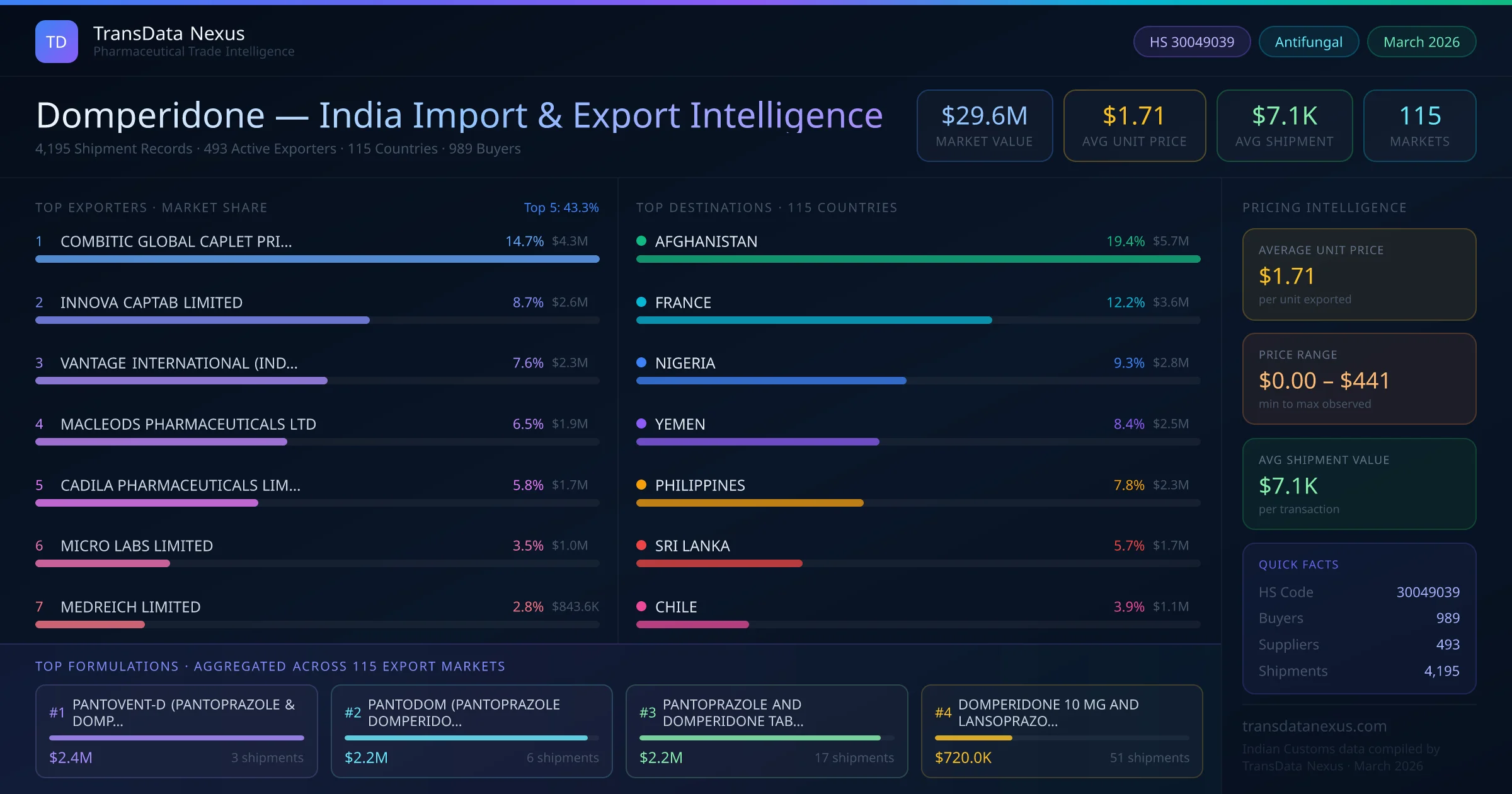
Importing finished pharmaceutical formulations containing Domperidone into India requires compliance with the Drugs and Cosmetics Act, 1940, and associated rules. The Central Drugs Standard Control Organization (CDSCO) mandates that all imported drugs possess a valid import license issued under Form 10 of the Drugs and Cosmetics Rules, 1945. Additionally, each product must be registered with CDSCO under Form 41, which serves as authorization for importation. Importers must also obtain an Importer Exporter Code (IEC) from the Directorate General of Foreign Trade (DGFT) to facilitate customs clearance. The registration process involves submitting comprehensive data on the drug's safety, efficacy, and quality, including stability studies in accordance with ICH guidelines. The timeline for obtaining these approvals can vary, but it typically ranges from several months to over a year, depending on the complexity of the product and the efficiency of the regulatory bodies. For formulations under HS Code 30049039, specific requirements include detailed product dossiers, manufacturing licenses from the country of origin, and a No Objection Certificate (NOC) from the manufacturer authorizing the import. It's crucial to ensure that all documentation is current and complies with Indian regulations to avoid delays or rejections.