Intas Pharmaceuticals Limited — Cytarabine Exporter Profile
Indian Pharmaceutical Exporter · #1 for Cytarabine · $5.1M export value · DGFT Verified
Intas Pharmaceuticals Limited is the #1 Indian exporter of Cytarabine with $5.1M in export value and 693 verified shipments. Intas Pharmaceuticals Limited holds a 47.6% market share in Cytarabine exports across 14 countries. The company exports 75 pharmaceutical products worth $372.3M across 19 therapeutic categories.
Intas Pharmaceuticals Limited — Cytarabine Export Profile: Buyers & Destinations
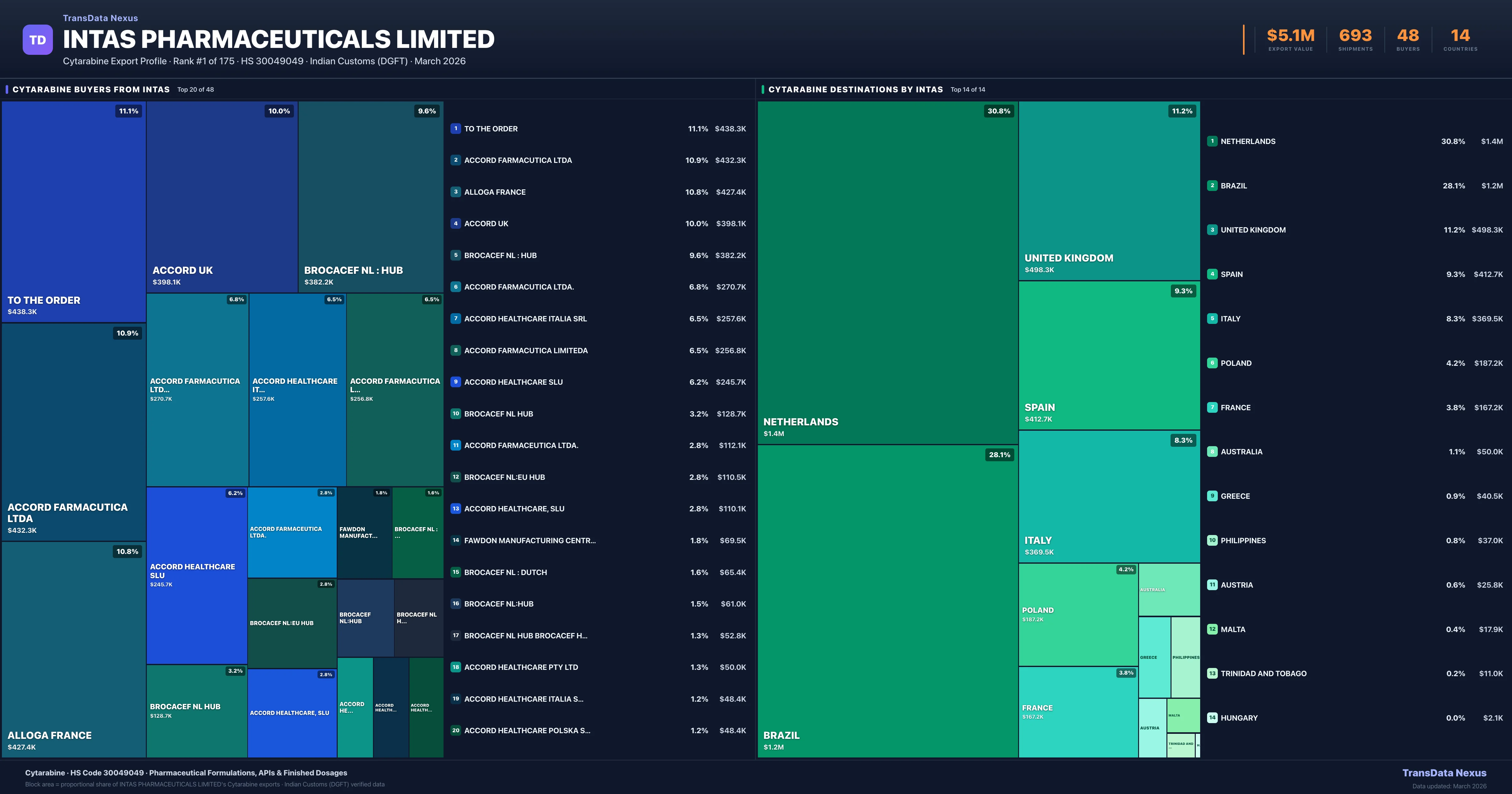
Where Does Intas Pharmaceuticals Limited Export Cytarabine?
| Country | Value | Shipments | Share |
|---|---|---|---|
| NETHERLANDS | $1.4M | 164 | 30.8% |
| BRAZIL | $1.2M | 39 | 28.1% |
| UNITED KINGDOM | $498.3K | 57 | 11.2% |
| SPAIN | $412.7K | 27 | 9.3% |
| ITALY | $369.5K | 39 | 8.3% |
| POLAND | $187.2K | 34 | 4.2% |
| FRANCE | $167.2K | 12 | 3.8% |
| AUSTRALIA | $50.0K | 1 | 1.1% |
| GREECE | $40.5K | 2 | 0.9% |
| PHILIPPINES | $37.0K | 9 | 0.8% |
Intas Pharmaceuticals Limited exports Cytarabine to 14 countries. The largest destination is NETHERLANDS accounting for 30.8% of Intas Pharmaceuticals Limited's Cytarabine shipments, followed by BRAZIL (28.1%) and UNITED KINGDOM (11.2%). These destinations reflect Intas Pharmaceuticals Limited's established distribution network and regulatory approvals in key markets.
Who Are the Top Buyers of Cytarabine from Intas Pharmaceuticals Limited?
| Buyer | Country | Value | Orders |
|---|---|---|---|
| TO THE ORDER | UNITED KINGDOM | $438.3K | 62 |
| ACCORD FARMACUTICA LTDA | BRAZIL | $432.3K | 13 |
| ALLOGA FRANCE | NETHERLANDS | $427.4K | 36 |
| ACCORD UK | UNITED KINGDOM | $398.1K | 25 |
| BROCACEF NL : HUB | NETHERLANDS | $382.2K | 42 |
| ACCORD FARMACUTICA LTDA. | BRAZIL | $270.7K | 9 |
| ACCORD HEALTHCARE ITALIA SRL | ITALY | $257.6K | 22 |
| ACCORD FARMACUTICA LIMITEDA | BRAZIL | $256.8K | 7 |
| ACCORD HEALTHCARE SLU | SPAIN | $245.7K | 15 |
| BROCACEF NL HUB | NETHERLANDS | $128.7K | 27 |
Intas Pharmaceuticals Limited supplies Cytarabine to 48 buyers globally. The largest buyer is TO THE ORDER (UNITED KINGDOM), followed by ACCORD FARMACUTICA LTDA (BRAZIL) and ALLOGA FRANCE (NETHERLANDS). These buyers represent pharmaceutical distributors, hospital groups, and government procurement agencies in their respective markets.
What Is India's Total Cytarabine Export Value and How Much Does Intas Pharmaceuticals Limited Contribute?
India exported $9.8M worth of Cytarabine through 1,825 shipments from 175 suppliers to 110 countries, serving 381 buyers globally. Intas Pharmaceuticals Limited contributes $5.1M to this total, accounting for 47.6% of India's Cytarabine exports. Intas Pharmaceuticals Limited ships Cytarabine to 14 countries through 48 buyers.
What Is the Average Shipment Value for Intas Pharmaceuticals Limited's Cytarabine Exports?
Intas Pharmaceuticals Limited's average Cytarabine shipment value is $7.4K per consignment, based on 693 shipments totaling $5.1M. The largest destination is NETHERLANDS (30.8% of Intas Pharmaceuticals Limited's Cytarabine exports).
How Does Intas Pharmaceuticals Limited Compare to Other Indian Cytarabine Exporters?
Intas Pharmaceuticals Limited ranks #1 among 175 Indian Cytarabine exporters with a 47.6% market share. The top 3 exporters are INTAS PHARMACEUTICALS LIMITED ($5.1M), FRESENIUS KABI ONCOLOGY LIMITED ($1.8M), NAPROD LIFE SCIENCES PRIVATE LIMITED ($557.0K). Intas Pharmaceuticals Limited processed 693 shipments to 14 destination countries.
What Cytarabine Formulations Does Intas Pharmaceuticals Limited Export?
| Product Description / Formulation | Value | Shipments |
|---|---|---|
| PHARMA DRUGS & MEDI CYTARABINE INJ,100MG | $357.5K | 40 |
| PHARMA DRUGS & MEDI CYTARABINE INJ BP 1000MG/10 ML VIAL AS PER INVOICE | $182.9K | 4 |
| PHARMA DRUGS MEDI CYTARABINE INJ BP 1000MG 10 ML VIAL AS PER INVOICE | $148.6K | 3 |
| PHARMA DRUG&MED:CYTARABINE INJ,100MG/ML, | $144.4K | 28 |
| PHARMA DRUG&MED:CYTARABINE INJ 100MG/ML, | $139.0K | 8 |
| PHARMA DRUGS AND MEDI CYTARABINE INJ BP1000MG/10 ML VIAL AS PER INVNOS | $136.4K | 3 |
| PHARMA DRUGS AND MEDI CYTARABINE INJ BP1000MG/10 ML VIAL AS PER INV | $115.3K | 3 |
| PHARMA DRUG&MED:CYTARABINE INJ BP 1000MG | $100.0K | 2 |
| PHARMA DRUGS & MEDI CYTARABINE INJ,100MG/ML,20MLX1V(ACCO-FR AS PER INVOICE | $96.0K | 3 |
| PHARMA DRUGS & MEDI CYTARABINE INJ 100MG/ML,20ML(ACCORD-UK AS PER INVOICE | $79.5K | 3 |
Intas Pharmaceuticals Limited exports 402 distinct Cytarabine formulations including tablets, capsules, syrups, and combination drugs. The top formulation is PHARMA DRUGS & MEDI CYTARABINE INJ,100MG with 40 shipments worth $357.5K. Notable combination products include fixed-dose combinations with other active ingredients, meeting diverse global regulatory requirements.
Regulatory Requirements: Exporting Cytarabine to Key Markets
What Intas Pharmaceuticals Limited must comply with to export Cytarabine to its top destination countries
Brazil — ANVISA
Approval Process
Generic registration under RDC 200/2017. ANVISA GMP inspection of Indian facility mandatory before approval.
Timeline: 12–24 months
GMP & Export Requirements
ANVISA GMP certificate (CBPF) for each manufacturing site; on-site inspection by ANVISA
Certificate of Pharmaceutical Product from CDSCO; Import Authorization via SISCOMEX
Note: ANVISA maintains active Indian pharma facility inspection program. Inspection backlog can extend timelines.
United Kingdom — MHRA
Approval Process
Post-Brexit UK Marketing Authorisation via national procedure. Separate from EU approval since January 2021.
Timeline: 12–15 months
GMP & Export Requirements
UK GMP compliance; MHRA inspection of manufacturing site
Manufacturer Import Authorisation (MIA) held by UK importer; Qualified Person (QP) batch release
Note: Post-Brexit: EU GMP certificates no longer automatically valid in UK. Separate MHRA recognition required.
Regulatory information is provided for general reference based on publicly available guidelines. Requirements vary by formulation, dosage form, and therapeutic classification. Exporters should consult qualified regulatory affairs professionals for case-specific guidance.
How Does Intas Pharmaceuticals Limited Compare to Nearest Cytarabine Exporters?
Exporters ranked immediately above and below #1 by export value
| # | Supplier | Export Value | Shipments | Countries | Avg/Shipment |
|---|---|---|---|---|---|
| 1 | INTAS PHARMACEUTICALS LIMITED ★ | $5.1M | 693 | 14 | $7.4K |
| 2 | FRESENIUS KABI ONCOLOGY LIMITED | $1.8M | 98 | 13 | $18.5K |
| 3 | NAPROD LIFE SCIENCES PRIVATE LIMITED | $557.0K | 52 | 8 | $10.7K |
Intas Pharmaceuticals Limited ranks #1 among 175 Indian Cytarabine exporters. Average shipment value of $7.4K compared to the market average of $56.0K. The closest competitors by value are FRESENIUS KABI ONCOLOGY LIMITED and NAPROD LIFE SCIENCES PRIVATE LIMITED.
Which Indian Ports Ship Cytarabine Exports?
| Port | Shipments | % Share |
|---|---|---|
| SAHAR AIR CARGO ACC (INBOM4) | 249 | 13.6% |
| SAHAR AIR | 242 | 13.3% |
| DELHI AIR | 159 | 8.7% |
| AHEMDABAD AIR ACC (INAMD4) | 145 | 7.9% |
| DELHI AIR CARGO ACC (INDEL4) | 144 | 7.9% |
| AHEMDABAD AIR | 142 | 7.8% |
| MUNDRA SEA (INMUN1) | 139 | 7.6% |
| MUNDRA SEA | 111 | 6.1% |
What Other Advanced Oncology Products Does Intas Pharmaceuticals Limited Export?
Intas Pharmaceuticals Limited also exports these advanced oncology products. Each links to the detailed product page.
Geopolitical & Trade Policy Impact on Intas Pharmaceuticals Limited's Cytarabine Exports
The current geopolitical landscape presents both challenges and opportunities for Indian pharmaceutical exporters like Intas Pharmaceuticals. The Israel-Iran tensions have led to increased security risks in the Strait of Hormuz and the Red Sea–Suez Canal corridor, prompting major carriers to reroute shipments around the Cape of Good Hope. This detour has extended transit times by 10–20 days and raised freight rates by 40–50% on key India–Europe routes, impacting the cost-effectiveness and timeliness of exports. (livemint.com)
Conversely, the ongoing US-China trade tensions have opened avenues for Indian pharmaceutical companies to capture a larger share of the U.S. market. Intas's acquisition of UDENYCA® and the FDA's acceptance of its biosimilar applications position the company to leverage these opportunities effectively. However, compliance with the European Union's Falsified Medicines Directive (FMD) remains a critical factor for market access. Intas's proactive approach in securing partnerships and ensuring regulatory compliance underscores its commitment to maintaining and expanding its presence in the European market.
Intas Pharmaceuticals Limited — Regulatory Compliance & Quality Standards
Maintaining stringent regulatory compliance is paramount for Indian pharmaceutical exporters. Intas Pharmaceuticals has demonstrated a strong commitment to adhering to international quality standards, as evidenced by its successful acquisitions and partnerships that necessitate compliance with FDA, WHO, and EU Good Manufacturing Practices (GMP). The company's proactive engagement in regulatory processes, such as the FDA's acceptance of its Biologics License Application for DMB-3115, reflects its dedication to quality and compliance. (intaspharma.com) As global regulatory requirements continue to evolve, Intas's focus on quality assurance and compliance positions it favorably in the competitive pharmaceutical export market.
About Intas Pharmaceuticals Limited
Intas Pharmaceuticals Limited exports 75 products worth $372.3M. Beyond Cytarabine, top products include Ras, Mycophenolate, Tacrolimus, Apixaban, Ezetimibe. View the complete Intas Pharmaceuticals Limited profile for full portfolio analysis.
Market Intelligence
Latest developments and market context
Detailed market intelligence for Cytarabine — including regulatory updates, trade policy changes, and competitive landscape analysis — will be available in an upcoming update. This section will feature AI-researched insights from verified industry sources, government notifications, and market reports.
Official References & Regulatory Resources
- CDSCO India
- Pharmexcil
- IBEF — India Pharma Industry
- Ministry of Commerce — Pharma Exports
- India Trade Statistics (DGFT)
Verify manufacturer licensing and export certifications with the official agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Product Matching: Cytarabine shipments identified using fuzzy matching on DGFT product description fields, validated against Indian Pharmacopoeia nomenclature.
- 2.Supplier Identification: Intas Pharmaceuticals Limited matched across shipments using name normalization, accounting for company name formatting variations.
- 3.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation. This removes the impact of unusually large one-off transactions that could distort company rankings.
- 4.Verified Records: This page is backed by 705 individual customs records matching Intas Pharmaceuticals Limited exporting Cytarabine, covering 402 formulations to 14 countries via 48 buyers.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
Global Coverage
Exports to 110+ countries, 381+ buyers
Expert-Reviewed
Analyzed by pharmaceutical trade specialists
Get Cytarabine Export Data from Intas Pharmaceuticals Limited
Access shipment-level details, pricing data, buyer connections, and competitive analysis for Intas Pharmaceuticals Limited's Cytarabine exports. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Intas Pharmaceuticals Limited
Full Company Profile →
75 products · $372.3M total trade · 19 categories
Cytarabine Stats
Company Overview
Top Products by Intas Pharmaceuticals Limited
Related Analysis
Need Detailed Export Data?
Access shipment-level records, pricing trends, and buyer contacts for Intas Pharmaceuticals Limited.
Request DemoView Pricing →Data Source
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Market share calculated relative to total Indian pharmaceutical exports for Cytarabine. For current shipment-level data, contact TransData Nexus.