Ethiopia to India Pharmaceutical Import
Bilateral Trade Intelligence · $25 Total Trade · 1 Foreign Suppliers · 1 Indian Buyers · DGFT Verified · Updated March 2026
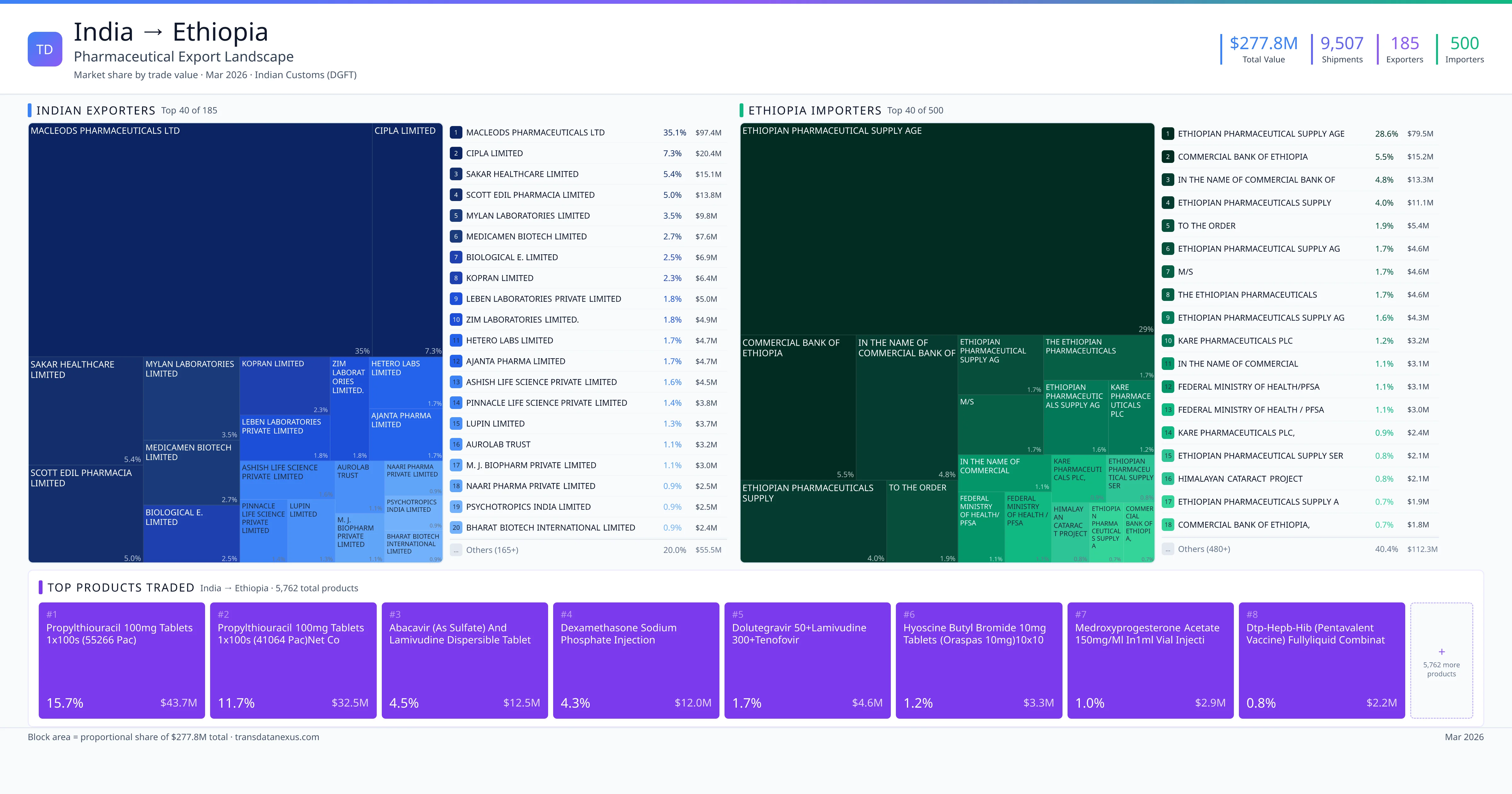
India imported $25 worth of pharmaceutical formulations from Ethiopia across 2 verified shipments, from 1 foreign suppliers to 1 Indian buyers. The top suppliers are ARMAUER HANSEN RESEARCH INSTITUTE ($25). The leading products are Plasma ($25). Average shipment value: $12.
Top Pharmaceutical Formulations — Ethiopia to India
| # | Formulation | Value | Share |
|---|---|---|---|
| 1 | Plasma | $25 | 100.0% |
India imports 1+ pharmaceutical formulations from Ethiopia with a combined trade value of $25. Key products include Plasma ($25). These are finished dosage forms — tablets, capsules, injectables, and combination drugs — shipped from Indian manufacturing facilities with FDA, WHO-GMP, and EU GMP certifications. Data from 2 verified Indian Customs (DGFT) shipment records.
Top Pharmaceutical Products — Ethiopia to India Trade Routes
These are the top pharmaceutical products exported from India to Ethiopia, each with a dedicated trade route analysis page. Click any product to see detailed export data including Indian suppliers, Ethiopia buyers, regulatory requirements, and logistics for that specific product corridor. Products include Plasma ($25) — all finished pharmaceutical formulations verified from Indian Customs (DGFT) records.
Top Ethiopia Suppliers to India
1 Indian pharmaceutical companies export finished formulations to Ethiopia. Leading exporters include Armauer Hansen Research Institute, . The top exporter accounts for 100.0% of total India–Ethiopia pharma exports. Source: Indian Customs (DGFT).
Top Indian Buyers from Ethiopia
1 companies in Ethiopia import pharmaceutical formulations from India. Top buyers include Cliantha Research Limited, . The largest buyer accounts for 100.0% of India–Ethiopia pharma imports. Source: Indian Customs (DGFT).
Trade Statistics
Other Import Origins
Product Routes
India–Ethiopia Pharmaceutical Trade Corridor Analysis
Historical evolution, India's market position, and recent developments
1Trade Corridor Evolution
The pharmaceutical trade corridor between Ethiopia and India has experienced modest activity from 2022 to 2026. Between 2022 and 2026, Ethiopia exported a total of $25 USD worth of finished pharmaceutical formulations to India, comprising two shipments. The sole supplier from Ethiopia during this period was the ARMAUER HANSEN RESEARCH INSTITUTE, which provided plasma products. The recipient in India was CLIANTHA RESEARCH LIMITED, which received both shipments. This limited trade volume indicates a nascent and underdeveloped trade relationship in the pharmaceutical sector between the two nations.
2Ethiopia's Role in India's Pharma Imports
Ethiopia's contribution to India's pharmaceutical imports is negligible, accounting for only $25 USD over the 2022-2026 period. This minimal share underscores Ethiopia's limited presence in India's pharmaceutical import landscape. The sole product imported from Ethiopia was plasma, highlighting the narrow scope of pharmaceutical categories involved in this trade.
3Recent Developments
Between 2024 and 2026, there were no significant policy changes, trade agreements, or regulatory harmonization efforts between Ethiopia and India in the pharmaceutical sector. The trade volume remained static, indicating a lack of substantial developments in this bilateral corridor during the specified period.
Ethiopia Regulatory Landscape for Indian Pharmaceutical Exports
Registration process, GMP requirements, import documentation
1CDSCO Registration Process
For Ethiopian companies aiming to export pharmaceutical products to India, obtaining registration with the Central Drugs Standard Control Organisation (CDSCO) is mandatory. The process involves several key steps:
1. Application Submission: Companies must register on the CDSCO SUGAM portal, providing details about the applicant and the products intended for import.
2. Document Submission: Essential documents include the Certificate of Pharmaceutical Product (COPP), Good Manufacturing Practice (GMP) certificate, free sale certificate, and a power of attorney.
3. Review and Approval: CDSCO reviews the application and may conduct inspections to ensure compliance with Indian standards.
4. License Issuance: Upon successful evaluation, CDSCO issues the necessary import license, authorizing the company to export pharmaceutical products to India.
The entire process can take several months, depending on the completeness of the application and the efficiency of the review process.
2GMP & Manufacturing Standards
India requires that manufacturing sites in Ethiopia adhere to Good Manufacturing Practice (GMP) standards as per WHO guidelines. Ethiopian manufacturers must obtain WHO prequalification, which involves demonstrating compliance with international quality standards. CDSCO may conduct inspections to verify adherence to these standards before granting approval for importation into India.
3Import Documentation
To import pharmaceutical products into India, Ethiopian companies must provide:
- Import License (Form 10): Issued by CDSCO, authorizing the import of specific pharmaceutical products.
- Registration Certificate: Confirms that the product is registered and approved for sale in the country of origin.
- No Objection Certificate (NOC): Indicates that the exporting country's regulatory authority has no objection to the export of the product.
- Test License: Allows for the importation of products for testing or analysis purposes.
Additionally, compliance with Indian customs procedures, including proper labeling and packaging, is essential for smooth importation.
Product Categories & Therapeutic Trends — India to Ethiopia
Dominant categories, emerging opportunities, and demand drivers
1Dominant Import Categories
The pharmaceutical imports from Ethiopia to India are limited to plasma products, with a total value of $25 USD over the 2022-2026 period. This singular product category indicates a lack of diversity in the types of pharmaceutical products imported from Ethiopia to India.
2Innovation & Specialty Imports
Given the minimal trade volume and limited product range, there is currently no significant importation of innovator drugs, novel formulations, or advanced therapies from Ethiopia to India. The trade is predominantly restricted to basic pharmaceutical products like plasma.
3Import Demand Drivers
The negligible import volume from Ethiopia to India suggests that factors such as patent protection, technology gaps, quality requirements, and disease burden are not significant drivers for importing pharmaceutical products from Ethiopia. The limited trade may be attributed to other considerations, including cost-effectiveness and availability of alternative suppliers.
Trade Policy & Tariff Intelligence — India and Ethiopia
Tariff structure, trade agreements, IP and patent landscape
1India's Import Tariff Structure
India's import tariff structure for pharmaceutical formulations includes:
- Most Favored Nation (MFN) Tariff Rates: Standard rates applied to imports from all WTO member countries.
- Basic Customs Duty: A duty imposed on imported goods, varying based on product classification.
- Integrated Goods and Services Tax (IGST): A tax levied on imported goods, applicable at the time of importation.
- Health Cess: An additional levy on imported medical devices and drugs, intended to fund health initiatives.
Specific duty rates and taxes depend on the product classification and prevailing government policies.
2Trade Agreements & Preferences
As of March 2026, there are no specific Free Trade Agreements (FTAs) or preferential duty rates between India and Ethiopia concerning pharmaceutical imports. Both countries are members of the World Trade Organization (WTO), which provides a framework for trade relations, but no bilateral agreements have been established to facilitate pharmaceutical trade.
3IP, Patents & Price Control
India's patent regime, particularly Section 3(d) of the Indian Patents Act, aims to prevent evergreening of patents and ensures that only genuine innovations receive patent protection. Compulsory licensing provisions allow the government to grant licenses to third parties under certain conditions, such as public health emergencies. The National Pharmaceutical Pricing Authority (NPPA) regulates the prices of essential drugs, including imported pharmaceuticals, to ensure affordability and accessibility.
Supply Chain & Logistics — India to Ethiopia Pharma Shipments
Shipping routes, port infrastructure, cold chain compliance
1Shipping Routes & Transit
Pharmaceutical shipments from Ethiopia to India are typically transported via sea routes, with transit times varying based on the specific ports of departure and arrival. Air freight is also an option, offering faster delivery times but at higher costs. The reliability of these routes depends on factors such as weather conditions, geopolitical stability, and port efficiency.
2Port Infrastructure
Ethiopia's primary export ports include the Port of Djibouti, which handles a significant portion of the country's international trade. In India, major import ports include the Jawaharlal Nehru Port in Mumbai and the Chennai Port, both equipped to handle pharmaceutical shipments. The efficiency and capacity of these ports are crucial for the timely and safe delivery of pharmaceutical products.
3Indian Customs & Cold Chain
Indian customs procedures for pharmaceutical imports involve:
- Customs Clearance: Ensuring that all import documentation is accurate and complete to facilitate smooth clearance.
- CDSCO Testing: Imported pharmaceutical products may be subject to testing by CDSCO to verify compliance with Indian standards.
- Cold Chain Compliance: Maintaining the required temperature conditions during storage and transportation to preserve product efficacy.
- Good Distribution Practice (GDP): Adhering to guidelines that ensure the quality and integrity of pharmaceutical products throughout the supply chain.
Market Opportunity Assessment — Ethiopia for Indian Pharma
Market size, healthcare system, growth outlook
1India's Import Dependency
Despite being a major pharmaceutical manufacturer, India imports certain specialized products, including plasma, to meet specific medical needs. The importation of plasma from Ethiopia may be driven by factors such as cost-effectiveness, availability, and the need to diversify sources.
2Ethiopia Companies in India
As of March 2026, there are no significant Ethiopian pharmaceutical companies operating in India. The limited trade volume suggests that Ethiopian companies have not established a substantial presence in the Indian market.
3Outlook & Self-Reliance
India's Atmanirbhar Bharat initiative and the Production-Linked Incentive (PLI) scheme aim to boost domestic manufacturing and reduce import dependency. While these efforts may lead to increased self-reliance, the demand for specialized products like plasma may continue to necessitate imports from countries like Ethiopia.
Competitive Landscape — India vs Other Pharmaceutical Suppliers to Ethiopia
Competing origins, India's edge, challenges and threats
1Competing Import Origins
India imports pharmaceutical products from various countries, including the USA, EU, China, Switzerland, and Japan. Ethiopia's share in this import landscape is minimal, with a total of $25 USD over the 2022-2026 period. This indicates that other countries are the primary suppliers of pharmaceutical products to India.
2Ethiopia's Competitive Advantages
Ethiopian pharmaceutical products may offer competitive advantages such as cost-effectiveness and the ability to meet specific quality standards. However, the limited trade volume suggests that these advantages have not been fully realized in the Indian market.
3Domestic Competition & Threats
Indian pharmaceutical manufacturers are continually developing alternatives to imported products, including
FAQ — India to Ethiopia Pharmaceutical Trade
What is the total value of India's pharmaceutical export to Ethiopia?
India exported pharmaceuticals worth $25 to Ethiopia across 2 verified shipments.
Who are the top Indian pharmaceutical exporters to Ethiopia?
1. ARMAUER HANSEN RESEARCH INSTITUTE — $25. Total: 1 suppliers.
Which companies in Ethiopia import pharmaceuticals from India?
1. CLIANTHA RESEARCH LIMITED — $25. 1 buyers total.
What pharmaceutical products does India export most to Ethiopia?
1. Plasma ($25, 100.0%)
Which ports handle pharmaceutical shipments from India to Ethiopia?
Export: . Import: .
Why does Ethiopia import pharmaceuticals from India?
India's cost-competitive generic drug manufacturing, WHO-GMP certified facilities, and broad product portfolio. This $25 corridor reflects quality compliance and pricing advantages.
What certifications do Indian pharmaceutical exporters need to supply Ethiopia?
WHO-GMP certification, EU GMP approval (for EU markets), product dossier registration (CTD format), and ICH guideline compliance.
What is the average shipment value for India to Ethiopia pharmaceutical trade?
$12 per consignment across 2 shipments.
How many Indian pharmaceutical companies export to Ethiopia?
1 Indian companies. Largest: ARMAUER HANSEN RESEARCH INSTITUTE with $25.
How can I find verified Indian pharmaceutical suppliers for Ethiopia?
TransData Nexus covers 1 active exporters with shipment history and trade values at transdatanexus.com.
Unlock the Full India to Ethiopia Pharmaceutical Export Dataset
Access complete shipment records, supplier intelligence, buyer histories, and price analytics for all 2 shipments.
Official References & Regulatory Resources
- WHO Essential Medicines List
- CDSCO India
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- Pharmexcil
Data sourced from Indian Customs (DGFT) records. Verify regulatory status with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Data Source: Indian Customs (DGFT) export shipping bill records covering all pharmaceutical shipments from India.
- 2.Country Matching: Shipments to Ethiopia identified using destination country codes from customs declarations.
- 3.Statistical Normalization: Values are statistically normalized to remove outlier transactions and ensure accurate market share representation.
- 4.Coverage: 2 verified shipments from 1 Indian exporters to 1 Ethiopia buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
1 Exporters
1 buyers tracked
Expert-Reviewed
By pharmaceutical trade specialists