offer exemptions or reduced tariffs for essential medicines to ensure public health needs are met.
France: As a member of the European Union (EU), France applies the EU's Common External Tariff. Pharmaceutical products under HS code 30049049 generally face low or zero MFN tariffs. The EU does not have an FTA with India covering pharmaceuticals, so no preferential rates apply. However, France's health authorities may provide exemptions or reduced tariffs for essential medicines to promote public health.
Vietnam: Vietnam, an ASEAN member, benefits from the ASEAN-India Free Trade Area (AIFTA). Under this agreement, tariffs on pharmaceutical products, including those under HS code 30049049, are often reduced or eliminated. Vietnam's Ministry of Health may also classify certain medications as essential, qualifying them for further exemptions or reduced tariffs.
Australia: Australia and India have a Comprehensive Economic Cooperation Agreement (CECA) under negotiation, but as of now, no FTA is in effect. Therefore, MFN tariff rates apply to imports of pharmaceutical products under HS code 30049049. Australia's Therapeutic Goods Administration may provide exemptions or reduced tariffs for essential medicines to ensure public health needs are met.
Classification Disputes & Misclassification Risks
TransData Nexus Gefitinib HS code trade compliance intelligence
1Classification Disputes & Misclassification Risks
Gefitinib, an anticancer medication, is typically classified under HS code 30049049, which covers "other medicaments consisting of mixed or unmixed products for therapeutic or prophylactic uses, put up in measured doses." However, classification disputes can arise, particularly between HS codes 3003 and 3004.
HS Code 3003 vs. 3004:
- HS Code 3003: This code pertains to "medicaments consisting of two or more constituents mixed together for therapeutic or prophylactic uses, not put up in measured doses or in forms or packings for retail sale."
- HS Code 3004: This code covers "medicaments consisting of mixed or unmixed products for therapeutic or prophylactic uses, put up in measured doses or in forms or packings for retail sale."
The primary distinction lies in the packaging and dosing form. Gefitinib, being supplied in tablet form with specific dosages, falls under HS code 3004.
Common Errors and Penalties:
Misclassification can occur if Gefitinib is incorrectly declared under HS code 3003 instead of 3004. Such errors may lead to:
- Incorrect Duty Assessment: Resulting in underpayment or overpayment of duties.
- Regulatory Non-Compliance: Potentially leading to fines, penalties, or shipment delays.
Recent Rulings:
While specific recent rulings from the World Customs Organization (WCO) or national authorities regarding Gefitinib are not available, it is crucial for importers and exporters to ensure accurate classification to avoid penalties.
Pharmacopoeia & Quality Standards
TransData Nexus Gefitinib HS code trade compliance intelligence
1Pharmacopoeia & Quality Standards
Gefitinib is recognized in various pharmacopoeias, including the United States Pharmacopeia (USP), British Pharmacopoeia (BP), European Pharmacopoeia (EP), and Indian Pharmacopoeia (IP).
Monograph Status:
- USP: Gefitinib Tablets are included in the USP 2025 edition. The monograph specifies that Gefitinib Tablets must contain not less than 90.0% and not more than 110.0% of the labeled amount of gefitinib (C₂₂H₂₄ClFN₄O₃).
- EP: Gefitinib is listed as a reference standard for system suitability in the EP.
Quality Requirements:
- Purity/Assay: The assay ensures the tablet contains the correct amount of active ingredient, typically within 90.0% to 110.0% of the labeled claim.
- Dissolution Testing: Dissolution tests are conducted to ensure the tablet releases the active ingredient appropriately. The USP specifies dissolution testing under general chapter <711>.
- Stability Testing: Stability testing is performed according to the International Council for Harmonisation (ICH) guidelines, considering different climatic zones. This ensures the product maintains its quality over its shelf life under various environmental conditions.
Adherence to these pharmacopoeial standards is essential to ensure the safety, efficacy, and quality of Gefitinib tablets in the market.
Frequently Asked Questions — Gefitinib HS Codes
Common questions about Gefitinib trade classification, answered with data from Indian Customs records.
What is the HS code for Gefitinib exports from India?
The most commonly used HS code for Gefitinib exports from India is 30049049 (Other gastrointestinal preparations (retail)), which accounts for 93.9% of all Gefitinib trade valued at $6.5M. In total, India exports Gefitinib under 11 distinct HS code classifications.
How many HS codes are used for Gefitinib trade?
Gefitinib is exported from India under 11 different HS code classifications, covering a total trade value of $6.9M across 307 shipments. The top 5 HS codes account for 99.9% of total trade.
What is HS code 30049049?
HS code 30049049 refers to "Other gastrointestinal preparations (retail)" under Chapter 30 (Pharmaceutical Products) of the Harmonized System. For Gefitinib, this code covers $6.5M in trade value with 196 transactions, 61 suppliers, and 101 buyers across 66 countries. The average unit price under this code is $16.20.
What is the difference between HS 30049049 and 30049099 for Gefitinib?
HS 30049049 (Other gastrointestinal preparations (retail)) accounts for 93.9% of Gefitinib trade ($6.5M) with an average unit price of $16.20, while HS 30049099 (Other medicaments, put up in measured doses or for retail sale) covers 5.2% ($359.8K) at $92.94 per unit. HS 30049049 involves 61 suppliers and 101 buyers, compared to 52 suppliers and 57 buyers for HS 30049099.
How much Gefitinib is exported under HS code 30049049?
Under HS code 30049049, India exports $6.5M worth of Gefitinib, representing 93.9% of total Gefitinib trade. This involves 196 shipments from 61 Indian exporters to 101 buyers in 66 countries.
Which countries import the most Gefitinib from India?
The top importing countries for Gefitinib from India are Brazil, Iraq, Philippines. The leading importer Brazil accounts for $2.8M in trade value. In total, Gefitinib is exported to buyers across multiple countries under 11 HS code classifications.
Who are the top Gefitinib exporters from India?
The leading Gefitinib exporters from India include CIPLA LIMITED ($3.8M), NATCO PHARMA LIMITED ($1.7M), JODAS EXPOIM PRIVATE LIMITED ($395.7K). These suppliers export across multiple HS code classifications with a combined trade value of $6.9M.
Which Gefitinib HS code has the highest unit price?
HS code 30049099.0 has the highest average unit price at $308.33 per unit for Gefitinib exports. This compares to the overall market average of $22517.31 per transaction. The most traded code HS 30049049 averages $16.20 per unit.
Official References & Regulatory Resources
HS code classifications follow the WCO Harmonized System. Verify tariff rates with your national customs authority.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.HS Code Classification: 11 HS code classifications identified for Gefitinib using Indian Customs Tariff (ITC-HS) mapping.
- 2.Product Categorization: Products under each HS code matched from 5 Indian exporters using DGFT product description fields.
- 3.Trade Value Aggregation: Total trade value aggregated from 307 individual shipping bill records. Values are FOB in USD.
- 4.Country Breakdown: Exports distributed across 10 destination markets with per-country value and shipment analysis.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
307 Verified Shipments
11 HS codes analyzed
Expert-Reviewed
By pharmaceutical trade specialists