Loading...
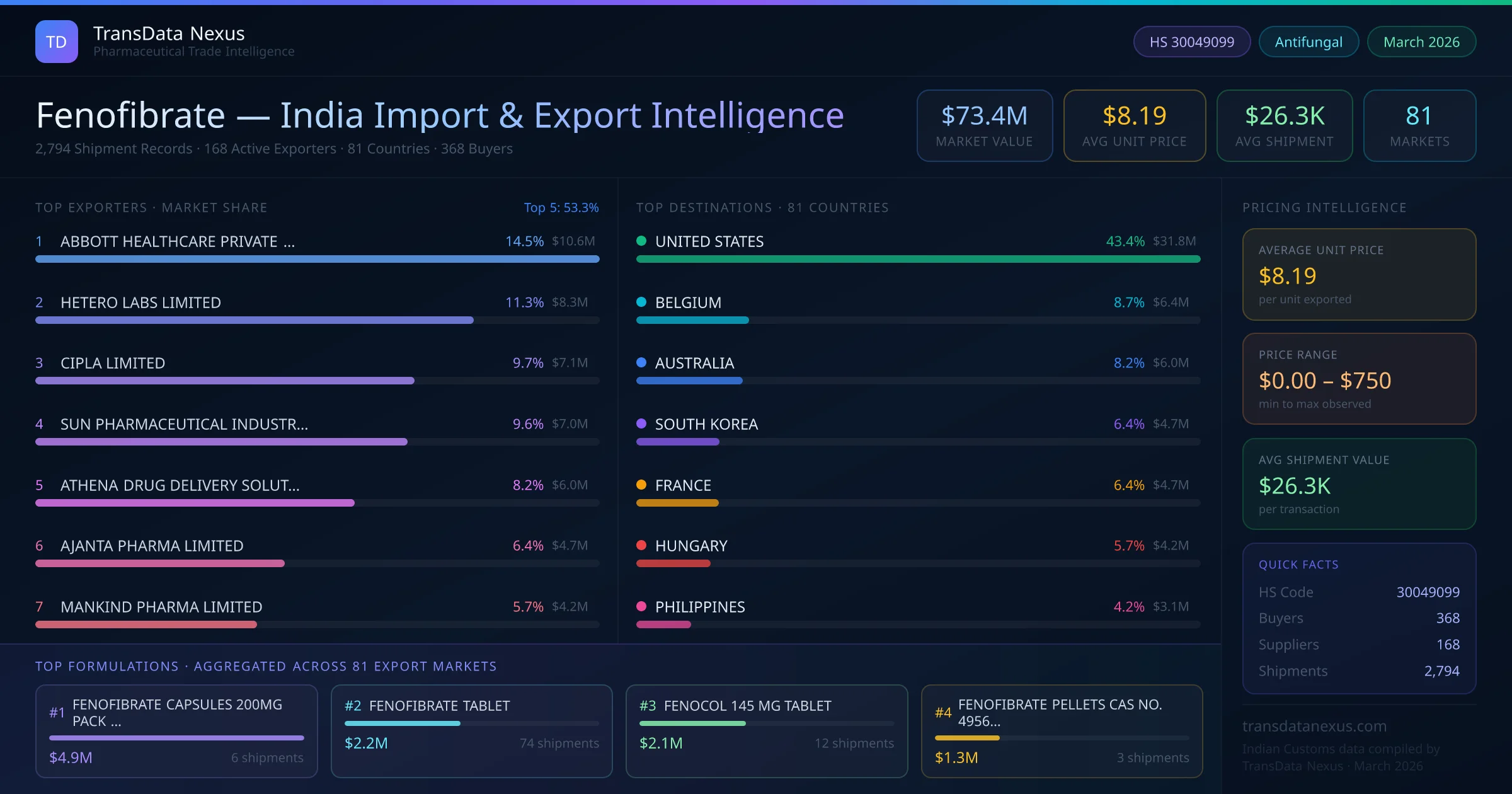
Who Supplies Fenofibrate to India from AUSTRALIA
India's fenofibrate imports from AUSTRALIA total $156 across 3 shipments from 2 foreign suppliers. Alphapharm Pty Ltd leads with $88 in import value; the top 5 suppliers together control 100.0% of this origin. Leading Indian buyers include MYLAN LABORATORIES LIMITED. This corridor reflects India's pharmaceutical import demand for fenofibrate — a concentrated sourcing relationship with select suppliers from AUSTRALIA.