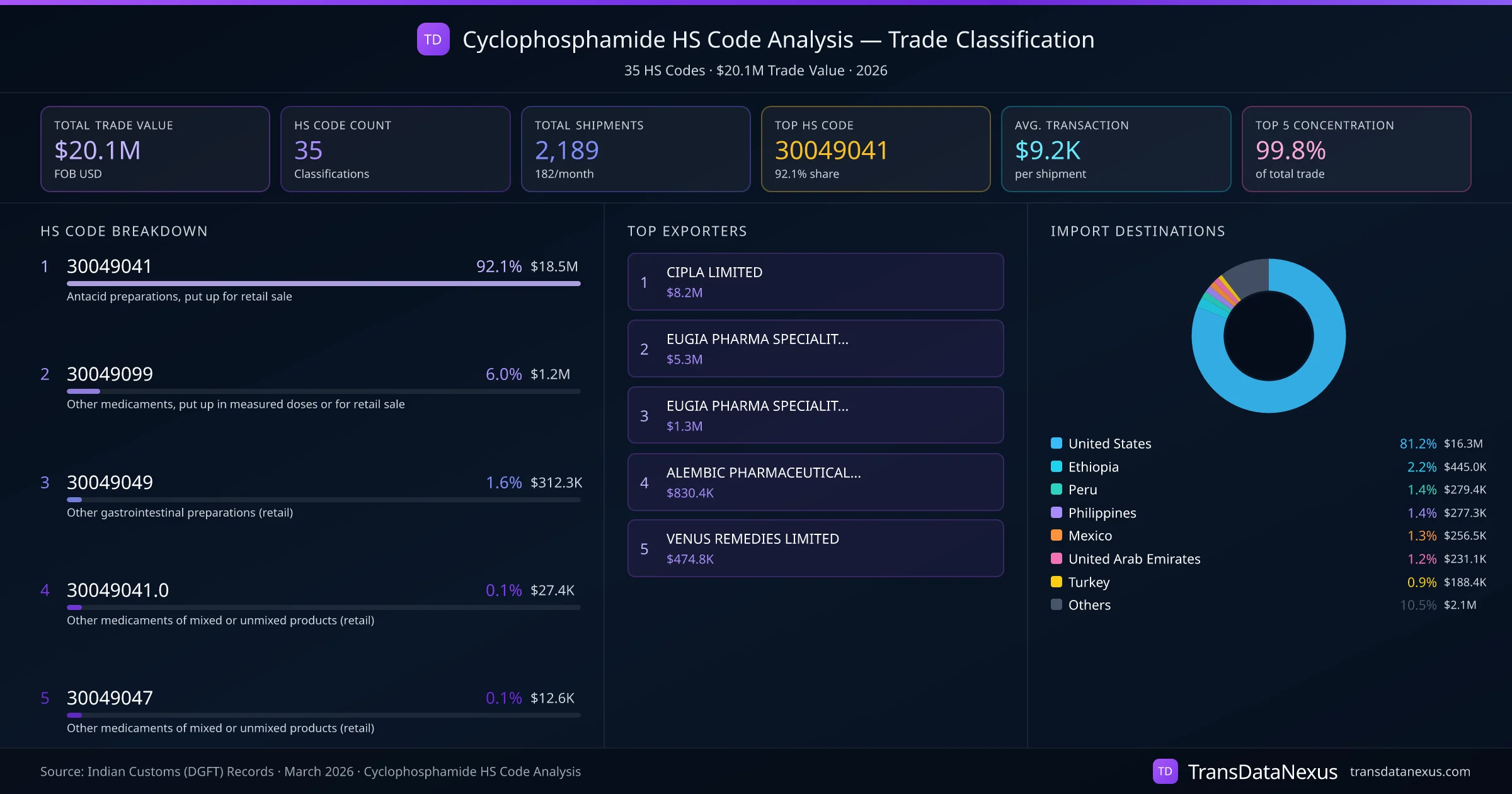
Cyclophosphamide, classified under HS code 30049041, is subject to varying tariff rates and exemptions across key importing countries.
United States: The Harmonized Tariff Schedule of the United States (HTSUS) lists HS code 300490 as "Other medicaments of mixed or unmixed products, for therapeutic or prophylactic uses, put up in measured doses or in forms or packings for retail sale." The Most Favored Nation (MFN) duty rate for this category is 0%. Additionally, the U.S. Food and Drug Administration (FDA) includes Cyclophosphamide in its list of essential medicines, which may qualify for duty-free treatment under specific provisions.
Ethiopia: According to the Ethiopian Revenue and Customs Authority, pharmaceutical products under HS code 300490 are generally exempt from import duties, aligning with the country's policy to facilitate access to essential medicines.
Peru: The Peruvian Customs Administration (SUNAT) applies an MFN tariff rate of 0% on pharmaceutical products classified under HS code 300490. Under the Peru-India Trade Agreement, preferential rates may apply, potentially offering further reductions or exemptions for Cyclophosphamide imports.
Philippines: The Philippine Tariff Commission specifies that medicaments under HS code 300490 are subject to an MFN tariff rate of 0%. The Philippines' Bureau of Customs also provides exemptions for essential medicines, which likely include Cyclophosphamide.
Mexico: The Mexican Tariff Schedule indicates that products under HS code 300490 are subject to an MFN tariff rate of 0%. The Mexico-India Preferential Trade Agreement may offer additional benefits, potentially reducing tariffs or providing exemptions for essential medicines like Cyclophosphamide.
United Arab Emirates (UAE): The UAE Federal Customs Authority applies an MFN tariff rate of 0% on pharmaceutical products under HS code 300490. The UAE's Ministry of Health and Prevention lists Cyclophosphamide as an essential medicine, which may qualify for duty exemptions.