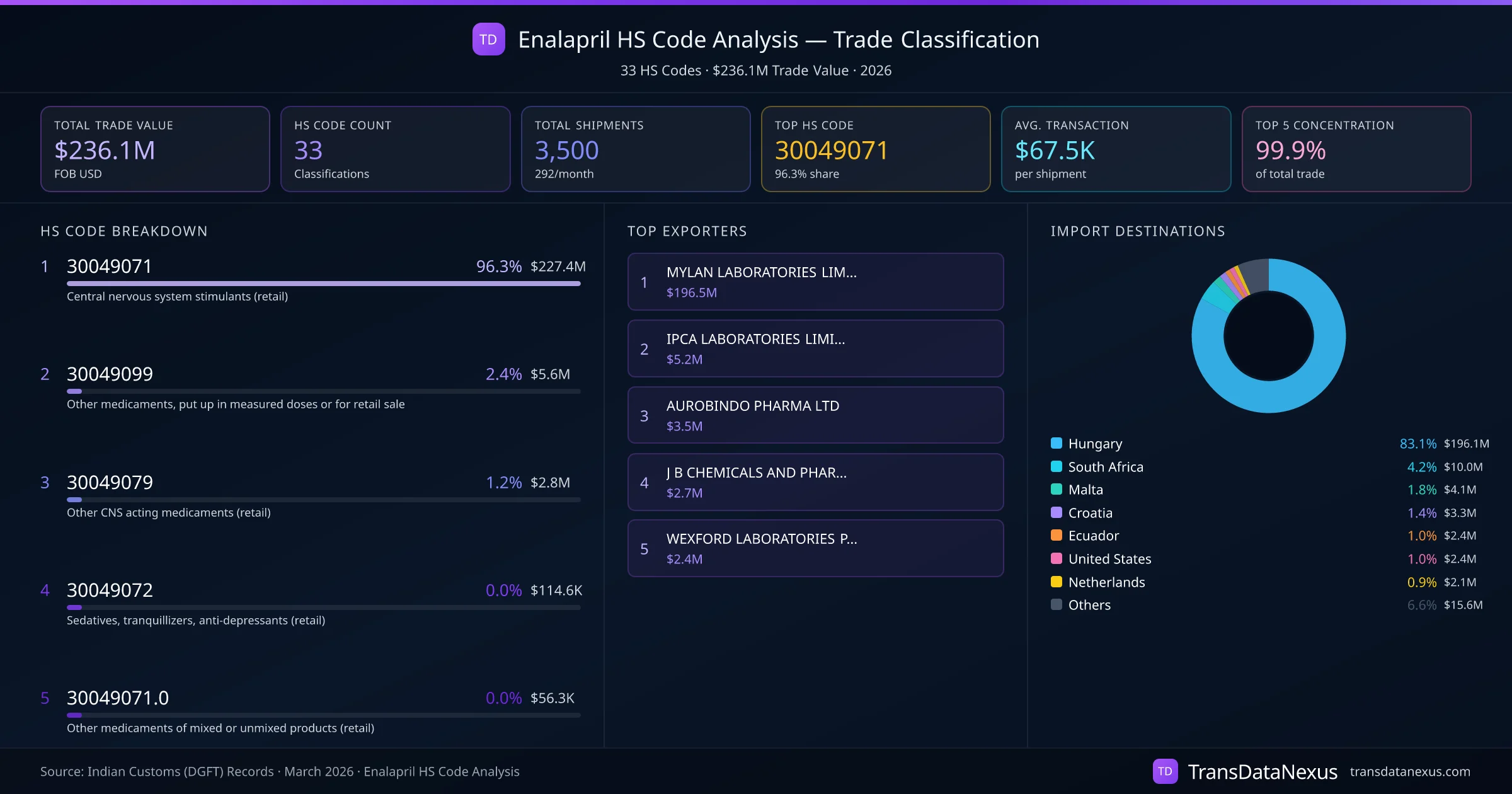
Enalapril, an angiotensin-converting enzyme (ACE) inhibitor used to treat hypertension and heart failure, is primarily classified under HS code 30049071. This code pertains to medicaments containing alkaloids or derivatives, excluding those containing ephedrine, pseudoephedrine, or norephedrine, and is specific to the Indian tariff schedule. In other countries, Enalapril is generally classified under HS code 300490, which covers "Other medicaments consisting of mixed or unmixed products for therapeutic or prophylactic uses, put up in measured doses or in forms or packings for retail sale."
Hungary, Malta, Croatia, Netherlands, and Finland (European Union Members):
As members of the European Union (EU), these countries adhere to the EU's Common External Tariff. The Most Favored Nation (MFN) tariff rate for products under HS code 300490 is 0%, indicating that imports of Enalapril are duty-free. Additionally, the EU maintains a list of essential medicines, which often benefit from tariff exemptions or reductions to ensure affordability and accessibility. Enalapril is included in the World Health Organization's (WHO) Model List of Essential Medicines, and the EU's tariff policies align with such classifications to facilitate the importation of critical pharmaceuticals.
South Africa:
South Africa's MFN tariff rate for HS code 300490 is 0%, allowing duty-free importation of Enalapril. South Africa is a member of the Southern African Development Community (SADC), which has a Free Trade Agreement (FTA) with India. Under this agreement, certain pharmaceutical products may benefit from preferential tariff rates. However, specific details regarding Enalapril would require consultation with the South African Revenue Service (SARS) or the SADC Secretariat. South Africa also maintains a list of essential medicines, and Enalapril's inclusion could further support duty-free status.